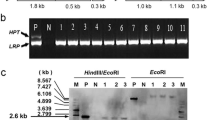
Abstract
Key message
This study demonstrated high expression and accumulation of human α-lactalbumin in transgenic maize, and significant improvement of lysine content in maize endosperm.
Abstract
As a high-yield crop, lack of lysine in endosperm storage protein is a major defect of maize (Zea mays L.). Specifically expression of foreign proteins is a potential way to improve lysine content in maize endosperm. Human α-lactalbumin is such a protein with high lysine content and high nutritional value. In this study, the codon-optimized human lactalbumin alpha (LALBA) gene was driven by maize endosperm-specific 27 kD γ-zein promoter, and transformed into maize. Five independent transgenic lines were obtained, and LALBA was highly expressed in endosperm in all these lines. Protein assay indicated that human α-lactalbumin was highly accumulated in maize endosperm. Immuno-localization assay indicated that human α-lactalbumin was mainly deposited into the protein body (PB). Protein interaction assay showed that human α-lactalbumin interacted with 16 kD γ-zein, which might lead to its deposition to the PBs. Amino acid analysis of two independent transgenic lines showed significant increase of lysine contents in transgenic endosperm, with 47.26% and 45.15% increase to their non-transgenic seeds, respectively. We obtained transgenic maize with endosperm-specific accumulation of human α-lactalbumin at high level and increased the lysine content in maize endosperm. This study demonstrated an effective way to improve the nutritional value of maize seeds.






Similar content being viewed by others
Data availability
All data supporting the findings of this study are available within the paper and within its.
References
Azevedo RA, Arruda P (2010) High-lysine maize: the key discoveries that have made it possible. Amino Acids 39:979–989. https://doi.org/10.1007/s00726-010-0576-5
Azevedo RA, Lancien M, Lea PJ (2006) The aspartic acid metabolic pathway, an exciting and essential pathway in plants. Amino Acids 30:143–162. https://doi.org/10.1007/s00726-005-0245-2
Bicar EH, Woodman-Clikeman W, Sangtong V, Peterson JM, Yang SS, Lee M, Scott MP (2008) Transgenic maize endosperm containing a milk protein has improved amino acid balance. Transgenic Res 17:59–71. https://doi.org/10.1007/s11248-007-9081-3
Burgoyne RD, Duncan JS (1998) Secretion of milk proteins. J Mammary Gland Biol Neoplasia 3:275–286. https://doi.org/10.1023/a:1018763427108
Chang Y, Shen E, Wen L, Yu J, Zhu D, Zhao Q (2015) Seed-specific expression of the Arabidopsis AtMAP18 gene increases both lysine and total protein content in maize. PLoS ONE 10:e0142952. https://doi.org/10.1371/journal.pone.0142952
Energy and protein requirements (1985) Report of a joint FAO/WHO/UNU Expert Consultation. World Health Organization technical report series 724:1–206
Feng Y, Ma Y, Feng F, Chen X, Qi W, Ma Z, Song R (2022) Accumulation of 22 kDa α-zein-mediated nonzein protein in protein body of maize endosperm. New Phytol 233:265–281. https://doi.org/10.1111/nph.17796
Ferreira RR, Varisi VA, Meinhardt LW, Lea PJ, Azevedo RA (2005) Are high-lysine cereal crops still a challenge? Brazil J Med Biol Res 38:985–994. https://doi.org/10.1590/s0100-879x2005000700002
Ferrari RC, Cruz BC, Gastaldi VD, Storl T, Ferrari EC, Boxall SF, Hartwell J, Freschi L (2020) Exploring C(4)-CAM plasticity within the Portulaca oleracea complex. Sci Rep 10:14237. https://doi.org/10.1038/s41598-020-71012-y
Frame B, Main M, Schick R, Wang K (2011) Genetic transformation using maize immature zygotic embryos. Methods Mol Biol (clifton, NJ) 710:327–341. https://doi.org/10.1007/978-1-61737-988-8_22
Galili G, Amir R (2013) Fortifying plants with the essential amino acids lysine and methionine to improve nutritional quality. Plant Biotechnol J 11:211–222. https://doi.org/10.1111/pbi.12025
Gao H, Smith J, Yang M, Jones S, Djukanovic V, Nicholson MG, West A, Bidney D, Falco SC, Jantz D, Lyznik LA (2010) Heritable targeted mutagenesis in maize using a designed endonuclease. Plant J Cell Mol Biol 61:176–187. https://doi.org/10.1111/j.1365-313X.2009.04041.x
Gomord V, Denmat LA, Fitchette-Lainé AC, Satiat-Jeunemaitre B, Hawes C, Faye L (1997) The C-terminal HDEL sequence is sufficient for retention of secretory proteins in the endoplasmic reticulum (ER) but promotes vacuolar targeting of proteins that escape the ER. Plant J Cell Mol Biol 11:313–325. https://doi.org/10.1046/j.1365-313x.1997.11020313.x
Guan Z, Meng X, Sun Z, Xu Z, Song R (2008) Characterization of duplicated Dunaliella viridis SPT1 genes provides insights into early gene divergence after duplication. Gene 423:36–42. https://doi.org/10.1016/j.gene.2008.06.029
Habben JE, Kirleis AW, Larkins BA (1993) The origin of lysine-containing proteins in opaque-2 maize endosperm. Plant Mol Biol 23:825–838. https://doi.org/10.1007/bf00021537
Hall L, Davies MS, Craig RK (1981) The construction, identification and characterisation of plasmids containing human alpha-lactalbumin cDNA sequences. Nucleic Acids Res 9:65–84. https://doi.org/10.1093/nar/9.1.65
Hobbs SL, Warkentin TD, DeLong CM (1993) Transgene copy number can be positively or negatively associated with transgene expression. Plant Mol Biol 21:17–26. https://doi.org/10.1007/BF00039614
Holding DR, Meeley RB, Hazebroek J, Selinger D, Gruis F, Jung R, Larkins BA (2010) Identification and characterization of the maize arogenate dehydrogenase gene family. J Exp Bot 61:3663–3673. https://doi.org/10.1093/jxb/erq179
Joseph M, Ludevid MD, Torrent M, Rofidal V, Tauzin M, Rossignol M, Peltier JB (2012) Proteomic characterisation of endoplasmic reticulum-derived protein bodies in tobacco leaves. BMC Plant Biol 12:36. https://doi.org/10.1186/1471-2229-12-36
Kapila J, De Rycke R, Van Montagu M, Angenon G (1997) An Agrobacterium-mediated transient gene expression system for intact leaves. Plant Sci 122:101–108. https://doi.org/10.1016/s0168-9452(96)04541-4
Kirma M, Araujo WL, Fernie AR, Galili G (2012) The multifaceted role of aspartate-family amino acids in plant metabolism. J Exp Bot 63:4995–5001. https://doi.org/10.1093/jxb/ers119
Lambert RJ, Alexander DE, Dudley JW (1969) Relative performance of normal and modified protein (Opaque-2) maize hybrids. Crop Sci 9:242–243. https://doi.org/10.2135/cropsci1969.0011183X000900020040x
Lending CR, Larkins BA (1989) Changes in the zein composition of protein bodies during maize endosperm development. Plant Cell 1:1011–1023. https://doi.org/10.1105/tpc.1.10.1011
Liang X, Qian G, Sun J, Yang M, Shi X, Yang H, Wu J, Wang Z, Zheng Y, Yue X (2021) Evaluation of antigenicity and nutritional properties of enzymatically hydrolyzed cow milk. Sci Rep 11:18623. https://doi.org/10.1038/s41598-021-98136-z
Liu C, Li S, Yue J, Xiao W, Zhao Q, Zhu D, Yu J (2015) Microtubule-associated protein SBgLR facilitates storage protein deposition and its expression leads to lysine content increase in transgenic maize endosperm. Int J Mol Sci 16:29772–29786. https://doi.org/10.3390/ijms161226199
Mather IH, Jack LJ, Madara PJ, Johnson VG (2001) The distribution of MUC1, an apical membrane glycoprotein, in mammary epithelial cells at the resolution of the electron microscope: implications for the mechanism of milk secretion. Cell Tissue Res 304:91–101. https://doi.org/10.1007/s004410100351
Mertz ET, Bates LS, Nelson OE (1964) Mutant gene that changes protein composition and increases lysine content of maize endosperm. Science (new York, NY) 145:279–280. https://doi.org/10.1126/science.145.3629.279
Mladenovic-Drinic S, Ignjatovic-Micic D, Eric I, Andjelkovic V, Jelovac D, Konstantinov K (2004) Biotechnology in maize breeding. Genetika 36:93–109. https://doi.org/10.2298/gensr0402093m
Osborne TB, Mendel LB, Ferry EL, Wakeman AJ (1914) Nutritive properties of proteins of the maize kernel. J Biol Chem 18:1–16. https://doi.org/10.1016/s0021-9258(18)88372-7
Permyakov EA, Berliner LJ (2000) α-Lactalbumin: structure and function. FEBS Lett 473:269–274. https://doi.org/10.1016/s0014-5793(00)01546-5
Rajeevkumar S, Anunanthini P, Sathishkumar R (2015) Epigenetic silencing in transgenic plants. Front Plant Sci 6:693. https://doi.org/10.3389/fpls.2015.00693
Rascon-Cruz Q, Sinagawa-Garcia S, Osuna-Castro JA, Bohorova N, Paredes-Lopez O (2004) Accumulation, assembly, and digestibility of amarantin expressed in transgenic tropical maize. Theor Appl Genet 108:335–342. https://doi.org/10.1007/s00122-003-1430-x
Scrimshaw NS (2006) Quality protein maize. Food Nutr Bull 27:265–266. https://doi.org/10.1177/156482650602700310
Shou H, Bordallo P, Wang K (2004) Expression of the Nicotiana protein kinase (NPK1) enhanced drought tolerance in transgenic maize. J Exp Bot 55:1013–1019. https://doi.org/10.1093/jxb/erh129
Takase K, Hagiwara K (1998) Expression of human alpha-lactalbumin in transgenic tobacco. J Biochem 123:440–444. https://doi.org/10.1093/oxfordjournals.jbchem.a021956
Torrent M, Llompart B, Lasserre-Ramassamy S, Llop-Tous I, Bastida M, Marzabal P, Westerholm-Parvinen A, Saloheimo M, Heifetz PB, Ludevid MD (2009) Eukaryotic protein production in designed storage organelles. BMC Biol 7:5. https://doi.org/10.1186/1741-7007-7-5
Vaucheret H, Beclin C, Elmayan T, Feuerbach F, Godon C, Morel JB, Mourrain P, Palauqui JC, Vernhettes S (1998) Transgene-induced gene silencing in plants. Plant J Cell Mol Biol 16:651–659. https://doi.org/10.1046/j.1365-313x.1998.00337.x
Wallace JC, Lopes MA, Paiva E, Larkins BA (1990) New methods for extraction and quantitation of zeins reveal a high content of gamma-zein in modified opaque-2 maize. Plant Physiol 92:191–196. https://doi.org/10.1104/pp.92.1.191
Wang M, Liu C, Li S, Zhu D, Zhao Q, Yu J (2013) Improved nutritive quality and salt resistance in transgenic maize by simultaneously overexpression of a natural lysine-rich protein gene, SBgLR, and an ERF transcription factor gene, TSRF1. Int J Mol Sci 14:9459–9474. https://doi.org/10.3390/ijms14059459
Wang G, Qi W, Wu Q, Yao D, Zhang J, Zhu J, Wang G, Wang G, Tang Y, Song R (2014) Identification and characterization of maize floury4 as a novel semidominant opaque mutant that disrupts protein body assembly. Plant Physiol 165:582–594. https://doi.org/10.1104/pp.114.238030
Wenefrida I, Utomo HS, Linscombe SD (2013) Mutational breeding and genetic engineering in the development of high grain protein content. J Agric Food Chem 61:11702–11710. https://doi.org/10.1021/jf4016812
Woo YM, Hu DW, Larkins BA, Jung R (2001) Genomics analysis of genes expressed in maize endosperm identifies novel seed proteins and clarifies patterns of zein gene expression. Plant Cell 13:2297–2317. https://doi.org/10.1105/tpc.010240
Wu Y, Messing J (2014) Proteome balancing of the maize seed for higher nutritional value. Front Plant Sci 5:240. https://doi.org/10.3389/fpls.2014.00240
Yang SH, Moran DL, Jia HW, Bicar EH, Lee M, Scott MP (2002) Expression of a synthetic porcine alpha-lactalbumin gene in the kernels of transgenic maize. Transgenic Res 11:11–20. https://doi.org/10.1023/a:1013996129125
Yu J, Peng P, Zhang X, Zhao Q, Zhu D, Sun X, Liu J, Ao G (2005) Seed-specific expression of the lysine-rich protein gene sb401 significantly increases both lysine and total protein content in maize seeds. Food Nutr Bull 26:427–431. https://doi.org/10.3390/ijms15045350
Yue J, Li C, Zhao Q, Zhu D, Yu J (2014) Seed-specific expression of a lysine-rich protein gene, GhLRP, from cotton significantly increases the lysine content in maize seeds. Int J Mol Sci 15:5350–5365. https://doi.org/10.3390/ijms15045350
Funding
This work was supported by the Research Program of Sanya Yazhou Bay Science and Technology City (SYND-2021-22 to R.S.), the Hainan Yazhou Bay Seed Lab (B21HJ8105 to R.S.), and the National Key Research and Development Program of China (grant 2016YFD0100503 to W.Q.).
Author information
Authors and Affiliations
Contributions
RTS and WWQ conceived and designed research. CWLL and LFJ performed experiments. CWLL and WM collected and analyzed the data. CWLL, WWQ and RTS wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Communicated by Li Tian.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
299_2022_2906_MOESM1_ESM.pdf
Supplementary file1 Figure S1. The sequence of 27 kD γ-zein promoter, modified LALBA (opening reading frame) and 27 kD γ-zein terminator. Figure S2. RT-qPCR analysis of LALBA expression in 18 DAP seeds with Ubiquitin as reference gene. Figure S3. Protein interaction between human α-lactalbumin and zeins. Figure S4. Schematic structure of chimeric constructs used in transient expression. Figure S5. Phenotypic features of human α-lactalbumin transgenic line #1. Table S1. Primers used in this research. Table S2. Amino acid content in #1 endosperm. Table S3. Amino acid content in #4 endosperm. Table S4. Other transgenic maize for increasing lysine content. Table S5. The contents of five amino acids in human α-lactalbumin and α-zeins (PDF 807 KB)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, C., Ma, W., Jin, L. et al. Endosperm-specific accumulation of human α-lactalbumin increases seed lysine content in maize. Plant Cell Rep 41, 2023–2035 (2022). https://doi.org/10.1007/s00299-022-02906-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-022-02906-6