Abstract
Introduction
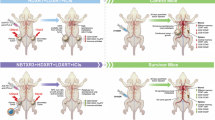
The current systemic anti-metastatic treatment is chemotherapy. Chemotherapy reacts mostly against replicating cells, which makes this therapy not specific. Moreover, resting cancer cells will not be destroyed. A better alternative is an engagement of the host immune system to react against tumor-associated antigens. An efficient immune-stimulating technique is an ablation of the tumor that results in the release of tumor antigens. Our ablation strategy is an innovative alpha-radiation-based technology, diffusing alpha-emitters radiation therapy (DaRT), which efficiently destroys local tumors and provides thereby an antigenic supply for antigen-presenting cells to stimulate T cells.
Methods
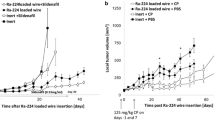
Mice bearing weakly immunogenic DA3 adenocarcinoma or highly immunogenic CT26 colon carcinoma were treated by DaRT. Anti-tumor immune responses following tumor destruction were evaluated by (1) the resistance to a tumor challenge; (2) scanning by a CT imaging device for elimination of lung metastases; (3) improved tumor control when combining DaRT with an immunoadjuvant (CpG).
Results
CT26 model: 63–77 % of DaRT-treated mice became resistant to a re-inoculated tumor compared to 29–33 % resistant mice in the control. DA3 model: (1) The growth rate of challenge tumors was the lowest in mice which their primary tumor was treated by DaRT. (2) Most (93 %) mice in the control group developed lung metastases compared to 56 % in the DaRT group. (3) Combining DaRT with CpG resulted in a better control of the primary tumor. Our study offers a technique to eliminate local and distant malignant cells, regardless of their replication status, by stimulating specific anti-tumor immunity through the supply of tumor antigens from the destroyed tumor.





Similar content being viewed by others
Abbreviations
- DaRT:
-
Diffusing alpha-emitters radiation therapy
- DCs:
-
Dendritic cells
- DMBA:
-
Dimethylbenzanthracene
- DMEM:
-
Dulbecco’s modified Eagle’s medium
- HBSS:
-
Hanks’ balanced salt solution
- NNMU:
-
N-Nitroso-N-methylurethane
- ODNs:
-
Oligodeoxynucleotides
- RPMI:
-
Roswell Park Memorial Institute
References
Weiss EM, Frey B, Rodel F, Herrmann M, Schlucker E, Voll RE, Fietkau R, Gaipl US (2010) Ex vivo– and in vivo–induced dead tumor cells as modulators of antitumor responses. Ann N.Y. Acad Sci 1209:109–117. doi:10.1111/j.1749-6632.2010.05743.x
Palucka K, Ueno H, Fay J, Banchereau J (2011) Dendritic cells and immunity against cancer. J Intern Med 269:64–73. doi:10.1111/j.1365-2796.2010.02317.x
Den Brok MH, Sutmuller RP, van der Voort R, Bennink EJ, Figdor CG, Ruers TJ, Adema GJ (2004) In situ tumor ablation creates an antigen source for the generation of antitumor immunity. Cancer Res 64:4024–4029
Arazi L, Cooks T, Schmidt M, Keisari Y, Kelson I (2007) Treatment of solid tumours by interstitial release of recoiling short-lived alpha emitters. Phys Med Biol 52:5025–5042
Cooks T, Arazi L, Schmidt M, Marshak G, Kelson I, Keisari Y (2008) Growth retardation and destruction of experimental Squamous cell carcinoma by interstitial radioactive wires releasing diffusing alpha-emitting atoms. Int J Cancer 122:1657–1664
Cooks T, Arazi L, Efrati M, Schmidt M, Marshak G, Kelson I, Keisari Y (2009) Interstitial wires releasing diffusing alpha-emitters combined with chemotherapy improved local tumor control and survival in squamous cell carcinoma bearing mice. Cancer 115:1791–1801. doi:10.1002/cncr.24191
Cooks T, Schmidt M, Bittan H, Lazarov E, Arazi L, Kelson I, Keisari Y (2009) Local control of lung derived tumors by diffusing alpha-emitting atoms released from intratumoral wires loaded with Radium-224. Int J Radiat Oncol Biol Phys 74:966–973. doi:10.1016/j.trsl.2011.08.009
Arazi L, Cooks T, Schmidt M, Keisari Y, Kelson I (2010) The treatment of solid tumors by alpha emitters released from 224Ra-loaded sources: internal dosimetry analysis. Phys Med Biol 55:1203–1218. doi:10.1088/0031-9155/55/4/020
Horev-Drori G, Cooks T, Bittan H, Lazarov E, Schmidt M, Arazi L, Efrati M, Kelson I, Keisari Y (2012) Local control of experimental malignant pancreatic tumors by treatment with a combination of chemotherapy and intratumoral 224Radium-loaded wires releasing alpha-emitting atoms. Transl Res 159:32–41. doi:10.1016/j.ijrobp.2009.02.063
Cooks T, Tal M, Raab S, Efrati M, Reitkopf S, Lazarov E, Etzyoni R, Schmidt M, Arazi L, Kelson I, Keisari Y (2012) Intratumoral 224Ra-loaded wires spread alpha-emitters inside solid human tumors in athymic mice achieving tumor control. Anticancer Res 32:5315–5321
Coulie PG, Van den Eynde BJ, Van der Bruggen P, Boon T (2014) Tumour antigens recognized by T lymphocytes: at the core of cancer immunotherapy. Nat Rev Cancer 14:135–146. doi:10.1038/nrc3670
Aranda F, Llopiz D, Díaz-Valdés N, Riezu-Boj JI, Bezunartea J, Ruiz M, Martínez M, Durantez M, Mansilla C, Prieto J, Lasarte JJ, Borrás-Cuesta F, Sarobe P (2011) Adjuvant combination and antigen targeting as a strategy to induce polyfunctional and high-avidity T-cell responses against poorly immunogenic tumors. Cancer Res 71:3214–3224. doi:10.1158/0008-5472.CAN-10-3259
Goldstein M, Varghese B, Brody J, Rajapaksa R, Kohrt H, Czerwinski D, Levy S, Levy RA (2011) CpG-loaded tumor cell vaccine induces antitumor CD4 T cells that are effective in adoptive therapy for large and established tumors. Blood 117:118–127. doi:10.1182/blood-2010-06-288456
Nierkens S, den Brok MH, Garcia Z, Togher S, Wagenaars J, Wassink M, Boon L, Ruers TJ, Figdor CG, Schoenberger SP, Adema GJ, Janssen EM (2011) Immune adjuvant efficacy of CpG oligonucleotide in cancer treatment is founded specifically upon TLR9 function in plasmacytoid dendritic cells. Cancer Res 71:6428–6437. doi:10.1158/0008-5472.CAN-11-2154
Nierkens S, den Brok MH, Sutmuller RP, Grauer OM, Bennink E, Morgan ME, Figdor CG, Ruers TJ, Adema GJ (2008) In vivo colocalization of antigen and CpG within dendritic cells is associated with the efficacy of cancer immunotherapy. Cancer Res 68:5390–5396. doi:10.1158/0008-5472.CAN-07-6023
den Brok MH, Sutmuller RP, Nierkens S, Bennink EJ, Toonen LW, Figdor CG, Ruers TJ, Adema GJ (2006) Synergy between in situ cryoablation and TLR9 stimulation results in a highly effective in vivo dendritic cell vaccine. Cancer Res 66:7285–7292
Yu B, Kusmartsev S, Cheng F, Paolini M, Nefedova Y, Sotomayor E, Gabrilovich D (2003) Effective combination of chemotherapy and dendritic cell administration for the treatment of advanced-stage experimental breast cancer. Clin Cancer Res 9:285–294
Robinson M et al (2009) Novel immunocompetent murine tumor model for evaluation of conditionally replication-competent (oncolytic) murine adenoviral vectors. J Virol 83:3450–3462. doi:10.1128/JVI.02561-08
Formenti SC, Demaria S (2013) Combining radiotherapy and cancer immunotherapy: a paradigm shift. J Natl Cancer Inst 105:256–265. doi:10.1093/jnci/djs629
Lindner M, Schirrmacher V (2002) Tumour cell-dendritic cell fusion for cancer immunotherapy: comparison of therapeutic efficiency of polyethylen-glycol versus electro-fusion protocols. Eur J Clin Invest 32:207–217
Robinson M, Li B, Ge Y, Ko D, Yendluri S, Harding T, VanRoey M, Spindler KR, Jooss K (2009) Novel immunocompetent murine tumor model for evaluation of conditionally replication –competent (oncolytic) murine adenoviral vectors. J Virol 83:3450–3462. doi:10.1128/JVI.02561-08
Plotnikov A, Fishman D, Tichler T, Korenstein R, Keisari Y (2004) Low electric field enhanced chemotherapy can cure mice with CT-26 colon carcinoma and induce anti-tumour immunity. Clin Exp Immunol 138:410–416
Plotnikov A, Tichler T, Korenstein R, Keisari Y (2005) Involvement of the immune response in the cure of metastatic murine CT-26 colon carcinoma by low electric field-enhanced chemotherapy. Int J Cancer 117:816–824
Hodge JW, Guha C, Neefjes J, James L, Gulley (2008) Synergizing radiation therapy and immunotherapy for curing incurable cancers: opportunities and challenges. Oncology 22:1064–1084
Demaria S, Ng B, Devitt ML, Babb JS, Kawashima N, Liebes L, Formenti SC (2004) Ionizing radiation inhibition of distant untreated tumors (abscopal effect) is immune mediated. Int J Radiat Oncol Biol Phys 58:862–870
Jahrsdörfer B, Weiner GJ (2008) CpG oligodeoxynucleotides as immunotherapy in cancer. Update Cancer Ther 3:27–32. doi:10.1016/j.uct.2007.11.003
Sakuishi K, Ngiow SF, Sullivan JM, Teng MW, Kuchroo VK, Smyth MJ, Anderson AC (2013) TIM3 + FOXP3 + regulatory T cells are tissue-specific promoters of T-cell dysfunction in cancer. Oncoimmunology 2:1–13
Youn JI, Nagaraj S, Gabrilovich MI (2008) Subset of myeolid: derived suppressor cells in tumor-bearing mice. J Immunol 181:5791–5802
Stap J, Krawczyk PM, Van Oven CH, Barendsen GW, Essers J, Kanaar R, Aten JA (2008) Induction of linear tracks of DNA double-strand breaks by alpha-particle irradiation of cells. Nat Methods 5:261–266. doi:10.1038/nmeth.f.206
Keisari Y (2013) Tumor ablation: effects on systemic and local anti-tumor immunity and on other tumor-microenvironment interactions. Springer, Dordrecht
Gaipl US, Multhoff G, Scheithauer H, Lauber K, Hehlgans S, Frey B, Rödel F (2014) Kill and spread the word: stimulation of antitumor immune responses in the context of radiotherapy. Immunotherapy 6:597–610. doi:10.2217/imt.14.38
Frey B, Rubner Y, Kulzer L, Werthmöller N, Weiss EM, Fietkau R, Gaipl US (2014) Antitumor immune responses induced by ionizing irradiation and further immune stimulation. Cancer Immunol Immunother 63:29–36. doi:10.1007/s00262-013-1474-y
Acknowledgments
We thank Dr. Gideon Halpern for assistance with the statistical analysis. This work was supported in part by The Roberts-Guthman Chair in Immunopharmacology and The German-Israeli Foundation. This work was performed in partial fulfillment of the requirements toward a PhD degree of Hila Confino, Sackler Faculty of Medicine, Tel Aviv University.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Confino, H., Hochman, I., Efrati, M. et al. Tumor ablation by intratumoral Ra-224-loaded wires induces anti-tumor immunity against experimental metastatic tumors. Cancer Immunol Immunother 64, 191–199 (2015). https://doi.org/10.1007/s00262-014-1626-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-014-1626-8