Abstract
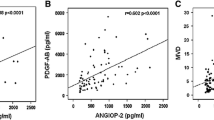
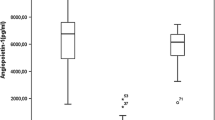
Increased angiogenesis in BCR-ABL1 negative myeloproliferative neoplasms (MPNs) has been recognized, but its connection with clinical and molecular markers needs to be defined. The aims of study were to (1) assess bone marrow (BM) angiogenesis measured by microvessel density (MVD) using CD34 and CD105 antibodies; (2) analyze correlation of MVD with plasma angiogenic factors including vascular endothelial growth factor, basic fibroblast growth factor, and interleukin-8; (3) examine the association of MVD with clinicopathological and molecular markers. We examined 90 de novo MPN patients (30 polycythemia vera (PV), primary myelofibrosis (PMF), essential thrombocythemia (ET)) and 10 age-matched controls. MVD was analyzed by immunohistochemistry “hot spot” method, angiogenic factors by immunoassay and JAK2V617F, and CALR mutations by DNA sequencing and allelic PCR. MVD was significantly increased in MPNs compared to controls (PMF > PV > ET). Correlation between MVD and plasma angiogenic factors was found in MPNs. MVD was significantly increased in patients with JAK2V617F mutation and correlated with JAK2 mutant allele burden (CD34-MVD: ρ = 0.491, p < 0.001; CD105-MVD: ρ = 0.276, p = 0.02) but not with CALR mutation. MVD correlated with leukocyte count, serum lactate dehydrogenase, hepatomegaly, and splenomegaly. BM fibrosis was significantly associated with CD34-MVD, CD105-MVD, interleukin-8, and JAK2 mutant allele burden. JAK2 homozygote status had positive predictive value (100%) for BM fibrosis. Patients with prefibrotic PMF had significantly higher MVD than patients with ET, and we could recommend MVD to be additional histopathological marker to distinguish these two entities. This study also highlights the strong correlation of MVD with plasma angiogenic factors, JAK2 mutant allele burden, and BM fibrosis in MPNs.





Similar content being viewed by others
References
Skoda RC, Duek A, Grisouard J (2015) Pathogenesis of myeloproliferative neoplasms. Exp Hematol 43:599–608
Kralovics R, Passamonti F, Buser AS, Teo SS, Tiedt R, Passweg JR et al (2005) A gain-of-function mutation of JAK2 in myeloproliferative disorders. N Engl J Med 352:1779–1790
Pardanani AD, Levine RL, Lasho T, Pikman Y, Mesa RA, Wadleigh M et al (2006) MPL515 mutations in myeloproliferative and other myeloid disorders: a study of 1182 patients. Blood 108:3472–3476
Klampfl T, Gisslinger H, Harutyunyan AS, Nivarthi H, Rumi E, Milosevic JD et al (2013) Somatic mutations of calreticulin in myeloproliferative neoplasms. N Engl J Med 369:2379–2390
Roskoski R Jr (2007) Vascular endothelial growth factor (VEGF) signaling in tumor progression. Crit Rev Oncol Hematol 62(3):179–213
Folkman J (1995) Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat Med 1:27–31
Rajkumar SV, Mesa RA, Fonseca R, Schroeder G, Plevak MF, Dispenzieri A et al (2002) Bone marrow angiogenesis in 400 patients with monoclonal gammopathy of undetermined significance, multiple myeloma, and primary amyloidosis. Clin Cancer Res 8:2210–2216
Padro T, Ruiz S, Bieker R, Bürger H, Steins M, Kienast J et al (2000) Increased angiogenesis in the bone marrow of patients with acute myeloid leukemia. Blood 95:2637–2644
Aguayo A, Armillas-Canseco FM, Martínez-Baños D (2011) Antiangiogenesis in myelodysplastic syndrome. Curr Cancer Drug Targets 11(9):1044–1052
Paesler J, Gehrke I, Poll-Wolbeck SJ, Kreuzer KA (2012) Targeting the vascular endothelial growth factor in hematologic malignancies. Eur J Haematol 89(5):373–384
Boveri E, Passamonti F, Rumi E, Pietra D, Elena C, Arcaini L et al (2008) Bone marrow microvessel density in chronic myeloproliferative disorders: a study of 115 patients with clinicopathological and molecular correlations. Br J Haematol 140(2):162–168
Kvasnicka HM, Thiele J (2004) Bone marrow angiogenesis: methods of quantification and changes evolving in chronic myeloproliferative disorders. Histol Histopathol 19(4):1245–1260
Gianelli U, Vener C, Raviele PR, Savi F, Somalvico F, Calori R et al (2007) VEGF expression correlates with microvessel density in Philadelphia chromosome negative chronic myeloproliferative disorders. Am J Clin Pathol 128:966–973
Musolino C, Calabro L, Bellomo G, Martello F, Loteta B, Pezzano C et al (2002) Soluble angiogenic factors: implications for chronic myeloproliferative disorders. Am J Hematol 69(3):159–163
Bauerle KT, Schweppe RE, Lund G, Kotnis G, Deep G, Agarwal R et al (2013) Nuclear factor κB-dependent regulation of angiogenesis, and metastasis in an in vivo model of thyroid cancer is associated with secreted interleukin-8. Leukemia 27(8):1697–1706
Swerdlow S, Campo E, Harris N, Jaffe E, Pileri S, Stein H et al (2008) WHO classification of tumors of haematopoietic and lymphoid tissues. IARC press, Lyon
Panteli K, Zagorianakou N, Bai M, Katsaraki A, Agnantis NJ, Bourantas K (2004) Angiogenesis in chronic myeloproliferative diseases detected by CD34 expression. Eur J Haematol 72(6):410–415
Lippert E, Boissinot M, Kralovics R, Girodon F, Dobo I, Praloran V et al (2006) The JAK2-V617F mutation is frequently present at diagnosis in patients with essential thrombocythemia and polycythemia vera. Blood 108(6):1865–1867
Furtado LV, Weigelin HC, Elenitoba-Johnson KS, Betz BL (2013) Detection of MPL mutations by a novel allele-specific PCR-based strategy. J Mol Diagn 15(6):810–818
Ponzoni M, Savage DG, Ferreri AJ, Pruneri G, Viale G, Servida P et al (2004) Chronic idiopathic myelofibrosis: independent prognostic importance of bone marrow microvascular density evaluated by CD105 (endoglin) immunostaining. Mod Pathol 17(12):1513–1520
Medinger M, Skoda R, Gratwohl A, Theocharides A, Buser A, Heim D et al (2009) Angiogenesis and vascular endothelial growth factor−/receptor expression in myeloproliferative neoplasms: correlation with clinical parameters and JAK2-V617F mutational status. Br J Haematol 146(2):150–157
Boiocchi L, Vener C, Savi F, Bonoldi E, Moro A, Fracchiolla NS et al (2011) Increased expression of vascular endothelial growth factor receptor 1 correlates with VEGF and microvessel density in Philadelphia chromosome-negative myeloproliferative neoplasms. J Clin Pathol 64(3):226–231
Panteli K, Bai M, Hatzimichael E, Zagorianakou N, Agnantis NJ, Bourantas K (2007 Dec) Serum levels, and bone marrow immunohistochemical expression of, vascular endothelial growth factor in patients with chronic myeloproliferative diseases. Hematology 12(6):481–486
Bock O, Schlue J, Lehmann U, von Wasielewski R, Langer F, Kreipe H (2002) Megakaryocytes from chronic myeloproliferative disorders show enhanced nuclear bFGF expression. Blood 100:2274–2275
Tefferi A, Vaidya R, Caramazza D, Lasho T, Pardanani A (2011) Circulating interleukin (IL)- 8, IL-2R, IL-12, and IL-15 levels are independently prognostic in primary myelofibrosis: a comprehensive cytokine profiling study. J Clin Oncol 29:1356–1363
Murphy P, Ahmed N, Hassan HT (2002) Increased serum levels of vascular endothelial growth factor correlate with splenomegaly in polycythemia vera. Leuk Res 26(11):1007–1010
Di Raimondo F, Azzaro MP, Palumbo GA, Bagnato S, Stagno F, Giustolisi GM et al (2001) Elevated vascular endothelial growth factor (VEGF) serum levels in idiopathic myelofibrosis. Leukemia 15:976–980
Martyré MC, Le Bousse-Kerdiles MC, Romquin N, Chevillard S, Praloran V, Demory JL et al (1997) Elevated levels of basic fibroblast growth factor in megakaryocytes and platelets from patients with idiopathic myelofibrosis. Br J Haematol 97:441–448
Mesa RA, Hanson CA, Rajkumar SV, Schroeder G, Tefferi A (2000) Evaluation and clinical correlations of bone marrow angiogenesis in myelofibrosis with myeloid metaplasia. Blood 96(10):3374–3380
Ni H, Barosi G, Hoffman R (2006) Quantitative evaluation of bone marrow angiogenesis in idiopathic myelofibrosis. Am J Clin Pathol 126:241–247
Steurer M, Zoller H, Augustin F, Fong D, Heiss S, Strasser-Weippl K et al (2007) Increased angiogenesis in chronic idiopathic myelofibrosis: vascular endothelial growth factor as a prominent angiogenic factor. Hum Pathol 38:1057–1064
Barbui T, Thiele J, Passamonti F, Rumi E, Boveri E, Ruggeri M et al (2011) Survival and disease progression in essential thrombocythemia are significantly influenced by accurate morphologic diagnosis: an international study. J Clin Oncol 29(23):3179–3184
Deng YQ, Zhao H, Ma AL, Zhou JY, Xie SB, Zhang XQ et al (2015) Selected cytokines serve as potential biomarkers for predicting liver inflammation and fibrosis in chronic hepatitis B patients with normal to mildly elevated aminotransferases. Medicine (Baltimore) 94(45):e2003. doi:10.1097/MD.0000000000002003
Lee JS, Shin JH, Choi BS (2015) Serum levels of IL-8 and ICAM-1 as biomarkers for progressive massive fibrosis in coal workers’ pneumoconiosis. J Korean Med Sci 30(2):140–144
Hermouet S, Godard A, Pineau D et al (2002) Abnormal production of interleukin (IL)-11 and IL-8 in polycythaemia vera. Cytokine 20:178–183
Kvasnicka HM, Beham-Schmid C, Bob R, Dirnhofer S, Hussein K, Kreipe H et al (2016) Problems and pitfalls in grading of bone marrow fibrosis, collagen deposition and osteosclerosis - a consensus-based study. Histopathology 68(6):905–915
Lopes FC, Traina F, Almeida CB, Leonardo FC, Franco-Penteado CF, Garrido VT (2015) Key endothelial cell angiogenic mechanisms are stimulated by the circulating milieu in sickle cell disease and attenuated by hydroxyurea. Haematologica 100(6):730–739
Acknowledgements
This work was supported by a grant from Ministry of education, science, and technological development of the Republic of Serbia (175053) and Swiss National Science Foundation through Joint research project (SCOPES) IZ73Z0_152420/1. We are grateful to pathologists Maja Perunicic-Jovanovic, Tatjana Terzic, and Ljubomir Jakovic who have done histolopathological analysis and evaluation of bone marrow fibrosis.
Author contribution
Danijela Lekovic, Mirjana Gotic, Radek Skoda, and Vladan Cokic designed the study, performed the research, analyzed data and wrote manuscript. Bojana Cokic- Beleslin, Olivera Mitrovic-Ajtic, Dijana Sefer, Tijana Suboticki, Dragana Markovic, Marijana Buac, Milos Diklic, and Ronny Nienhold performed research. Natasa Milic performed statistical analyses.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Informed consent
Informed consent was obtained from all patients for being included in the study.
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Supplemental Table 1.
List of antibodies used for analyzing microvessel density (DOCX 11 kb)
Supplemental table 2.
The following primers were used for allelic PCR (DOCX 11 kb)
Rights and permissions
About this article
Cite this article
Lekovic, D., Gotic, M., Skoda, R. et al. Bone marrow microvessel density and plasma angiogenic factors in myeloproliferative neoplasms: clinicopathological and molecular correlations. Ann Hematol 96, 393–404 (2017). https://doi.org/10.1007/s00277-016-2890-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-016-2890-9