Abstract
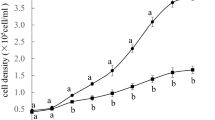
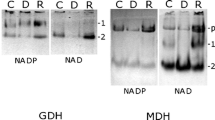
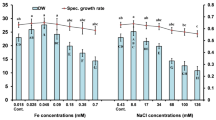
Haematococcus pluvialis is an ideal natural source of strong antioxidant astaxanthin. Sodium acetate (NaAc) was proven an effective organic carbon source for improving algal growth and astaxanthin production; however, the underlying mechanism remains obscure. To reveal the mechanism of NaAc at the green vegetative stage of H. pluvialis, the physiochemical characteristics and the global protein expression profiles obtained using a tandem mass tag labeling approach were compared between the control (CK) and two NaAc-addition groups. Results show that after NaAc addition, the biomass, nitrate consumption rate, and activities of three carbohydrate metabolism enzymes of H. pluvialis were significantly increased, and the net photosynthetic rate and chlorophyll content decreased. In addition, astaxanthin, total carbohydrates, and total lipids were accumulated, and some red cells appeared in the NaAc5 group. Moreover, 317 differentially expressed proteins (DEPs) with the most altered expression patterns were screened out in the CK vs. NaAc5 comparison in our proteomics study. All the DEPs involved in carbohydrate metabolism and lipid metabolism were significantly increased, while most of the photosynthesis-related proteins were depressed in the two NaAc-treated groups. The proteomics results were verified and supported by parallel reaction monitoring approach and physiochemical data. Our findings demonstrate that NaAc promoted the tricarboxylic acid cycle, glyoxylate cycle, and amino acid and lipid synthesis, and inhibited the photosynthesis-related activities, which consequently speeded up the growth and astaxanthin accumulation in this alga.
Similar content being viewed by others
Data Availability Statement
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Benvenuti G, Bosma R, Cuaresma M, Janssen M, Barbosa M J, Wijffels R H. 2015. Selecting microalgae with high lipid productivity and photosynthetic activity under nitrogen starvation. Journal of Applied Phycology, 27(4): 1425–1431, https://doi.org/10.1007/s10811-014-0470-8.
Bharte S, Desai K. 2019. The enhanced lipid productivity of Chlorella minutissima and Chlorella pyrenoidosa by carbon coupling nitrogen manipulation for biodiesel production. Environmental Science and Pollution Research, 26(4): 3492–3500, https://doi.org/10.1007/s11356-018-3757-5.
Bollivar D W, Beale S I. 1996. The chlorophyll biosynthetic enzyme Mg-protoporphyrin IX monomethyl ester (oxidative) cyclase (characterization and partial purification from Chlamydomonas reinhardtii and Synechocystis sp. PCC 6803). Plant Physiology, 112(1): 105–114, https://doi.org/10.1104/pp.112.1.105.
Breuer G, Lamers P P, Martens D E, Draaisma R B, Wijffels R H. 2012. The impact of nitrogen starvation on the dynamics of triacylglycerol accumulation in nine microalgae strains. Bioresource Technology, 124: 217–226, https://doi.org/10.1016/j.biortech.2012.08.003.
Chen Z, Luo L, Chen R F, Hu H H, Pan Y F, Jiang H B, Wan X, Jin H, Gong Y M. 2018. Acetylome profiling reveals extensive lysine acetylation of the fatty acid metabolism pathway in the diatom Phaeodactylum tricornutum. Molecular & Cellular Proteomics, 17(3): 399–412, https://doi.org/10.1074/mcp.RA117.000339.
Cheng J, Li K, Yang Z B, Zhou J H, Cen K F. 2016. Enhancing the growth rate and astaxanthin yield of Haematococcus pluvialis by nuclear irradiation and high concentration of carbon dioxide stress. Bioresource Technology, 204: 49–54, https://doi.org/10.1016/j.biortech.2015.12.076.
Cheng Y S, Zheng Y, VanderGheynst J S. 2011. Rapid quantitative analysis of lipids using a colorimetric method in a microplate format. Lipids, 46(1): 95–103, https://doi.org/10.1007/s11745-010-3494-0.
Cronan J EJr, Laporte D. 2005. Tricarboxylic acid cycle and glyoxylate bypass. EcoSal Plus, 1(2): 1–23, https://doi.org/10.1128/ecosalplus.3.5.2.
Du F C, Hu C Y, Sun X, Xu N J. 2021. Transcriptome analysis reveals pathways responsible for the promoting effect of sucrose on astaxanthin accumulation in Haematococcus pluvialis under high light condition. Aquaculture, 530: 735757, https://doi.org/10.1016/j.aquaculture.2020.735757.
Ganeteg U, Külheim C, Andersson J, Jansson S. 2004. Is each light-harvesting complex protein important for plant fitness? Plant Physiology, 134(1): 502–509, https://doi.org/10.1104/pp.103.033324.
Gong X D, Chen F. 1997. Optimization of culture medium for growth of Haematococcus pluvialis. Journal of Applied Phycology, 9(5): 437–444, https://doi.org/10.1023/A:1007944922264.
He B X, Hou L L, Dong M M, Shi J W, Huang X Y, Ding Y T, Cong X M, Zhang F, Zhang X C, Zang X N. 2018. Transcriptome analysis in Haematococcus pluvialis: astaxanthin induction by high light with acetate and Fe2+. International Journal of Molecular Sciences, 19(1): 175, https://doi.org/10.3390/ijms19010175.
Hu C Y, Cui D D, Sun X, Shi J X, Xu N J. 2020. Primary metabolism is associated with the astaxanthin biosynthesis in the green algae Haematococcus pluvialis under light stress. Algal Research, 46: 101768, https://doi.org/10.1016/j.algal.2019.101768.
Hussein G, Sankawa U, Goto H, Matsumoto K, Watanabe H. 2006. Astaxanthin, a carotenoid with potential in human health and nutrition. Journal of Natural Products, 69(3): 443–449, https://doi.org/10.1021/np050354+.
Imamoglu E, Dalay M C, Sukan F V. 2009. Influences of different stress media and high light intensities on accumulation of astaxanthin in the green alga Haematococcus pluvialis. New Biotechnology, 26(3–4): 199–204, https://doi.org/10.1016/j.nbt.2009.08.007.
Lee Y J, Jang J W, Kim K J, Maeng P J. 2011. TCA cycle-independent acetate metabolism via the glyoxylate cycle in Saccharomyces cerevisiae. Yeast, 28(2): 153–166, https://doi.org/10.1002/yea.1828.
Le-Feuvre R, Moraga-Suazo P, Gonzalez J, Martin S S, Henríquez V, Donoso A, Agurto-Muñoz C. 2020. Biotechnology applied to Haematococcus pluvialis Fotow: challenges and prospects for the enhancement of astaxanthin accumulation. Journal of Applied Phycology, 32(6): 3831–3852, https://doi.org/10.1007/s10811-020-02231-z.
Li K, Cheng J, Ye Q, He Y, Zhou J H, Cen K F. 2017. In vivo kinetics of lipids and astaxanthin evolution in Haematococcus pluvialis mutant under 15% CO2 using Raman microspectroscopy. Bioresource Technology, 244: 1439–1444, https://doi.org/10.1016/j.biortech.2017.04.116.
Li X B, Jin L, Pan X H, Yang L, Guo W D. 2019. Proteins expression and metabolite profile insight into phenolic biosynthesis during highbush blueberry fruit maturation. Food Chemistry, 290: 216–228, https://doi.org/10.1016/j.foodchem.2019.03.115.
Liu L J, Zhao Y, Jiang X X, Wang X Y, Liang W Y. 2018. Lipid accumulation of Chlorella pyrenoidosa under mixotrophic cultivation using acetate and ammonium. Bioresource Technology, 262: 342–346, https://doi.org/10.1016/j.biortech.2018.04.092.
Mertins P, Mani D R, Ruggles K V, Gillette M A, Clauser K R, Wang P, Wang X L, Qiao J W, Cao S, Petralia F, Kawaler E, Mundt F, Krug K, Tu Z D, Lei J T, Gatza M L, Wilkerson M, Perou C M, Yellapantula V, Huang K L, Lin C W, Mclellan M D, Yan P, Davies S R, Townsend R R, Skates S J, Wang J, Zhang B, Kinsinger C R, Mesri M, Rodriguez H, Ding L, Paulovich A G, Fenyö D, Ellis M J, Carr S A. 2016. Proteogenomics connects somatic mutations to signalling in breast cancer. Nature, 534(7605): 55–62, https://doi.org/10.1038/nature18003.
Nahidian B, Ghanati F, Shahbazi M, Soltani N. 2018. Effect of nutrients on the growth and physiological features of newly isolated Haematococcus pluvialis TMU1. Bioresource Technology, 255: 229–237, https://doi.org/10.1016/j.biortech.2018.01.130.
Nakazawa M. 2017. C2 metabolism in Euglena. In: Schwartzbach S, Shigeoka S eds. Euglena: Biochemistry, Cell and Molecular Biology. Springer, Cham. p.39–45, https://doi.org/10.1007/978-3-319-54910-1_3.
Pang N, Chen S L. 2017. Effects of C5 organic carbon and light on growth and cell activity of Haematococcus pluvialis under mixotrophic conditions. Algal Research, 21: 227–235, https://doi.org/10.1016/j.algal.2016.12.003.
Roosens N H, Bitar F A, Loenders K, Angenon G, Jacobs M. 2002. Overexpression of ornithine-δ-aminotransferase increases proline biosynthesis and confers osmotolerance in transgenic plants. Molecular Breeding, 9(2): 73–80, https://doi.org/10.1023/A:1026791932238.
Sakihama Y, Hidese R, Hasunuma T, Kondo A. 2019. Increased flux in acetyl-CoA synthetic pathway and TCA cycle of Kluyveromyces marxianus under respiratory conditions. Scientific Reports, 9(1): 5319, https://doi.org/10.1038/s41598-019-41863-1.
Sarada R, Bhattacharya S, Bhattacharya S, Ravishankar G A. 2002. A response surface approach for the production of natural pigment astaxanthin from green alga, Haematococcus pluvialis: effect of sodium acetate, culture age, and sodium chloride. Food Biotechnology, 16(2): 107–120, https://doi.org/10.1081/FBT-120014322.
Slabas A R, Fawcett T. 1992. The biochemistry and molecular biology of plant lipid biosynthesis. Plant Molecular Biology, 19(1): 169–191, https://doi.org/10.1007/BF00015613.
Starai V J, Escalante-Semerena J C. 2004. Acetyl-coenzyme A synthetase (AMP forming). Cellular and Molecular Life Sciences, 61(16): 2020–2030, https://doi.org/10.1007/s00018-004-3448-x.
Su Y X, Wang J X, Shi M L, Niu X F, Yu X H, Gao L J, Zhang X Q, Chen L, Zhang W W. 2014. Metabolomic and network analysis of astaxanthin-producing Haematococcus pluvialis under various stress conditions. Bioresource Technology, 170: 522–529, https://doi.org/10.1016/j.biortech.2014.08.018.
Tran N P, Park J K, Hong S J, Lee C G. 2009. Proteomics of proteins associated with astaxanthin accumulation in the green algae Haematococcus lacustris under the influence of sodium orthovanadate. Biotechnology Letters, 31(12): 1917–1922, https://doi.org/10.1007/s10529-009-0095-1.
Tripathi U, Sarada R, Ravishankar G A. 2002. Effect of culture conditions on growth of green alga-Haematococcus pluvialis and astaxanthin production. Acta Physiologiae Plantarum, 24(3): 323–329, https://doi.org/10.1007/s11738-002-0058-9.
Vorapreeda T, Thammarongtham C, Cheevadhanarak S, Laoteng K. 2012. Alternative routes of acetyl-CoA synthesis identified by comparative genomic analysis: involvement in the lipid production of oleaginous yeast and fungi. Microbiology, 158(1): 217–228, https://doi.org/10.1099/mic.0.051946-0.
Walker J M. 1994. The bicinchoninic acid (BCA) assay for protein quantitation. In: Walker J M ed. Basic Protein and Peptide Protocols. Humana Press, Clifton. p.5–8, https://doi.org/10.1385/0-89603-268-X:5.
Wan M X, Zhang Z, Wang J, Huang J K, Fan J H, Yu A Q, Wang W L, Li Y G. 2015. Sequential heterotrophy-dilution-photoinduction cultivation of Haematococcus pluvialis for efficient production of astaxanthin. Bioresource Technology, 198: 557–563, https://doi.org/10.1016/j.biortech.2015.09.031.
Wang L, Wang H L, Chen X R, Xu Y, Zhou T J, Wang X X, Lu Q, Ruan R. 2018. Using Chlorella vulgaris to treat toxic excess sludge extract, and identification of its response mechanism by proteomics approach. Bioresource Technology, 253: 188–196, https://doi.org/10.1016/j.biortech.2018.01.039.
Wang S B, Chen F, Sommerfeld M, Hu Q. 2004. Proteomic analysis of molecular response to oxidative stress by the green alga Haematococcus pluvialis (Chlorophyceae). Planta, 220(1): 17–29, https://doi.org/10.1007/s00425-004-1323-5.
Wang Y, Chiu S Y, Ho S H, Liu Z, Hasunuma T, Chang T T, Chang K F, Chang J S, Ren N Q, Kondo A. 2016. Improving carbohydrate production of Chlorella sorokiniana NIES-2168 through semi-continuous process coupled with mixotrophic cultivation. Biotechnology Journal, 11(8): 1072–1081, https://doi.org/10.1002/biot.201500270.
Wen Z W, Liu Z Y, Hou Y Y, Liu C F, Gao F, Zheng Y B, Chen F J. 2015. Ethanol induced astaxanthin accumulation and transcriptional expression of carotenogenic genes in Haematococcus pluvialis. Enzyme and Microbial Technology, 78: 10–17, https://doi.org/10.1016/j.enzmictec.2015.06.010.
Xi T Q, Kim D G, Roh S W, Choi J S, Choi Y E. 2016. Enhancement of astaxanthin production using Haematococcus pluvialis with novel LED wavelength shift strategy. Applied Microbiology and Biotechnology, 100(14): 6231–6238, https://doi.org/10.1007/s00253-016-7301-6.
Ye X, Chen J N, Hu C Y, Xu N J, Sun X. 2020. Promotion of the rapid growth in Haematococcus pluvialis under 0.16% CO2 condition revealed by transcriptome and metabolomic analysis. Journal of Plant Growth Regulation, 39(3): 1177–1190, https://doi.org/10.1007/s00344-019-10055-6.
Ye X, Yu X L, Hu C Y, Xu N J, Sun X. 2021. Effects of CO2 and sodium acetate on the growth and astaxanthin accumulation of the green alga Haematococcus pluvialis. Journal of Nuclear Agricultural Sciences, 35(3): 613–622, https://doi.org/10.11869/j.issn.100-8551.2021.03.0613. (in Chinese with English abstract)
You J, Hu H H, Xiong L Z. 2012. An ornithine δ-aminotransferase gene OsOAT confers drought and oxidative stress tolerance in rice. Plant Science, 197: 59–69, https://doi.org/10.1016/j.plantsci.2012.09.002.
Zhang C H, Zhang L T, Liu J G. 2019. Exogenous sodium acetate enhances astaxanthin accumulation and photoprotection in Haematococcus pluvialis at the non-motile stage. Journal of Applied Phycology, 31(2): 1001–1008, https://doi.org/10.1007/s10811-018-1622-z.
Zhao J. 2014. Determination of nitrate in vegetables by ultraviolet spectrophotometry method. Journal of Anhui Agricultural Sciences, 42(27): 9553–9554, https://doi.org/10.13989/j.cnki.0517-6611.2014.27.108. (in Chinese with English abstract)
Zheng S Y, He M L, Sui Y S, Gebreluel T, Zhou S M, Kemuma N D, Wang C H. 2017. Kelp waste extracts combined with acetate enhances the biofuel characteristics of Chlorella sorokiniana. Bioresource Technology, 225: 142–150, https://doi.org/10.1016/j.biortech.2016.11.060.
Zhou Y, Cai H M, Xiao J H, Li X H, Zhang Q F, Lian X M. 2009. Over-expression of aspartate aminotransferase genes in rice resulted in altered nitrogen metabolism and increased amino acid content in seeds. Theoretical and Applied Genetics, 118(7): 1381–1390, https://doi.org/10.1007/s00122-009-0988-3.
Zhuo P L, Li Y H, Zhong J L, Zheng M S, Zhu W R, Xu N J. 2019. Comprehensive effects of exogenous salicylic acid and light on chlorophyll fluorescence parameters and photosynthetic oxygen evolution in Ulva prolifera. Photosynthetica, 57(1): 342–349, https://doi.org/10.32615/ps.2019.035.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China (No. 31572638) and the K. C. Wong Magna Fund in Ningbo University
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Yu, X., Ye, X., Hu, C. et al. Sodium acetate can promote the growth and astaxanthin accumulation in the unicellular green alga Haematococcus pluvialis as revealed by a proteomics approach. J. Ocean. Limnol. 40, 2052–2067 (2022). https://doi.org/10.1007/s00343-021-1271-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00343-021-1271-y