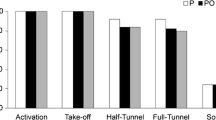
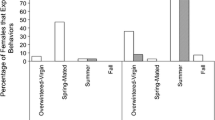
Abstract
Mating might significantly affect the host selection behaviors of phytophagous insects. Here, we investigated the post-mating changes in behavioral and antennal responses of Pieris rapae (Lepidoptera: Pieridae) females to host plant volatiles. In two-choice bioassays using artificial plant models, mated females visited the model scented with synthetic blends (15-, 9-, or 6-components) of cabbage plant volatiles more frequently than the unscented control, whereas virgin females did not exhibit this preference. Because single compounds and the 3-component blend did not induce preferential visiting, mated females apparently utilized complex odor blends as their host-finding cue. Moreover, 2- to 4-day-old mated females visited the models, scented and unscented, more frequently than did their virgin counterparts. Therefore, mating enhanced the host-finding behavior of young females and their responsiveness to plant volatiles. Gas chromatography–electroantennographic detector analysis revealed that eight of the 15 compounds in the cabbage plant volatiles elicited responses from female antennae. However, post-mating and age-dependent changes in antennal responses were not detected. Because female peripheral (antennal) sensitivity to volatiles remained practically unchanged after emergence, post-mating changes in host selection might be attributed to changes in the central nervous system.



Similar content being viewed by others
References
Anton S, Dufour M-C, Gadenne C (2007) Plasticity of olfactory guided behavior and its neurobiological basis: lessons from moths and locusts. Entomol Exp Appl 123:1–11. https://doi.org/10.1111/j.1570-7458.2007.00516.x
Bartlet E, Blight MM, Hick AJ, Williams IH (1993) The responses of the cabbage seed weevil (Ceutorhynchus assimilis) to the odour of oilseed rape (Brassica napus) and to some volatile isothiocyanates. Entomol Exp Appl 68:295–302. https://doi.org/10.1046/j.1570-7458.1997.00256.x
Baur R, Feeny P (1994) Comparative electrophysiological analysis of plant odor perception in females of three Papilio species. Chemoecology 5:26–36. https://doi.org/10.1007/BF01259970
Baur R, Feeny P, Städler E (1993) Oviposition stimulants for the black swallowtail butterfly: identification of electrophysiologically active compounds in carrot volatiles. J Chem Ecol 19:919–937. https://doi.org/10.1007/BF00992528
Bronström G (2017) glmmML: Generalized linear models with clustering. R package version 1.0.2. http://CRAN.R-project.org/package=glmmML. Accessed 20 June 2017
Bruce TJA, Pickett JA (2011) Perception of plant volatile blends by herbivorous insects—finding the right mix. Phytochemistry 72:1605–1611. https://doi.org/10.1016/j.phytochem.2011.04.011
Bruce TJA, Wadhams LJ, Woodcock CM (2005) Insect host location: a volatile situation. Trends Plant Sci 10:269–274. https://doi.org/10.1016/j.tplants.2005.04.003
Cha DH, Nojima S, Hesler SP, Zhang A, Linn CE Jr, Roelofs WL, Loeb GM (2008) Identification and field evaluation of grape shoot volatiles attractive to female grape berry moth (Paralobesia viteana). J Chem Ecol 34:1180–1189. https://doi.org/10.1007/s10886-008-9517-0
de Bruyne M, Baker TC (2008) Odor detection in insects: volatile codes. J Chem Ecol 34:882–897. https://doi.org/10.1007/s10886-008-9485-4
de Pinho PG, Valentão P, Gonçalves RF, Sousa C, Seabra RM, Andrade PB (2009) Volatile composition of Brassica oleracea L. var. costata DC leaves using solid-phase microextraction and gas chromatography/ion trap mass spectrometry. Rapid Commun Mass Sp 23:2292–2300. https://doi.org/10.1002/rcm.4148
Fadamiro H, Chen L, Akotsen-Mensah C, Setzer WN (2010) Antennal electrophysiological responses of the giant swallowtail butterfly, Papilio cresphontes, to the essential oils of Zanthoxylum clava-herculis and related plants. Chemoecology 20:25–33. https://doi.org/10.1007/s00049-009-0039-1
Feeny P, Städler E, Åhman I, Carter M (1989) Effects of plant odor on oviposition by the black swallowtail butterfly, Papilio polyxenes (Lepidoptera: Papilionidae). J Insect Behav 2:803–827. https://doi.org/10.1007/BF01049402
Finch S (1978) Volatile plant chemicals and their effect on host plant finding by the cabbage root fly (Delia brassicae). Entomol Exp Appl 24:150–159. https://doi.org/10.1111/j.1570-7458.1978.tb02793.x
Fraser AM, Mechaber WL, Hildebrand JG (2003) Electroantennographic and behavioral responses of the sphinx moth Manduca sexta to host plant headspace volatiles. J Chem Ecol 29:1813–1833. https://doi.org/10.1023/A:1024898127549
Gadenne C, Barrozo RB, Anton S (2016) Plasticity in insect olfaction: to smell or not to smell? Annu Rev Entomol 61:317–333. https://doi.org/10.1146/annurev-ento-010715-023523
Geervliet JBF, Posthumus MA, Vet LEM, Dicke M (1997) Comparative analysis of headspace volatiles from different caterpillar-infested or uninfested food plants of Pieris species. J Chem Ecol 23:2935–2954. https://doi.org/10.1023/A:1022583515142
Gols R, Bullock JM, Dicke M, Bukovinszky T, Harvey JA (2011) Smelling the wood from the trees: non-linear parasitoid responses to volatile attractants produced by wild and cultivated cabbage. J Chem Ecol 37:795–807. https://doi.org/10.1007/s10886-011-9993-5
Gossard TW, Jones RE (1977) The effects of age and weather on egg-laying in Pieris rapae L. J Appl Ecol 14:65–71
Honda K, Ômura H, Hayashi N (1998) Identification of floral volatiles from Ligustrum japonicum that stimulate flower-visiting by cabbage butterfly, Pieris rapae. J Chem Ecol 24:2167–2180. https://doi.org/10.1023/A:1020750029362
Hopkins RJ, van Loon JJA (2001) The effect of host acceptability on oviposition and egg accumulation by the small white butterfly, Pieris rapae. Physiol Entomol 26:149–157. https://doi.org/10.1046/j.1365-3032.2001.00228.x
Ikeura H, Kobayashi F, Hayata Y (2010) How do Pieris rapae search for Brassicaceae host plants? Biochem Syst Ecol 38:1199–1203. https://doi.org/10.1016/j.bse.2010.12.007
Li J, Wakui R, Tebayashi S, Kim C-S (2010) Volatile attractants for the common bluebottle, Graphium sarpedon nipponum, from the host, Cinnamomum camphora. Biosci Biotech Biochem 74:1987–1990. https://doi.org/10.1271/bbb.100118
Martel V, Andersson P, Hansson BS, Schlyter F (2009) Peripheral modulation of olfaction by physiological state in the Egyptian leaf worm Spodoptera littoralis (Lepidoptera: Noctuidae). J Insect Physiol 55:793–797. https://doi.org/10.1016/j.jinsphys.2009.04.012
Mercader RJ, Stelinski LL, Scriber JM (2008) Differential antennal sensitivities of the generalist butterflies Papilio glaucus and P. canadensis to host plant and non-host plant extracts. J Lepid Soc 62:84–88
Myers JH (1985) Effect of physiological condition of the host plant on the ovipositional choice of the cabbage white butterfly, Pieris rapae. J Anim Ecol 54:193–204
Ômura H, Honda K, Hayashi N (1999) Chemical and chromatic bases for preferential visiting by the cabbage butterfly, Pieris rapae, to rape flowers. J Chem Ecol 25:1895–1906. https://doi.org/10.1023/A:1020990018111
Ômura H, Honda K, Hayashi N (2000) Floral scent of Osmanthus fragrans discourages foraging behavior of cabbage butterfly, Pieris rapae. J Chem Ecol 26:655–666. https://doi.org/10.1023/A:1005424121044
Pearson GA, Schal C (1999) Electroantennogram responses of both sexes of grape root borer (Lepidoptera: Sesiidae) to synthetic female sex pheromone. Environ Entomol 28:943–946. https://doi.org/10.1093/ee/28.6.943
Pinto CF, Troncoso AJ, Urzúa A, Niemeyer M (2009) Use of volatiles of Aristolochia chilensis (Aristolochiaceae) in host searching by fourth-instar larvae and adults of Battus polydamas archidamas (Lepidoptera: Papilionidae: Troidini). Eur J Entomol 106:63–68. https://doi.org/10.14411/eje.2009.010
R Core Team (2016) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rajapakse CNK, Walter GH, Moore CJ, Hull CD, Cribb BW (2006) Host recognition by a polyphagous lepidopteran (Helicoverpa armigera): primary host plants, host produced volatiles and neurosensory stimulation. Physiol Entomol 31:270–277. https://doi.org/10.1111/j.1365-3032.2006.00517.x
Reddy GVP, Guerrero A (2000) Behavioral responses of the diamondback moth, Plutella xylostella, to green leaf volatiles of Brassica oleracea subsp. capitata. J Agric Food Chem 48:6025–6029. https://doi.org/10.1021/jf0008689
Riffell JA, Lei H, Christensen TA, Hildebrand JG (2009) Characterization and coding of behaviorally significant odor mixtures. Curr Biol 19:335–340. https://doi.org/10.1016/j.cub.2009.01.041
Saveer AM, Kromann SH, Birgersson G, Bengtsson M, Lindblom T, Balkenius A, Hansson BS, Witzgall P, Becher PG, Ignell R (2012) Floral to green: mating switches moth olfactory coding and preference. Proc R Soc B 279:2314–2322. https://doi.org/10.1098/rspb.2011.2710
Saxena KN, Goyal S (1978) Host-plant relations of the Citrus butterfly Papilio demoleus L.: orientational and ovipositional responses. Entomol Exp Appl 24:1–10. https://doi.org/10.1111/j.1570-7458.1978.tb02750.x
Schmidt-Büsser D, Von Arx M, Connétable S, Guerin PM (2011) Identification of host-plant chemical stimuli for the European grape berry moth Eupoecilia ambiguella. Physiol Entomol 36:101–110. https://doi.org/10.1111/j.1365-3032.2010.00766.x
Shu S, Grant GG, Langevin D, Lombardo DA, MacDonald L (1997) Oviposition and electroantennogram responses of Dioryctria abietivorella (Lepidoptera: Pyralidae) elicited by monoterpenes and enantiomers from eastern white pine. J Chem Ecol 23:35–50. https://doi.org/10.1023/B:JOEC.0000006344.18966.c6
Snell-Rood EC, Papaj DR (2009) Patterns of phenotypic plasticity in common and rare environments: a study of host use and color learning in the cabbage white butterfly Pieris rapae. Am Nat 173:615–631. https://doi.org/10.1086/597609
Späthe A, Reinecke A, Olsson SB, Kesavan S, Knaden M, Hansson BS (2013) Plant species- and status-specific odorant blends guide oviposition choice in the moth Manduca sexta. Chem Senses 38:147–159. https://doi.org/10.1093/chemse/bjs089
Suzuki Y (1978) Adult longevity and reproductive potential of the small cabbage white, Pieris rapae crucivora Boisduval (Lepidoptera: Pieridae). Appl Entomol Zool 13:312–313
Tasin M, Bäckman A-C, Bengtsson M, Ioriatti C, Witzgall P (2006) Essential host plant cues in the grapevine moth. Naturwissenschaften 93:141–144. https://doi.org/10.1007/s00114-005-0077-7
Tasin M, Bäckman A-C, Coracini M, Casado D, Ioriatti C, Witzgall P (2007) Synergism and redundancy in a plant volatile blend attracting grapevine moth females. Phytochemistry 68:203–209. https://doi.org/10.1016/j.phytochem.2006.10.015
Topazzini A, Mazza M, Pelosi P (1990) Electroantennogram responses of five Lepidoptera species to 26 general odourants. J Insect Physiol 36:619–624. https://doi.org/10.1016/0022-1910(90)90065-N
Traynier RMM (1986) Visual learnings in assays of sinigrin solution as an oviposition releaser for the cabbage butterfly, Pieris rapae. Entomol Exp Appl 40:25–33. https://doi.org/10.1111/j.1570-7458.1986.tb02151.x
van Loon JAA, Frentz WH, van Eeuwijk FA (1992) Electroantennogram responses to plant volatiles in two species of Pieris butterflies. Entomol Exp Appl 62:253–260. https://doi.org/10.1111/j.1570-7458.1992.tb00665.x
Visser JH (1986) Host odor perception in phytophagous insects. Annu Rev Entomol 31:121–144. https://doi.org/10.1146/annurev.en.31.010186.001005
Wedell N (2005) Female receptivity in butterflies and moths. J Exp Biol 208:3433–3440. https://doi.org/10.1242/jeb.01774
Yildizhan S, van Loon J, Sramkova A, Ayasse M, Arsene C, ten Broeke C, Schulz S (2009) Aphrodisiac pheromones from the wings of the small cabbage white and large cabbage white butterflies, Pieris rapae and Pieris brassicae. ChemBioChem 10:1666–1677. https://doi.org/10.1002/cbic.200900183
Acknowledgements
This study was supported by a Grant-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (JSPS) to HO (No. 16K08099).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Itoh, Y., Okumura, Y., Fujii, T. et al. Effects of mating on host selection by female small white butterflies Pieris rapae (Lepidoptera: Pieridae). J Comp Physiol A 204, 245–255 (2018). https://doi.org/10.1007/s00359-017-1237-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-017-1237-x