Abstract
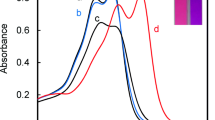
We report a new sensing strategy for Zn2+ by a colorimetric chemosensor based on commercially available negatively charged pyrogallol red (PR) and positively charged poly(diallyldimethylammonium chloride) (PDADMAC) ensemble in perfect aqueous solution at pH 7.0. The addition of ten equivalents of PDADMAC to PR resulted in PR aggregation by electrostatic interactions with PDADMAC. PR and PDADMAC ensemble showed a selective and distinct color change from red to blue in the presence of Zn2+ over other competitive metal ions. The color change was clearly visible to the naked eye, corresponding to a large red shift in the absorption peak from 544 to 595 nm. This result indicated that the PR and PDADMAC ensemble can serve as a potential candidate of a naked-eye chemosensor for Zn2+ in aqueous solution. This study demonstrates that the combination of organic dyes and oppositely charged polyelectrolytes is a versatile and useful tool, and shows great potential for the determination of important analytes in environmental and biological systems.











Similar content being viewed by others
References
Quang DT, Kim JS (2010) Fluoro- and chromogenic chemodosimeters for heavy metal ion detection in solution and biospecimens. Chem Rev 110:6280–6301
Sharma H, Kaur N, Singh A, Kuwar A, Singh N (2016) Optical chemosensors for water sample analysis. J Mater Chem C 4:5154–5194
Jeong Y, Yoon J (2012) Recent progress on fluorescent chemosensors for metal ions. Inorg Chim Acta 381:2–14
Outten CE, O’Halloran TV (2001) Femtomolar sensitivity of metalloregulatory proteins controlling zinc homeostasis. Science 292:2488–2492
Berg JM, Shi Y (1996) The galvanization of biology: a growing appreciation for the roles of zinc. Science 271:1081–1085
Frederickson CJ, Koh JY, Bush AI (2005) The neurobiology of zinc in health and disease. Nat Rev Neurosci 6:449–462
Assaf SY, Chung SH (1984) Release of endogenous Zn2+ from brain-tissue during activity. Nature 308:734–736
Bush AI, Pettingell WH, Multhaup G, Paradis M, Vonsattel JP, Gusella JF, Beyreuther K, Masters CL, Tanzi RE (1994) Rapid induction of Alzheimer A beta amyloid formation by zinc. Science 265:1464–1467
Cuajungco MP, Lees GJ (1997) Zinc metabolism in the brain: relevance to human neurodegenerative disorders. Neurobiol Dis 4:137–169
Onis M, Frongillo EA, Blcssner M (2000) Is malnutrition declining? An analysis of changes in levels of child malnutrition since 1980. Bull WHO 78:1222–1233
Walker CF, Black RE (2004) Zinc and the risk for infectious disease. Annu Rev Nutr 24:255–275
Wang H, Sun CL, Yue YH, Yin FF, Jiang JQ, Wu HR, Zhang HL (2013) New molecular probe for the selective detection of zinc ion. Analyst 138:5576–5579
Razi CH, Akelma AZ, Akin O, Kocak M, Ozdemir O, Celik A, Kislal FM (2012) Hair zinc and selenium levels in children with recurrent wheezing. Pediatr Pulmonol 47:1185–1191
Priya MDL, Geetha A (2011) Level of trace elements (copper, zinc, magnesium and selenium) and toxic elements (lead and mercury) in the hair and nail of children with autism. Biol Trace Elem Res 142:148–158
Buldini PL, Ferri D, Sharma JL (1997) Determination of some inorganic species in edible vegetable oils and fats by ion chromatography. J Chromatogr A 789:549–555
Hu QF, Yang GY, Zhao YY, Yin JY (2003) Determination of copper, nickel, cobalt, silver, lead, cadmium, and mercury ions in water by solid-phase extraction and the RP-HPLC with UV–Vis detection. Anal Bioanal Chem 375:831–835
Yue W, Bange A, Riehl BL, Johnson JM, Papautsky I, Heineman WR (2013) The application of nafion metal catalyst free carbon nanotube modified gold electrode: voltammetric zinc detection in serum. Electroanalysis 25:2259–2267
Carter KP, Young AM, Palmer AE (2014) Fluorescent sensors for measuring metal ions in living systems. Chem Rev 114:4564–4601
Xu Z, Kim GH, Han SJ, Jou MJ, Lee C, Shin I, Yoon J (2009) An NBD-based colorimetric and fluorescent chemosensor for Zn2+ and its use for detection of intracellular zinc ions. Tetrahedron 65:2307–2312
Kaur P, Kaur S, Mahajan A, Singh K (2008) Highly selective colorimetric sensor for Zn2+ based on hetarylazo derivative. Inorg Chem Commun 11:626–629
Du P, Lippard SJ (2010) A highly selective turn-on colorimetric, red fluorescent sensor for detecting mobile zinc in living cells. Inorg Chem 49:10753–10755
Ivanov VM, Mamedov AM (2006) 3,4,5-Trihydroxyfluorones as analytical reagents. J Anal Chem 61:1040–1062
Ivanov VM, Mamedov AM (2006) Pyrogallol red and bromopyrogallol red in new optical methods for the determination of molybdenum(VI) and tungsten(VI). J Anal Chem 61:261–268
Pelit L, Koçak S, Pelit FO, Turkmena H, Ertas FN (2013) A spectrophotometric method for determination of molybdenum in water samples by using pyrogallol red and a water soluble ionic liquid. Anal Methods 5:5792–5798
Higby K, Suiter CR, Silerkhodr T (1995) A comparison between two screening methods for detection of microproteinuria. Am J Obstet Gynecol 173:1111–1114
Behr S, Trumel C, Palanche F, Braun JP (2003) Assessment of a pyrogallol red technique for total protein measurement in the cerebrospinal fluid of dogs. J Small Anim Pract 44:530–533
Ensafi AA, Khayamian T, Khaloo SS (2004) Application of adsorptive cathodic differential pulse stripping method for simultaneous determination of copper and molybdenum using pyrogallol red. Anal Chim Acta 505:201–207
Rojas C, Arancibia V, Gómez M, Nagles E (2012) Adsorptive stripping voltammetric determination of cobalt in the presence of nickel and zinc using pyrogallol red as chelating agent. Int J Electrochem Sci 7:979–990
Safavi A, Shams E (1999) Determination of trace amounts of copper(II) by adsorptive stripping voltammetry of its complex with pyrogallol red. Anal Chim Acta 385:265–272
Arancibia V, Muñoz C (2007) Determination of aluminium in water samples by adsorptive cathodic stripping voltammetry in the presence of pyrogallol red and a quaternary ammonium salt. Talanta 73:546–552
Guo H, Li Y, Chen X, Nie L, He N (2005) Determination of trace antimony (III) by adsorption voltammetry at carbon paste electrode. Sensors 5:284–292
Sharma RK, Chelladurai C, Tiwari AD, Rajor HK, Mehta S, Otsuka M (2008) Studies on inhibition mechanism of transcription factor NF-κB and DNA binding by chelator pyrogallol red on the basis of its interaction with metal ions. Bioorg Med Chem 16:9018–9022
Ensafi AA, Aboutalebi A (2005) A versatile stable cobalt optical sensor based on pyrogallol red immobilization on cellulose acetate film. Sens Actuators B 105:479–483
Butler GB, Angelo RJ (1957) Preparation and polymerization of unsaturated quaternary ammonium compounds VIII A proposed alternating intramolecular-intermolecular chain propagation. J Am Chem Soc 79:3128–3131
Assem Y, Chaffey-Millar H, Barner-Kowollik C, Wegner G, Agarwal S (2007) Controlled/living ring-closing cyclopolymerization of diallyldimethylammonium chloride via the reversible addition fragmentation chain transfer process. Macromolecules 2007:3907–3913
Wang Y, Chen J, Jiao H, Chen Y, Li W, Zhang Q, Yu C (2013) Polymer-templated perylene-probe noncovalent self-assembly: a new strategy for label-free ultrasensitive fluorescence turn-on biosensing. Chem Eur J 19:12846–12852
Dubas ST, Limsavarn L, Iamsamai C, Potiyaraj P (2006) Assembly of polyelectrolyte multilayers on nylon fibers. J Appl Polym Sci 101:3286–3290
Job P (1928) Formation and stability of inorganic complexes in solution. Ann Chim-Puris 9:113–203
Irving HMNH, Freiser H, West TS (1978) IUPAC compendium of analytical nomenclature, definitive rules. Pergamon Press, Oxford
Benesi AH, Hildebrand HJ (1949) A spectrophotometric investigation of the interaction of iodine with aromatic hydrocarbons. J Am Chem Soc 71:2703–2707
Dahm KG, Guerra KL, Xu P, Drewes JE (2011) Composite geochemical database for coalbed methane produced water quality in the rocky mountain region. Environ Sci Technol 45:7655–7663
WHO (2008) Guidelines for drinking water quality, Geneva
Kumar YP, King P, Prasad VSKR (2006) Zinc biosorption on Tectona grandis L.f. leaves biomass:equilibrium and kinetic studies. Chem Eng J 124:63–70
Kim KB, Kim H, Song EJ, Kim S, Noh I, Kim C (2013) A cap-type schiff base acting as a fluorescence sensor for zinc(II) and a colorimetric sensor for iron(II), copper(II), and zinc(II) in aqueous media. Dalton Trans 42:16569–16577
Tsikalas GK, Lazarou P, Klontzas E, Pergantis SA, Spanopoulos I, Trikalitis PN, Froudakis GE, Katerinopoulos HE (2014) A “turn-on”-turning-to-ratiometric sensor for zinc(II) ions in aqueous media. RSC Adv 4:693–696
Kim KB, Kim H, Song EJ, Kim S, Noh I, Kim C (2013) A cap-type schiff base acting as a fluorescence sensor for zinc(II) and a colorimetric sensor for iron(II), copper(II), and zinc(II) in aqueous media. Dalton Trans 42:16569–16577
Kim TS, Lee JJ, Lee SY, Jo TG, Kim C (2016) A highly sensitive benzimidazole-based chemosensor for the colorimetric detection of Fe(II) and Fe(III) and the fluorometric detection of Zn(II) in aqueous media. RSC Adv 6:61505–61515
Li M, Lu HY, Liu RL, Chen JD, Chen CF (2012) Turn-on fluorescent sensor for selective detection of Zn2+, Cd2+, and Hg2+ in water. J Org Chem 77:3670–3673
Mikata Y, Kawata K, Takeuchi S, Nakanishi K, Konno H, Itami S, Yasuda K, Tamotsu S, Burdette SC (2014) Isoquinoline-derivatized tris(2-pyridylmethyl)-amines as fluorescent zinc sensors with strict Zn2+/Cd2+ selectivity. Dalton Trans 43:10751–10759
Aragoni MC, Arca M, Bencini A, Caltagirone C, Garau A, Isaia F, Light ME, Lippolis V, Lodeiro C, Mameli M, Montis R, Mostallino MC, Pintusa A, Puccioni S (2013) Zn2+/Cd2+ optical discrimination by fluorescent chemosensors based on 8-hydroxyquinoline derivatives and sulfur-containing macrocyclic units. Dalton Trans 42:14516–14530
Madden T, Datye A, Fulton M, Prairie M, Majumdar S, Stange B (1997) Oxidation of metal-EDTA complexes by TiO2 photocatalysis. Environ Sci Technol 31:3475–3481
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sakamaki, M., Aikawa, S. & Fukushima, Y. Colorimetric chemosensor for Zn2+ based on pyrogallol red and poly(diallyldimethylammonium chloride) in aqueous solution. Polym. Bull. 75, 1667–1680 (2018). https://doi.org/10.1007/s00289-017-2119-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-017-2119-7