Abstract
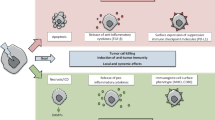
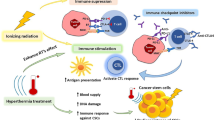
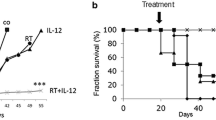
The therapy of cancer emerged as multimodal treatment strategy. The major mode of action of locally applied radiotherapy (RT) is the induction of DNA damage that triggers a network of events that finally leads to tumor cell cycle arrest and cell death. Along with this, RT modifies the phenotype of the tumor cells and their microenvironment. Either may contribute to the induction of specific and systemic antitumor immune responses. The latter are boosted when additional immune therapy (IT) is applied at distinct time points during RT. We will focus on therapy-induced necrotic tumor cell death that is immunogenic due to the release of damage-associated molecular patterns. Immune-mediated distant bystander (abscopal) effects of RT when combined with dendritic cell-based IT and the role of fractionation of radiation in the induction of immunogenic tumor cell death will be discussed. Autologous whole-tumor-cell-based vaccines generated by high hydrostatic pressure technology will be introduced and the influence of cytokines and the immune modulator AnnexinA5 on the ex vivo generated or in situ therapy-induced vaccine efficacy will be outlined. RT should be regarded as immune adjuvant for metastatic disease and as a tool for the generation of an in situ vaccine when applied at distinct fractionation doses or especially in combination with IT to generate immune memory against the tumor. To identify the most beneficial combination and chronology of RT with IT is presumably one of the biggest challenges of innovative tumor research and therapies.



Similar content being viewed by others
References
Weiss EM, Wunderlich R, Ebel N et al (2012) Selected anti-tumor vaccines merit a place in multimodal tumor therapies. Front Oncol 2:132. doi:10.3389/fonc.2012.00132
Gabrilovich DI, Ostrand-Rosenberg S, Bronte V (2012) Coordinated regulation of myeloid cells by tumours. Nat Rev Immunol 12:253–268. doi:10.1038/nri3175
Frey B, Rubner Y, Wunderlich R, Weiss EM, Pockley AG, Fietkau R, Gaipl US (2012) Induction of abscopal anti-tumor immunity and immunogenic tumor cell death by ionizing irradiation: implications for cancer therapies. Curr Med Chem 19:1751–1764
Apetoh L, Ghiringhelli F, Tesniere A et al (2007) Toll-like receptor 4-dependent contribution of the immune system to anticancer chemotherapy and radiotherapy. Nat Med 13:1050–1059. doi:10.1038/nm1622
Voll RE, Herrmann M, Roth EA, Stach C, Kalden JR, Girkontaite I (1997) Immunosuppressive effects of apoptotic cells. Nature 390:350–351. doi:10.1038/37022
Gregory CD, Pound JD (2010) Microenvironmental influences of apoptosis in vivo and in vitro. Apoptosis 15:1029–1049. doi:10.1007/s10495-010-0485-9
Lauber K, Ernst A, Orth M, Herrmann M, Belka C (2012) Dying cell clearance and its impact on the outcome of tumor radiotherapy. Front Oncol 2:116. doi:10.3389/fonc.2012.00116
Krysko DV, Garg AD, Kaczmarek A, Krysko O, Agostinis P, Vandenabeele P (2012) Immunogenic cell death and DAMPs in cancer therapy. Nat Rev Cancer 12:860–875. doi:10.1038/nrc3380
Hagemann T, Balkwill F, Lawrence T (2007) Inflammation and cancer: a double-edged sword. Cancer Cell 12:300–301. doi:10.1016/j.ccr.2007.10.005
Schildkopf P, Frey B, Ott OJ, Rubner Y, Multhoff G, Sauer R, Fietkau R, Gaipl US (2011) Radiation combined with hyperthermia induces HSP70-dependent maturation of dendritic cells and release of pro-inflammatory cytokines by dendritic cells and macrophages. Radiother Oncol 101:109–115. doi:10.1016/j.radonc.2011.05.056
Ma Y, Adjemian S, Mattarollo SR et al (2013) Anticancer chemotherapy-induced intratumoral recruitment and differentiation of antigen-presenting cells. Immunity 38:729–741. doi:10.1016/j.immuni.2013.03.003
Frey B, Gaipl US (2011) The immune functions of phosphatidylserine in membranes of dying cells and microvesicles. Semin Immunopathol 33:497–516. doi:10.1007/s00281-010-0228-6
Bondanza A, Zimmermann VS, Rovere-Querini P et al (2004) Inhibition of phosphatidylserine recognition heightens the immunogenicity of irradiated lymphoma cells in vivo. J Exp Med 200:1157–1165. doi:10.1084/jem.20040327
Gaipl US, Beyer TD, Baumann I, Voll RE, Stach CM, Heyder P, Kalden JR, Manfredi A, Herrmann M (2003) Exposure of anionic phospholipids serves as anti-inflammatory and immunosuppressive signal–implications for antiphospholipid syndrome and systemic lupus erythematosus. Immunobiology 207:73–81. doi:10.1078/0171-2985-00217
Frey B, Schildkopf P, Rodel F, Weiss EM, Munoz LE, Herrmann M, Fietkau R, Gaipl US (2009) AnnexinA5 renders dead tumor cells immunogenic–implications for multimodal cancer therapies. J Immunotoxicol 6:209–216. doi:10.3109/15476910903204058
Michels T, Shurin GV, Naiditch H, Sevko A, Umansky V, Shurin MR (2012) Paclitaxel promotes differentiation of myeloid-derived suppressor cells into dendritic cells in vitro in a TLR4-independent manner. J Immunotoxicol 9:292–300. doi:10.3109/1547691x.2011.642418
Timke C, Winnenthal HS, Klug F et al (2011) Randomized controlled phase I/II study to investigate immune stimulatory effects by low dose radiotherapy in primarily operable pancreatic cancer. BMC Cancer 11:134. doi:10.1186/1471-2407-11-134
Postow MA, Callahan MK, Barker CA et al (2012) Immunologic correlates of the abscopal effect in a patient with melanoma. N Engl J Med 366:925–931. doi:10.1056/NEJMoa1112824
Darby S, McGale P, Correa C et al (2011) Effect of radiotherapy after breast-conserving surgery on 10-year recurrence and 15-year breast cancer death: meta-analysis of individual patient data for 10,801 women in 17 randomised trials. Lancet 378:1707–1716. doi:10.1016/s0140-6736(11)61629-2
Demaria S, Ng B, Devitt ML, Babb JS, Kawashima N, Liebes L, Formenti SC (2004) Ionizing radiation inhibition of distant untreated tumors (abscopal effect) is immune mediated. Int J Radiat Oncol Biol Phys 58:862–870. doi:10.1016/j.ijrobp.2003.09.012
Frey B, Lotter M, Stangl S, Rodel F, Fietkau R, Gaipl US (2012) Adaptive and innate immune cells contribute to reduced tumor progression after local irradiation of CT26 tumors in Balb/c mice. Strahlenther Onkol 188 [Abstractband zur 18. Jahrestagung der Deutschen Gesellschaft für Radioonkologie (DEGRO)]:132
Bogdandi EN, Balogh A, Felgyinszki N, Szatmari T, Persa E, Hildebrandt G, Safrany G, Lumniczky K (2010) Effects of low-dose radiation on the immune system of mice after total-body irradiation. Radiat Res 174:480–489. doi:10.1667/rr2160.1
Wunderlich R, Frischholz B, Roedel F, Fietkau R, Frey B, Gaipl US (2013) Ionising radiation with single doses up to clinical relevant 2Gy does not influence viability and phagocytosis capability of single activated macrophages of radiosensitive BALB/c mice. Strahlenther Onkol 189 [Abstractband zur 19. Jahrestagung der Deutschen Gesellschaft für Radioonkologie (DEGRO)]:122
Bauer M, Goldstein M, Heylmann D, Kaina B (2012) Human monocytes undergo excessive apoptosis following temozolomide activating the ATM/ATR pathway while dendritic cells and macrophages are resistant. PLoS One 7:e39956. doi:10.1371/journal.pone.0039956
Bauer M, Goldstein M, Christmann M, Becker H, Heylmann D, Kaina B (2011) Human monocytes are severely impaired in base and DNA double-strand break repair that renders them vulnerable to oxidative stress. Proc Natl Acad Sci USA 108:21105–21110. doi:10.1073/pnas.1111919109
Lee Y, Auh SL, Wang Y et al (2009) Therapeutic effects of ablative radiation on local tumor require CD8+ T cells: changing strategies for cancer treatment. Blood 114:589–595. doi:10.1182/blood-2009-02-206870
Gupta A, Probst HC, Vuong V et al (2012) Radiotherapy promotes tumor-specific effector CD8+ T cells via dendritic cell activation. J Immunol 189:558–566. doi:10.4049/jimmunol.1200563
Frey B, Weber J, Kohn K, Lotter M, Stangl S, Rodel F, Fietkau R, Gaipl US (2013) Hypofractionated local radiotherapy induces a timely restricted infiltration of immune cells in CT26 colon cancer tumors of BALB/c mice. Strahlenther Onkol 189 [Abstractband zur 19. Jahrestagung der Deutschen Gesellschaft für Radioonkologie (DEGRO)]:116
Ma Y, Aymeric L, Locher C et al (2011) Contribution of IL-17-producing gamma delta T cells to the efficacy of anticancer chemotherapy. J Exp Med 208:491–503. doi:10.1084/jem.20100269
Takeshima T, Chamoto K, Wakita D, Ohkuri T, Togashi Y, Shirato H, Kitamura H, Nishimura T (2010) Local radiation therapy inhibits tumor growth through the generation of tumor-specific CTL: its potentiation by combination with Th1 cell therapy. Cancer Res 70:2697–2706. doi:10.1158/0008-5472.can-09-2982
Matsunaga A, Ueda Y, Yamada S, Harada Y, Shimada H, Hasegawa M, Tsujii H, Ochiai T, Yonemitsu Y (2010) Carbon-ion beam treatment induces systemic antitumor immunity against murine squamous cell carcinoma. Cancer 116:3740–3748. doi:10.1002/cncr.25134
Weiss EM, Meister S, Janko C et al (2010) High hydrostatic pressure treatment generates inactivated mammalian tumor cells with immunogeneic features. J Immunotoxicol 7:194–204. doi:10.3109/15476911003657414
Weiss JM, Subleski JJ, Wigginton JM, Wiltrout RH (2007) Immunotherapy of cancer by IL-12-based cytokine combinations. Expert Opin Biol Ther 7:1705–1721. doi:10.1517/14712598.7.11.1705
Kerkar SP, Goldszmid RS, Muranski P et al (2011) IL-12 triggers a programmatic change in dysfunctional myeloid-derived cells within mouse tumors. J Clin Invest 121:4746–4757. doi:10.1172/jci58814
Ni J, Miller M, Stojanovic A, Garbi N, Cerwenka A (2012) Sustained effector function of IL-12/15/18-preactivated NK cells against established tumors. J Exp Med 209:2351–2365. doi:10.1084/jem.20120944
Hodge JW, Sharp HJ, Gameiro SR (2012) Abscopal regression of antigen disparate tumors by antigen cascade after systemic tumor vaccination in combination with local tumor radiation. Cancer Biother Radiopharm 27:12–22. doi:10.1089/cbr.2012.1202
Formenti SC, Demaria S (2013) Combining radiotherapy and cancer immunotherapy: a paradigm shift. J Natl Cancer Inst 105:256–265. doi:10.1093/jnci/djs629
Weng D, Song B, Koido S, Calderwood SK, Gong J (2013) Immunotherapy of radioresistant mammary tumors with early metastasis using molecular chaperone vaccines combined with ionizing radiation. J Immunol 191:755–763. doi:10.4049/jimmunol.1203286
Wheeler CJ, Black KL, Liu G et al (2008) Vaccination elicits correlated immune and clinical responses in glioblastoma multiforme patients. Cancer Res 68:5955–5964. doi:10.1158/0008-5472.can-07-5973
Van Gool S, Maes W, Ardon H, Verschuere T, Van Cauter S, De Vleeschouwer S (2009) Dendritic cell therapy of high-grade gliomas. Brain Pathol 19:694–712. doi:10.1111/j.1750-3639.2009.00316.x
Muth C, Schmidt V, Rubner Y, Fietkau R, Semrau S, Buslei R, Gaipl US (2013) Comparative analysis of the danger signals HMGB1, Hsp70, and Calreticulin in primary tumors and relapses of glioblastoma patients. Strahlenther Onkol 189 [Abstractband zur 19. Jahrestagung der Deutschen Gesellschaft für Radioonkologie (DEGRO)]:124
Rubner Y, Sieber R, Buslei R, Fietkau R, Frey B, Gaipl US (2013) Fractionated radiotherapy and not chemotherapy with temozolomide and/or valproic acid is the main stimulus for the induction of immunogenic glioma cell death in p53 mutated glioma cell lines. Strahlenther Onkol 189 [Abstractband zur 19. Jahrestagung der Deutschen Gesellschaft für Radioonkologie (DEGRO)]: 115–116
Gastpar R, Gehrmann M, Bausero MA, Asea A, Gross C, Schroeder JA, Multhoff G (2005) Heat shock protein 70 surface-positive tumor exosomes stimulate migratory and cytolytic activity of natural killer cells. Cancer Res 65:5238–5247. doi:10.1158/0008-5472.can-04-3804
Durante M, Reppingen N, Held KD (2013) Immunologically augmented cancer treatment using modern radiotherapy. Trends Mol Med. doi:10.1016/j.molmed.2013.05.007
Dewan MZ, Galloway AE, Kawashima N, Dewyngaert JK, Babb JS, Formenti SC, Demaria S (2009) Fractionated but not single-dose radiotherapy induces an immune-mediated abscopal effect when combined with anti-CTLA-4 antibody. Clin Cancer Res 15:5379–5388. doi:10.1158/1078-0432.ccr-09-0265
Schaue D, Ratikan JA, Iwamoto KS, McBride WH (2012) Maximizing tumor immunity with fractionated radiation. Int J Radiat Oncol Biol Phys 83:1306–1310. doi:10.1016/j.ijrobp.2011.09.049
Kulzer L, Schaft N, Dorrie J, Rubner Y, Frey B, Gaipel U, Fietkau R (2013) Influence of fractionated and hypofractionated radiotherapy on the activation of human dendritic cells. Strahlenther Onkol 189 [Abstractband zur 19. Jahrestagung der Deutschen Gesellschaft für Radioonkologie (DEGRO)]:119
Vanneman M, Dranoff G (2012) Combining immunotherapy and targeted therapies in cancer treatment. Nat Rev Cancer 12:237–251. doi:10.1038/nrc3237
Mantel F, Frey B, Haslinger S et al (2010) Combination of ionising irradiation and hyperthermia activates programmed apoptotic and necrotic cell death pathways in human colorectal carcinoma cells. Strahlenther Onkol 186:587–599. doi:10.1007/s00066-010-2154-x
Werthmoller N, Frey B, Fietkau R, Gaipl US (2013) The influence of standard combinatory tumor treatments and the apoptosis inhibitor zVAD-fmk on B16 melanoma cell death forms and consecutively on macrophages and dendritic cells. Strahlenther Onkol 189 [Abstractband zur 19. Jahrestagung der Deutschen Gesellschaft für Radioonkologie (DEGRO)]:120
Scheithauer HR, Schulz DS, Belka C (2011) Endometrial cancer: reduce to the minimum. A new paradigm for adjuvant treatments? Radiat Oncol 6:164. doi:10.1186/1748-717x-6-164
Sckisel GD, Murphy WJ (2012) Specificity may be overrated in cancer immunotherapy: getting to know the nonspecific side of memory T Cells. Oncoimmunology 1:1208–1210. doi:10.4161/onci.20779
Acknowledgments
This work was supported by the German Research Foundation (DFG -Graduiertenkolleg 1660: Key signals of the adaptive immune response and GA 1507/1-1), the German Federal Ministry of Education and Research (BMBF; m4 Cluster, 16EX1021R and GREWIS, 02NUK017G), and the European Commissions (DoReMi, European Atomic Energy Community’s Seventh Framework Programme (FP7/2007-2011) under Grant Agreement No. 249689).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
This paper is a Focussed Research Review based on a presentation given at the Third International Conference on Cancer Immunotherapy and Immunomonitoring (CITIM 2013), held in Krakow, Poland, 22nd–25th April 2013. It is part of a CII series of Focussed Research Reviews and meeting report.
Rights and permissions
About this article
Cite this article
Frey, B., Rubner, Y., Kulzer, L. et al. Antitumor immune responses induced by ionizing irradiation and further immune stimulation. Cancer Immunol Immunother 63, 29–36 (2014). https://doi.org/10.1007/s00262-013-1474-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-013-1474-y