Abstract
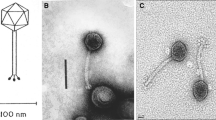
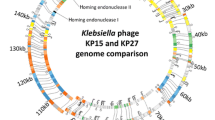
This work describes the newly isolated Klebsiella pneumoniae phage vB_KpnS-Carvaje that presents unique features in relation to other phages reported to date. These findings provide new insights into the diversity and evolutionary pathways of Klebsiella phages. The genome characterization of the Carvaje phage revealed that its genome length is approximately 57 kb with 99 open reading frames (ORFs), 33 of which have assigned functions while 66 are unknown. This phage differs from other sequenced Klebsiella phages, showing the closest resemblance (up to 65.32%) with Salmonella phages belonging to the Nonanavirus and Sashavirus genera. Comparisons at the amino acid level and phylogeny analysis among homologous genomes indicate that the Klebsiella Carvaje phage forms a novel sister taxon within the node of the Nonanaviruses and Sashaviruses cluster. Due to the unique features of the Carvaje phage, we propose the constitution of a new genus within the Caudoviricetes class. Further studies include the exploitation of this phage and its identified proteins for the control of Klebsiella infections and as recognition molecules in diagnostic methods.









Similar content being viewed by others
Data availability
Phage infectivity, one-step growth curves, genome assembly and annotation, whole-genome alignment with closest relatives, phylogenetic analysis of vB_KpnS-Carvaje’s whole and partial (Open Reading Frames) genome, detailed bioinformatic analysis of most relevant phage proteins. vB_KpnS-Carvaje whole-genome is deposited in Genbank under the Accession number: OL604152. Further relevant data are within the paper and its Supporting Information file.
References
Abedon ST (2018) Detection of bacteriophages: phage plaques. In: Harper D, Abedon S, Burrowes B, McConville M (eds) Bacteriophages. Springer, Cham, pp 507–538
Adams MH (1959) Bacteriophages. Interscience Publishers, New York
Alikhan N, Petty NK, Ben ZNL, Beatson SA (2011) BLAST ring image generator (BRIG): simple prokaryote genome comparisons. BMC Genomics 12:402. https://doi.org/10.1186/1471-2164-12-402
Altschup SF, Gish W, Miller W et al (1990) Basic local alignment search tool. J Mol Biol 215:403–410. https://doi.org/10.1016/S0022-2836(05)80360-2
Aziz RK, Bartels D, Best A et al (2008) The RAST Server: rapid annotations using subsystems technology. BMC Genomics 9:75. https://doi.org/10.1186/1471-2164-9-75
Baerends RJ, Smits WK, de Jong A et al (2004) Genome2D: a visualzation tool for rapid analysis of bacterial transcriptome data. Genome Biol 5:R37. https://doi.org/10.1186/gb-2004-5-5-r37
Bankevich A, Nurk S, Antipov D et al (2012) SPAdes: a new genome assembly algorithm and its applications to single-cell sequencing. J Comput Biol 19:455–477. https://doi.org/10.1089/cmb.2012.0021
Bloch S, Lewandowska N, Grzegorz W (2021) Bacteriophages as sources of small non-coding RNA molecules. Plasmid 113:102527. https://doi.org/10.1016/j.plasmid.2020.102527
Bolger AM, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30:2114–2120. https://doi.org/10.1093/bioinformatics/btu170
Bossche AVD, Ceyssens P, De SJ et al (2014) Systematic identification of hypothetical bacteriophage proteins targeting key protein complexes of pseudomonas aeruginosa. J Proteome Res 13:4446–4456. https://doi.org/10.1021/pr500796n
Carvalho CM, Gannon BW, Halfhide DE et al (2010) The in vivo efficacy of two administration routes of a phage cocktail to reduce numbers of campylobacter coli and campylobacter jejuni in chickens. BMC Microbiol 10:232. https://doi.org/10.1186/1471-2180-10-232
Carver T, Harris SR, Berriman M et al (2012) Artemis: an integrated platform for visualization and analysis of high-throughput sequence-based experimental data. Bioinformatics 28:464–469. https://doi.org/10.1093/bioinformatics/btr703
Ciccarelli FD, Doerks T, Creevey CJ et al (2006) Toward automatic reconstruction of a highly resolved tree of life. Science (80-) 311:1283–1287. https://doi.org/10.1126/science.1121745
Coppens L, Lavigne R (2020) SAPPHIRE: a neural network based classifier for σ 70 promoter prediction in pseudomonas. BMC Bioinformatics 21:1–8. https://doi.org/10.1186/s12859-020-03730-z
D’Hérelle F (1917) Sur un microbe invisible antagoniste des bacilles dysentériques, 1st edn. Comptes rendus hebdomadaires des séances de l’Académie des Sciences, Paris
Darling ACE, Mau B, Blattner FR, Perna NT (2004) Mauve: multiple alignment of conserved genomic sequence with rearrangements. Genome Res 14:1394–1403. https://doi.org/10.1101/gr.2289704.tion
Desplats C, Wilson WH, Krisch HM et al (2001) A conserved genetic module that encodes the major virion components in both the coliphage T4 and the marine cyanophage S-PM2. Proc Natl Acad Sci 98:11411–11416. https://doi.org/10.1073/pnas.191174498
Dong D, Liu W, Li H et al (2015) Survey and rapid detection of Klebsiella pneumoniae in clinical samples targeting rcsA gene in Beijing. China Front Microbiol 6:519. https://doi.org/10.3389/fmicb.2015.00519
Donlan RM (2009) Preventing biofilms of clinically relevant organisms using bacteriophage. Trends Microbiol 17:66–72. https://doi.org/10.1016/j.tim.2008.11.002
Dunstan RA, Bamert RS, Belousoff MJ et al (2021) Mechanistic insights into the capsule-targeting depolymerase from a klebsiella pneumoniae bacteriophage. Microbiol Spectr 9(1):e0102321. https://doi.org/10.1128/Spectrum.01023-21
Eckstein S, Stender J, Mzoughi S et al (2021) Isolation and characterization of lytic phage TUN1 specific for Klebsiella pneumoniae K64 clinical isolates from Tunisia. BMC Microbiol. https://doi.org/10.1186/s12866-021-02251-w
European centre for disease prevention and control (2019) Surveillance of antimicrobial resistance in Europe 2018. Stockholm: ECDC; 2019
Farris JS (1972) Estimating phylogenetic trees from distance matrices. Am Nat 106:645–668. https://doi.org/10.1086/282802
Fernandes E, Martins VC, Nóbrega C et al (2014) A bacteriophage detection tool for viability assessment of Salmonella cells. Biosens Bioelectron 52:239–246. https://doi.org/10.1016/j.bios.2013.08.053
Fokine A, Rossmann MG (2014) Molecular architecture of tailed double-stranded DNA phages. Bacteriophage 4:e28281. https://doi.org/10.4161/bact.28281
Founou RC, Founou LL, Essack SY (2017) Clinical and economic impact of antibiotic resistance in developing countries: a systematic review and meta-analysis. PLoS ONE 12:e0189621. https://doi.org/10.1371/journal.pone.0189621
Garrido-Maestu A, Fuciños P, Azinheiro S et al (2019) Specific detection of viable Salmonella Enteritidis by phage amplification combined with qPCR (PAA-qPCR) in spiked chicken meat samples. Food Control 99:79–83. https://doi.org/10.1016/j.foodcont.2018.12.038
Göker M, García-Blázquez G, Voglmayr H et al (2009) Molecular taxonomy of phytopathogenic fungi a case study in Peronospora. PLoS ONE 4:e6319. https://doi.org/10.1371/journal.pone.0006319
Gu J, Liu X, Li Y et al (2012) A Method for generation phage cocktail with great therapeutic potential. PLoS ONE 7:e31698. https://doi.org/10.1371/journal.pone.0031698
Gurevich A, Saveliev V, Vyahhi N, Tesler G (2013) QUAST: quality assessment tool for genome assemblies. Bioinformatics 29:1072–1075. https://doi.org/10.1093/bioinformatics/btt086
Hecht A, Glasgow J, Jaschke PR et al (2017) Measurements of translation initiation from all 64 codons in E. coli. Nucleic Acids Res 45:3615–3626. https://doi.org/10.1101/063800
Herridge WP, Shibu P, O’Shea J et al (2020) Bacteriophages of Klebsiella spp., their diversity and potential therapeutic uses. J Med Microbiol 69:176–194. https://doi.org/10.1099/jmm.0.001141
Hsu C-R, Lin T-L, Pan Y-J et al (2013) Isolation of a bacteriophage specific for a new capsular type of klebsiella pneumoniae and characterization of its polysaccharide depolymerase. PLoS ONE 8:e70092. https://doi.org/10.1371/journal.pone.0070092
Ilyina TV, Gorbalenya AE, Koonin EV (1992) Organization and evolution of bacterial and bacteriophage primase-helicase systems. J Mol Evol 34:351–357. https://doi.org/10.1007/BF00160243
Jamal M, Hussain T, Das CR, Andleeb S (2015) Characterization of siphoviridae phage Z and studying its efficacy against multidrug-resistant Klebsiella pneumoniae planktonic cells and biofilm. J Med Microbiol 64:454–462. https://doi.org/10.1099/jmm.0.000040
King AMQ, Lefkowitz E, Adams MJ, Carstens EB (2012) Order – Caudovirales. In: virus taxonomy: ninth report of the international committee on taxonomy of viruses. Elsevier, San Diego, pp 39–45
Komisarova EV, Kislichkina AA, Krasilnikova VM et al (2017) Complete nucleotide sequence of klebsiella pneumoniae bacteriophage vB_KpnM_KpV477. Genome Announc 5:e00694-e717. https://doi.org/10.1128/genomeA.00694-17
Kumar S, Tripathy S, Jyoti A, Singh SG (2019) Recent advances in biosensors for diagnosis and detection of sepsis: a comprehensive review. Biosens Bioelectron 124–125:205–215. https://doi.org/10.1016/j.bios.2018.10.034
Lefort V, Desper R, Gascuel O (2015) FastME 2.0: a comprehensive, accurate, and fast distance-based phylogeny inference program. Mol Biol Evol 32:2798–2800. https://doi.org/10.1093/molbev/msv150
Li P, Lo C, Anderson JJ et al (2017) Enabling the democratization of the genomics revolution with a fully integrated web-based bioinformatics platform. Nucleic Acids Res 45:67–80. https://doi.org/10.1093/nar/gkw1027
Li P, Zhang Y, Yan F, Zhou X (2021) Characteristics of a bacteriophage, vB _ Kox _ ZX8, isolated from clinical klebsiella oxytoca and its therapeutic effect on mice bacteremia. Front Microbiol 12:1–15. https://doi.org/10.3389/fmicb.2021.763136
Lim SH, Mix S, Xu Z et al (2014) Colorimetric sensor array allows fast detection and simultaneous identification of sepsis-causing bacteria in spiked blood culture. J Clin Microbiol 52:592–598. https://doi.org/10.1128/JCM.02377-13
McNair K, Zhou C, Dinsdale EA et al (2019) PHANOTATE: a novel approach to gene identification in phage genomes. Bioinformatics 35:4537–4542. https://doi.org/10.1093/bioinformatics/btz265
Meier-Kolthoff JP, Göker M (2017) VICTOR: genome-based phylogeny and classification of prokaryotic viruses. Bioinformatics 33:3396–3404. https://doi.org/10.1093/bioinformatics/btx440
Meier-Kolthoff JP, Auch AF, Klenk H-P, Göker M (2013) Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinformatics 14:60. https://doi.org/10.1186/1471-2105-14-60
Meier-kolthoff JP, Hahnke RL, Petersen J et al (2014) Complete genome sequence of DSM 30083 T, the type strain (U5/41 T) of Escherichia coli, and a proposal for delineating subspecies in microbial taxonomy. Stand Genomic Sci 9:2. https://doi.org/10.1186/1944-3277-9-2
Min L, Lessor L, Leary CO et al (2019) Complete genome sequence of klebsiella pneumoniae myophage mulock. Microbiol Resour Announc 8:e01338-e1419. https://doi.org/10.1128/MRA.01338-19
Moraru C, Varsani A, Kropinski AM (2020) VIRIDIC—a novel tool to calculate the intergenomic similarities of prokaryote-infecting viruses. Viruses 12:1268. https://doi.org/10.3390/v12111268
Murphy J, Bottacini F, Mahony J et al (2016) Comparative genomics and functional analysis of the 936 group of lactococcal Siphoviridae phages. Sci Rep 6:21345. https://doi.org/10.1038/srep21345
Naville M, Ghuillot-gaudeffroy A, Marchais A, Gautheret D (2011) ARNold: a web tool for the prediction of Rho-independent transcription terminators. RNA Biol 8:11–13. https://doi.org/10.4161/rna.8.1.13346
Nishimura Y, Yoshida T, Kuronishi M et al (2017) Genome analysis ViPTree : the viral proteomic tree server. Bioinformatics 33:2379–2380. https://doi.org/10.1093/bioinformatics/btx157
Nobrega FL, Costa AR, Kluskens LD, Azeredo J (2015) Revisiting phage therapy: new applications for old resources. Trends Microbiol 23:185–191. https://doi.org/10.1016/j.tim.2015.01.006
Nobrega FL, Vlot M, Jonge PA et al (2018) Targeting mechanisms of tailed bacteriophages. Nat Rev Microbiol 16:760–773. https://doi.org/10.1038/s41579-018-0070-8
Nogueira CL, Pires DP, Monteiro R et al (2021) Exploitation of a klebsiella bacteriophage receptor-binding protein as a superior biorecognition molecule. ACS Infect Dis 7:3077–3087. https://doi.org/10.1021/acsinfecdis.1c00366
Oliveira A, Ribeiro HG, Silva AC et al (2017) Synergistic antimicrobial interaction between honey and phage against escherichia coli biofilms. Front Microbiol 8:2407. https://doi.org/10.3389/fmicb.2017.02407
Oliveira A, Sousa JC, Silva AC et al (2018) Chestnut honey and bacteriophage application to control pseudomonas aeruginosa and escherichia coli biofilms: evaluation in an ex vivo wound model. Front Microbiol 9:1725. https://doi.org/10.3389/FMICB.2018.01725
Peng Q, Fang M, Liu X et al (2020) Isolation and characterization of a novel phage for controlling multidrug-resistant klebsiella pneumoniae. Microorganisms 8:542. https://doi.org/10.3390/microorganisms8040542
Pires D, Sillankorva S, Faustino A, Azeredo J (2011) Use of newly isolated phages for control of Pseudomonas aeruginosa PAO1 and ATCC 10145 biofilms. Res Microbiol 162:798–806. https://doi.org/10.1016/j.resmic.2011.06.010
Pires DP, Vilas Boas D, Sillankorva S, Azeredo J (2015) Phage therapy: a step forward in the treatment of Pseudomonas aeruginosa infections. J Virol 89:7449–7456. https://doi.org/10.1128/JVI.00385-15
Pringle CR (1998) Virus taxonomy. Arch Virol 143:1449–1459. https://doi.org/10.1007/s007050050389
Renner LD, Zan J, Hu LI et al (2017) Detection of ESKAPE bacterial pathogens at the point of care using isothermal DNA-based assays in a portable degas-actuated microfluidic diagnostic assay platform. Appl Environ Microbiol 83:e02449-e2516. https://doi.org/10.1128/AEM.02449-16
Sambrook J, Russell DW (2001) Molecular Cloning: A Laboratory Manual, 3rd edn. Cold Spring Harbor Laboratory Press, New York
Schindelin J, Arganda-Carreras I, Frise E et al (2012) Fiji: An open-source platform for biological-image analysis. Nat Methods 9:676–682. https://doi.org/10.1038/nmeth.2019
Schmelcher M, Loessner MJ (2014) Application of bacteriophages for detection of foodborne pathogens. Bacteriophage 4:e28137. https://doi.org/10.4161/bact.28137
Sievers F, Wilm A, Dineen D et al (2011) Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal Omega. Mol Syst Biol 7:539. https://doi.org/10.1038/msb.2011.75
Sillankorva S, Azeredo J (2014) Bacteriophage Attack as an Anti-biofilm Strategy. In: Donelli G (ed) Microbial biofilms. Methods in molecular biology (Methods and Protocols), vol 1147. Humana Press, New York, pp 277–285
Singh A, Arya SK, Glass N et al (2010) Bacteriophage tailspike proteins as molecular probes for sensitive and selective bacterial detection. Biosens Bioelectron 26:131–138. https://doi.org/10.1016/j.bios.2010.05.024
Snyder EE, Kampanya N, Lu J et al (2007) PATRIC : the VBI pathosystems resource integration center. Nucleic Acids Res 35:401–406. https://doi.org/10.1093/nar/gkl858
Söding J, Biegert A, Lupas AN (2005) The HHpred interactive server for protein homology detection and structure prediction. Nucleic Acids Res 33:W244–W248. https://doi.org/10.1093/nar/gki408
Sullivan MJ, Petty NK, Beatson SA (2011) Easyfig: a genome comparison visualizer. Bioinformatics 27:1009–1010. https://doi.org/10.1093/bioinformatics/btr039
Townsend EM, Kelly L, Gannon L et al (2021) Isolation and characterization of klebsiella phages for phage therapy. Phage (new Rochelle) 2:26–42. https://doi.org/10.1089/phage.2020.0046
Turner D, Kropinski AM, Adriaenssens EM (2021) A roadmap for genome-based phage taxonomy. Viruses 13:506. https://doi.org/10.3390/v13030506
Twort FW (1915) An investigation on the nature of ultra-microscopic viruses. Lancet 186:1241–1243. https://doi.org/10.1016/S0140-6736(01)20383-3
Waterhouse AM, Procter JB, Martin DMA et al (2009) Jalview version 2 — a multiple sequence alignment editor and analysis workbench. Bioinformatics 25:1189–1191. https://doi.org/10.1093/bioinformatics/btp033
Wyres KL, Lam MMC, Holt KE (2020) Population genomics of Klebsiella pneumoniae. Nat Rev Microbiol 18:344–359. https://doi.org/10.1038/s41579-019-0315-1
Xu J, Hendrix RW, Duda RL (2004) Conserved translational frameshift in dsDNA bacteriophage tail assembly genes. Mol Cell 16:11–21. https://doi.org/10.1016/j.molcel.2004.09.006
Xu L, Dong Z, Fang L et al (2019) OrthoVenn2: a web server for whole-genome comparison and annotation of orthologous clusters across multiple species. Nucleic Acids Res 47:W52–W58. https://doi.org/10.1093/nar/gkz333
Yaung SJ, Esvelt KM, Church GM (2015) Complete genome sequences of T4-like bacteriophages RB3, RB5, RB6, RB7, RB9, RB10, RB27, RB33, RB55, RB59, and RB68. Genome Announc 3:e01122-e1214. https://doi.org/10.1128/genomeA.01122-14
Yazdi M, Bouzari M, Ghaemi EA (2019) Genomic analyses of a novel bacteriophage (VB_PmiS-Isfahan) within Siphoviridae family infecting Proteus mirabilis. Genomics 111:1283–1291. https://doi.org/10.1016/j.ygeno.2018.08.008
Zeng C, Gilcrease EB, Hendrix RW et al (2019) DNA Packaging and genomics of the salmonella 9NA-like phages. J Virol 93:1–21. https://doi.org/10.1128/JVI.00848-19
Acknowledgements
The authors would like to thank the Clinical Academic Center of Braga (2CA-Braga) for establishing the ethical agreement between the Hospital of Braga and our research group, which led to the provision of the clinical isolates.
Funding
This study was supported by the Portuguese Foundation for Science and Technology (FCT) under the scope of the project “Phages‐on‐chip” PTDC/BTM‐SAL/32442/2017 (POCI‐01‐0145‐FEDER‐032442) and the strategic funding of the research unit INESC MN (UID/05367/2020) through pluriannual BASE and PROGRAMATICO financing. JCS acknowledges financial support from FCT through the grant SFRH/BD/144992/2019. SS acknowledges funding by FCT through the individual scientific employment program contract (2020.03171.CEECIND).
Author information
Authors and Affiliations
Contributions
CMC: conceived and coordinated the study. JCS: performed the laboratory experiments and curated raw sequencing data. JCS, SS and CMC: performed and analyzed whole and partial genome alignments and phylogenetics. AF isolated and characterized the hospital clinical isolates. JCS: wrote the original draft. SS and CC: performed the writing review and editing. All the authors reviewed and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflict of interest to disclose, either financial or non-financial.
Additional information
Communicated by Michael Polymenis.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sousa, J.C., Sillankorva, S., Faustino, A. et al. Suggestion for a new bacteriophage genus for the Klebsiella pneumoniae phage vB_KpnS-Carvaje. Curr Genet 68, 393–406 (2022). https://doi.org/10.1007/s00294-022-01242-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00294-022-01242-2