Abstract
Purpose
To evaluate the diagnostic accuracy of multiparametric magnetic resonance imaging (MRI)- and microultrasound (microUS)-guided targeted biopsy (TBx) in detecting prostate cancer (PCa) and clinically significant (cs) PCa among men with Prostate Imaging Reporting and Data System (PI-RADS 5) lesions and to compare this combined TBx (CTBx) strategy with CTBx plus systemic biopsy (SBx).
Methods
One hundred and thirty-six biopsy-naïve patients with PI-RADS 5 lesion at multiparametric MRI undergoing CTBx plus SBx were retrospectively evaluated. Analysis of diagnostic performance of microUS-TBx, MRI-TBx, CTBx, SBx and combined CTBx plus SBx was performed. Cost (downgrade, upgrade and biopsy core) to effectiveness (detection rate) was compared.
Results
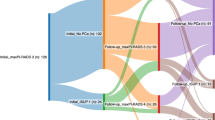
CTBx achieved a comparable detection rate to CTBx plus SBx in diagnosis of PCa and csPCa (PCa: 78.7% [107/136] vs 79.4% [108/136]; csPCa: 67.6% [92/136] vs 67.6% [92/136]; p > 0.05) and outperformed SBx (PCa: 58.8% [80/136]; csPCa: 47.8% [65/136]; p < 0.001). Using CTB would have avoided 41.1% (56/136) unnecessary SBx, without missing any csPCa. The rate of any upgrading or csPCa upgrading was significantly higher by SBx than by CTBx [33/65 (50.8%) vs 17/65 (26.1%) and 20/65 (30.8%) vs 4/65 (6.15%), respectively, p < 0.05]. Considering csPCa detection rate, microUS showed high sensitivity and positive predictive value (94.6%, 87.9%, respectively), with lower specificity and negative predictive value (25.0% and 44.4%, respectively). At multivariable logistic regression models, positive microUS was identified as an independent predictor of csPCa (p = 0.024).
Conclusions
A combined microUS/MRI-TBx approach could be the ideal imaging tool for characterizing primary disease in PI-RADS five patients, allowing SBx to be avoided.

Similar content being viewed by others
Data availability
Not Available.
References
Kasivisvanathan V, Rannikko AS, Borghi M et al (2018) MRI-targeted or standard biopsy for prostate-cancer diagnosis. N Engl J Med 378:1767–1777. https://doi.org/10.1056/NEJMoa1801993
Rouvière O, Puech P, Renard-Penna R et al (2019) Use of prostate systematic and targeted biopsy on the basis of multiparametric MRI in biopsy-naive patients (MRI-FIRST): a prospective, multicentre, paired diagnostic study. Lancet Oncol 20:100–109. https://doi.org/10.1016/S1470-2045(18)30569-2
Klotz L, Chin J, Black PC et al (2021) Comparison of multiparametric magnetic resonance imaging-targeted biopsy with systematic transrectal ultrasonography biopsy for biopsy-naive men at risk for prostate cancer: a phase 3 randomized clinical trial. JAMA Oncol 7:534. https://doi.org/10.1001/jamaoncol.2020.7589
Ahmed HU, El-Shater Bosaily A, Brown LC et al (2017) Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): a paired validating confirmatory study. Lancet 389:815–822. https://doi.org/10.1016/S0140-6736(16)32401-1
Hugosson J, Månsson M, Wallström J et al (2022) Prostate cancer screening with PSA and MRI followed by targeted biopsy only. N Engl J Med 387:2126–2137. https://doi.org/10.1056/NEJMoa2209454
Preisser F, Theissen L, Wenzel M et al (2021) Performance of combined magnetic resonance imaging/ultrasound fusion-guided and systematic biopsy of the prostate in biopsy-naïve patients and patients with prior biopsies. Eur Urol Focus 7:39–46. https://doi.org/10.1016/j.euf.2019.06.015
Ahdoot M, Wilbur AR, Reese SE et al (2020) MRI-targeted, systematic, and combined biopsy for prostate cancer diagnosis. N Engl J Med 382:917–928. https://doi.org/10.1056/NEJMoa1910038
Cornford P, van den Bergh RCN, Briers E et al (2021) EAU-EANM-ESTRO-ESUR-SIOG guidelines on prostate cancer. Part II—2020 update: treatment of relapsing and metastatic prostate cancer. Eur Urol 79:263–282. https://doi.org/10.1016/j.eururo.2020.09.046
Eastham JA, Boorjian SA, Kirkby E (2022) Clinically Localized Prostate Cancer: AUA/ASTRO Guideline. J Urol 208:505–507. https://doi.org/10.1097/JU.0000000000002854
Dariane C, Ploussard G, Barret E et al (2022) Micro-ultrasound-guided biopsies versus systematic biopsies in the detection of prostate cancer: a systematic review and meta-analysis. World J Urol. https://doi.org/10.1007/s00345-022-04087-z
Tafuri A, Iwata A, Shakir A et al (2021) Systematic biopsy of the prostate can be omitted in men with PI-RADS™ 5 and prostate specific antigen density greater than 15%. J Urol 206:289–297. https://doi.org/10.1097/JU.0000000000001766
Emmett L, Buteau J, Papa N et al (2021) The additive diagnostic value of prostate-specific membrane antigen positron emission tomography computed tomography to multiparametric magnetic resonance imaging triage in the diagnosis of prostate cancer (PRIMARY): a prospective multicentre study. Eur Urol 80:682–689. https://doi.org/10.1016/j.eururo.2021.08.002
Correas J-M, Halpern EJ, Barr RG et al (2021) Advanced ultrasound in the diagnosis of prostate cancer. World J Urol 39:661–676. https://doi.org/10.1007/s00345-020-03193-0
Lughezzani G, Maffei D, Saita A et al (2021) Diagnostic accuracy of microultrasound in patients with a suspicion of prostate cancer at magnetic resonance imaging: a single-institutional prospective study. Eur Urol Focus 7:1019–1026. https://doi.org/10.1016/j.euf.2020.09.013
Ghai S, Perlis N, Atallah C et al (2022) Comparison of micro-US and Multiparametric MRI for prostate cancer detection in biopsy-naive men. Radiology 305:390–398. https://doi.org/10.1148/radiol.212163
Weinreb JC, Barentsz JO, Choyke PL et al (2016) PI-RADS prostate imaging—reporting and data system: 2015, version 2. Eur Urol 69:16–40. https://doi.org/10.1016/j.eururo.2015.08.052
Turkbey B, Rosenkrantz AB, Haider MA et al (2019) Prostate imaging reporting and data system version 2.1: 2019 update of prostate imaging reporting and data system version 2. Eur Urol 76:340–351. https://doi.org/10.1016/j.eururo.2019.02.033
Ghai S, Eure G, Fradet V et al (2016) Assessing cancer risk on novel 29 MHz micro-ultrasound images of the prostate: creation of the micro-ultrasound protocol for prostate risk identification. J Urol 196:562–569. https://doi.org/10.1016/j.juro.2015.12.093
Epstein JI, Egevad L, Srigley JR, Humphrey PA (2016) The 2014 International Society of Urological Pathology (ISUP) consensus conference on gleason grading of prostatic carcinoma. Am J Surg Pathol 40:9
Fasulo V, Buffi NM, Regis F et al (2022) Use of high-resolution micro-ultrasound to predict extraprostatic extension of prostate cancer prior to surgery: a prospective single-institutional study. World J Urol 40:435–442. https://doi.org/10.1007/s00345-021-03890-4
Abouassaly R, Klein EA, El-Shefai A, Stephenson A (2020) Impact of using 29 MHz high-resolution micro-ultrasound in real-time targeting of transrectal prostate biopsies: initial experience. World J Urol 38:1201–1206. https://doi.org/10.1007/s00345-019-02863-y
Williams C, Ahdoot M, Daneshvar MA et al (2022) Why does magnetic resonance imaging-targeted biopsy miss clinically significant cancer? J Urol 207:95–107. https://doi.org/10.1097/JU.0000000000002182
Bhanji Y, Rowe SP, Pavlovich CP (2022) New imaging modalities to consider for men with prostate cancer on active surveillance. World J Urol 40:51–59. https://doi.org/10.1007/s00345-021-03762-x
Sountoulides P, Pyrgidis N, Polyzos SA et al (2021) Micro-ultrasound–guided vs multiparametric magnetic resonance imaging-targeted biopsy in the detection of prostate cancer: a systematic review and meta-analysis. J Urol 205:1254–1262. https://doi.org/10.1097/JU.0000000000001639
Rodríguez Socarrás ME, Gomez Rivas J, Cuadros Rivera V et al (2020) Prostate mapping for cancer diagnosis: the madrid protocol. transperineal prostate biopsies using multiparametric magnetic resonance imaging fusion and micro-ultrasound guided biopsies. J Urol 204:726–733. https://doi.org/10.1097/JU.0000000000001083
Kuppermann D, Calais J, Marks LS (2022) Imaging prostate cancer: clinical utility of prostate-specific membrane antigen. J Urol 207:769–778. https://doi.org/10.1097/JU.0000000000002457
Lopci E, Saita A, Lazzeri M et al (2018) 68 Ga-PSMA positron emission tomography/computerized tomography for primary diagnosis of prostate cancer in men with contraindications to or negative multiparametric magnetic resonance imaging: a prospective observational study. J Urol 200:95–103. https://doi.org/10.1016/j.juro.2018.01.079
Mannaerts CK, Gayet M, Verbeek JF et al (2018) Prostate cancer risk assessment in biopsy-naïve patients: the Rotterdam prostate cancer risk calculator in multiparametric magnetic resonance imaging-transrectal ultrasound (TRUS) fusion biopsy and systematic TRUS biopsy. Eur Urol Oncol 1:109–117. https://doi.org/10.1016/j.euo.2018.02.010
Faiena I, Salmasi A, Mendhiratta N et al (2019) PI-RADS version 2 category on 3 tesla multiparametric prostate magnetic resonance imaging predicts oncologic outcomes in gleason 3 + 4 prostate cancer on biopsy. J Urol 201:91–97. https://doi.org/10.1016/j.juro.2018.08.043
Meissner VH, Rauscher I, Schwamborn K et al (2022) Radical prostatectomy without prior biopsy following multiparametric magnetic resonance imaging and prostate-specific membrane antigen positron emission tomography. Eur Urol 82:156–160. https://doi.org/10.1016/j.eururo.2021.11.019
Funding
Giovanni Lughezzani certifies that this research did not receive any specific grant from funding agencies in the public, commercial or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
PPA was involved in protocol/project development, data analysis and manuscript—writing/editing. VF was involved in data collection or management and data analysis. RS-S and NMB were involved in manuscript—writing/editing. DM, NF and MP were involved in data collection or management. ML, AS, RH, GG and PC contributed to protocol/project development. GL contributed to protocol/project development and involved in manuscript—writing/editing.
Corresponding author
Ethics declarations
Conflict of interest
Giovanni Lughezzani certifies that all conflicts of interest, including specific financial interests and relationships and affiliations relevant to the subject matter or materials discussed in the manuscript (e.g., employment/affiliation, grants or funding, consultancies, honoraria, stock ownership or options, expert testimony, royalties or patent filed, received or pending), are the following: nothing to disclose.
Research involving human participants and/or animals
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. All participants provided informed consent for the clinical trial (ICH 003 v1.0 27/09/2017; study number 2004).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Avolio, P.P., Fasulo, V., Sanchez-Salas, R. et al. Diagnostic accuracy of multiparametric MRI- and microultrasound-targeted biopsy in biopsy-naïve patients with a PI-RADS 5 lesion: a single-institutional study. World J Urol 41, 2335–2342 (2023). https://doi.org/10.1007/s00345-023-04480-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-023-04480-2