Abstract
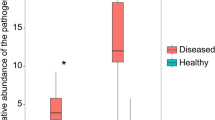
Characterizing the relationship between soil biotic and abiotic properties and plant health status is crucial to understanding the pathogenesis of soil-borne diseases. Here, we compared these properties in the soils of lisianthus with different disease incidence plots and report the cause-effect relationship between soil properties and plant health status using heat treatment coupled with microbiota self/across re-inoculations. The relative importance of soil bacterial and fungal communities in predicting plant health was also analyzed. Results showed that the soils with low and high disease incidences (LDS and HDS) harbored differential microbial communities and physicochemical properties. The LDS soil had relatively low Fusarium oxysporum abundance, electrical conductivity (EC), and NO3−-N content. Soil microbial community was the direct determinant of plant health. The disease-suppressive activity of the microbiome in the LDS soil could be transferred to the HDS soil. Also, the relative importance of the fungal community in predicting plant health status was greater than that of the bacterial community, as reflected by (1) the fungal community could drive more complex networks related to healthy plants and (2) the diversity and core taxa of the fungal community had higher mean predictor importance values for plant health. The relative abundances of core genera Acremonium, Mycothermus, and Chryseolinea were significantly and negatively correlated with the disease incidence and the abundances of pathogens, identifying these genera as potential disease-suppressive agents. Taken together, our results reveal a direct relationship between soil properties and plant health status, in which the fungal community composition is most important for predicting plant health status.
Key points
• Soil with differing pathological groups harbors distinct microbial communities.
• Soil microbial communities directly determine the plant’s health status.
• Fungal community is a better predictor of plant health than the bacterial community.





Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Change history
30 August 2021
A Correction to this paper has been published: https://doi.org/10.1007/s00253-021-11544-z
References
Adam G, Duncan H (2001) Development of a sensitive and rapid method for the measurement of total microbial activity using fluorescein diacetate (FDA) in a range of soils. Soil Biol Biochem 33(8):943–951. https://doi.org/10.1016/S0038-0717(00)00244-3
Alabouvette C, Olivain C, Steinberg C (2006) Biological control of plant diseases: the European situation. Eur J Plant Pathol 114(3):329–341. https://doi.org/10.1007/s10658-005-0233-0
Bastian M, Heymann S, Jacomy M (2009) Gephi: an open source software for exploring and manipulating networks. ICWSM 8:361–362
Berg G (2009) Plant–microbe interactions promoting plant growth and health: perspectives for controlled use of microorganisms in agriculture. Appl Microbiol Biot 84(1):11–18. https://doi.org/10.1007/s00253-009-2092-7
Bolger A, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30(15):2114–2120. https://doi.org/10.1093/bioinformatics/btu170
Breen JP (1994) Acremonium endophyte interactions with enhanced plant resistance to insects. Ann Rev Entomol 39(1):401–423. https://doi.org/10.1146/annurev.en.39.010194.002153
Bremner JM (1960) Determination of nitrogen in soil by the Kjeldahl method. J Agr Sci 55(1):11–33. https://doi.org/10.1017/S0021859600021572
Bremner JM, Jenkinson DS (1960) Determination of organic carbon in soil: I. oxidation by dichromate of organic matter in soil and plant materials. Eur J Soil Sci 11(2):394–402. https://doi.org/10.1111/j.1365-2389.1960.tb01093.x
Bulluck LR, Ristaino JB (2002) Effect of synthetic and organic soil fertility amendments on southern blight, soil microbial communities, and yield of processing tomatoes. Phytopathology 92(2):181–189. https://doi.org/10.1094/PHYTO.2002.92.2.181
Cha JY, Han S, Hong HJ, Cho H, Kim D, Kwon Y, Kwon SK, Crüsemann M, Lee YB, Kim JF, Giaever G, Nislow C, Moor BS, Thomashow LS, Weller DM, Kwak YS (2016) Microbial and biochemical basis of a Fusarium wilt-suppressive soil. ISME J 10(1):119–129. https://doi.org/10.1038/ismej.2015.95
Chaparro JM, Sheflin AM, Manter DK, Vivanco JM (2012) Manipulating the soil microbiome to increase soil health and plant fertility. Biol Fert Soils 48(5):489–499. https://doi.org/10.1007/s00374-012-0691-4
Cole JR, Wang Q, Fish JA, Chai B, Mcgarrell DM, Sun YN, Brown CT, Porrasalfaro A, Kuske CR, Tiedje JM (2014) Ribosomal database project: data and tools for high throughput rRNA analysis. Nucleic Acids Res 42(D1):633–642. https://doi.org/10.1093/nar/gkt1244
Ecker R, Barzilay A, Osherenko E (1994) The genetic relations between length of time to germination and seed dormancy in lisianthus (Eustoma grandiflorum). Euphytica 80(1):125–128. https://doi.org/10.1007/BF00039307
Edgar RC, Haas BJ, Clemente JC, Quince C, Knight R (2011) UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 27(16):2194–2200. https://doi.org/10.1093/bioinformatics/btr381
Fierer N (2017) Embracing the unknown: disentangling the complexities of the soil microbiome. Nat Rev Microbiol 15(10):579–590. https://doi.org/10.1038/nrmicro.2017.87
Fu L, Xiong W, Dini-Andreote F, Wang BB, Tao CY, Ruan YZ, Shen ZZ, Li R, Shen QR (2020) Changes in bulk soil affect the disease-suppressive rhizosphere microbiome against Fusarium wilt disease. Front Agr Sci Eng 7(3):307–316. https://doi.org/10.15302/J-FASE-2020328
Gamliel A, Austerweil M, Kritzman G (2000) Non-chemical approach to soilborne pest management–organic amendments. Crop Prot 19(8–10):847–853. https://doi.org/10.1016/S0261-2194(00)00112-5
Grunewaldt-Stöcker G, von Alten H (2003) Plant health effects of Acremonium root endophytes compared to those of arbuscular mycorrhiza. In: Abe J (ed) The dynamic interface between plants and the earth: the 6th symposium of the international society of root research, 11–15 November 2001, Nagoya, Japan. Springer, Dordrecht, pp 445–454
Herrmann L, Lesueur D (2013) Challenges of formulation and quality of biofertilizers for successful inoculation. Appl Microbiol Biot 97(20):8859–8873. https://doi.org/10.1007/s00253-013-5228-8
Huang XQ, Zhou X, Zhang JB, Cai ZC (2019a) Highly connected taxa located in the microbial network are prevalent in the rhizosphere soil of healthy plant. Biol Fert Soils 55(3):299–312. https://doi.org/10.1007/s00374-019-01350-1
Huang XQ, Zhao J, Zhou X, Zhang JB, Cai ZC (2019b) Differential responses of soil bacterial community and functional diversity to reductive soil disinfestation and chemical soil disinfestation. Geoderma 348:124–134. https://doi.org/10.1016/j.geoderma.2019.04.027
Huang XQ, Liu SZ, Liu X, Zhang SR, Li L, Zhao HT, Zhao J, Zhang JB, Cai ZC (2020) Plant pathological condition is associated with fungal community succession triggered by root exudates in the plant-soil system. Soil Biol Biochem 151:108046. https://doi.org/10.1016/j.soilbio.2020.108046
Huang Y, Xiao X, Huang HY, Jing JQ, Zhao HJ, Wang L, Long XE (2018) Contrasting beneficial and pathogenic microbial communities across consecutive cropping fields of greenhouse strawberry. Appl Microbiol Biot 102(13):5717–5729. https://doi.org/10.1007/s00253-018-9013-6
Janvier C, Villeneuve F, Alabouvette C, Edel-Hermann V, Mateille T, Steinberg C (2007) Soil health through soil disease suppression: which strategy from descriptors to indicators? Soil Biol Biochem 39(1):1–23. https://doi.org/10.1016/j.soilbio.2006.07.001
Kõljalg U, Nilsson RH, Abarenkov K, Tedersoo L, Taylor AFS, Bahram M, Bates ST, Brus TD, Bengtsson-Palme J, Callaghan TM (2013) Towards a unified paradigm for sequence-based identification of fungi. Mol Ecol 22(21):5271–5277. https://doi.org/10.1111/mec.12481
Liaw A, Wiener M, Wiener TC, Liaw JC (2002) Classification and regression by randomforest. R News 2(3):18–22
Li YL, Wang BY, Chang YF, Yang YT, Yao CZ, Huang XQ, Zhang JB, Cai ZC, Zhao J (2019) Reductive soil disinfestation effectively alleviates the replant failure of sanqi ginseng through allelochemical degradation and pathogen suppression. Appl Microbiol Biot 103:3581–3595. https://doi.org/10.1007/s00253-019-09676-4
Li YL, Dai SY, Wang BY, Jiang YT, Ma YY, Pan LL, Wu K, Huang XQ, Zhang JB, Cai ZC, Zhao J (2020) Autotoxic ginsenoside disrupts soil fungal microbiomes by stimulating potentially pathogenic microbes. Appl Environ Microb 86(9):e00130-e220. https://doi.org/10.1128/AEM.00130-20
Liu LL, Huang XQ, Zhao J, Zhang JB, Cai ZC (2019) Characterizing the key agents in a disease-suppressed soil managed by reductive soil disinfestation. Appl Environ Microb 85(7):e02992-e3018. https://doi.org/10.1128/AEM.02992-18
Liu LL, Huang XQ, Zhang JB, Cai ZC, Jiang K, Chang YY (2020) Deciphering the relative importance of soil and plant traits on the development of rhizosphere microbial communities. Soil Biol Biochem 148:107909. https://doi.org/10.1016/j.soilbio.2020.107909
Liu YP, Chen L, Wu GW, Feng HC, Zhang GS, Shen QR, Zhang RF (2017) Identification of root-secreted compounds involved in the communication between cucumber, the beneficial Bacillus amyloliquefaciens, and the soil-borne pathogen Fusarium oxysporum. Mol Plant Microbe in 30(1):53–62. https://doi.org/10.1094/MPMI-07-16-0131-R
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15(12):550–550. https://doi.org/10.1186/s13059-014-0550-8
Magoc T, Salzberg SL (2011) FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics 27(21):2957–2963. https://doi.org/10.1093/bioinformatics/btr507
McMurdie PJ, Holmes S, Michael W (2013) Phyloseq: an R package for reproducible interactive analysis and graphics of microbiome census data. PLoS ONE 8(4):e61217. https://doi.org/10.1371/journal.pone.0061217
Mendes R, Kruijt M, de Bruijn I, Dekkers E, van der Voort M, Schneider JHM, Piceno YM, DeSantis TZ, Andersen GL, Bakker PAHM, Raaijmakers JM (2011) Deciphering the rhizosphere microbiome for disease-suppressive bacteria. Science 332(6033):1097–1100. https://doi.org/10.1126/science.1203980
Meng TZ, Ren GD, Wang GF, Ma Y (2019a) Impacts on soil microbial characteristics and their restorability with different soil disinfestation approaches in intensively cropped greenhouse soils. Appl Microbiol Biot 103(2):6369–6383. https://doi.org/10.1007/s00253-019-09964-z
Meng TZ, Wang QJ, Abbasi PA, Ma Y (2019b) Deciphering differences in the chemical and microbial characteristics of healthy and Fusarium wilt-infected watermelon rhizosphere soils. Appl Microbiol Biot 103(3):1497–1509. https://doi.org/10.1007/s00253-018-9564-6
Nachmias A, Kaufman Z, Livescu L, Tsror L, Meiri A, Caligari PDS (1993) Effects of salinity and its interactions with disease incidence on potatoes grown in hot climates. Phytoparasitica 21(3):245–255. https://doi.org/10.1007/BF02980946
Oksanen J, Blanchet FG, Friednly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner H (2020) Vegan: community ecology package. R package version 2.5–7. http://cran.rproject.org/web/packages/vegan/index.html. Published 28 November 2020
Olanrewaju OS, Ayangbenro AS, Glick BR, Babalola OO (2019) Plant health: feedback effect of root exudates-rhizobiome interactions. Appl Microbiol Biot 103(3):1155–1166. https://doi.org/10.1007/s00253-018-9556-6
Ou YN, Penton CR, Geisen S, Shen ZZ, Sun YF, Lv NN, Wang BB, Ruan YZ, Xiong W, Li R, Shen QR (2019) Deciphering underlying drivers of disease suppressiveness against pathogenic Fusarium oxysporum. Front Microbiol 10:2535. https://doi.org/10.3389/fmicb.2019.02535
Peng HX, Sivasithamparam K, Turner D (1999) Chlamydospore germination and Fusarium wilt of banana plantlets in suppressive and conducive soils are affected by physical and chemical factors. Soil Biol Biochem 31(10):1363–1374. https://doi.org/10.1016/S0038-0717(99)00045-0
Raaijmakers JM, Paulitz TC, Steinberg C, Alabouvette C, Moenneloccoz Y (2009) The rhizosphere: a playground and battlefield for soilborne pathogens and beneficial microorganisms. Plant Soil 321(1):341–361. https://doi.org/10.1007/s11104-008-9568-6
Rohart F, Gautier B, Singh A, Cao KL (2017) mixOmics: an R package for ‘omics feature selection and multiple data integration. PLOS Comput Biol 13(11):e1005752. https://doi.org/10.1371/journal.pcbi.1005752
Romaniuk R, Giuffre L, Costantini A, Nannipieri P (2011) Assessment of soil microbial diversity measurements as indicators of soil functioning in organic and conventional horticulture systems. Ecol Indic 11(5):1345–1353. https://doi.org/10.1016/j.ecolind.2011.02.008
Shi WM, Yao J, Yan F (2009) Vegetable cultivation under greenhouse conditions leads to rapid accumulation of nutrients, acidification and salinity of soils and groundwater contamination in South-Eastern China. Nutr Cycl Agroecosys 83(1):73–84. https://doi.org/10.1007/s10705-008-9201-3
Shi Y, Delgado-Baquerizo M, Li YT, Yang YF, Zhu YG, Peñuelas J, Chu HY (2020) Abundance of kinless hubs within soil microbial networks are associated with high functional potential in agricultural ecosystems. Environ Int 142:105869. https://doi.org/10.1016/j.envint.2020.105869
Steinhauser D, Krall L, Müssig C, Büssis D, Usadel B (2007) Correlation networks. In: Junker BH, Schreiber F (eds) Analysis of biological networks. John Wiley & Sons Inc, New Jersey, pp 305–333
Toju H, Tanabe AS, Sato H (2018a) Network hubs in root-associated fungal metacommunities. Microbiome 6(1):116–140. https://doi.org/10.1186/s40168-018-0497-1
Toju H, Peay KG, Yamamichi M, Narisawa K, Hiruma K, Naito K, Fukuda S, Ushio M, Nakaoka S, Onoda Y, Yoshida K, Schlaeppi K, Bai Y, Sugiura R, Ichihashi Y, Minamisawa Y, Kiers ET (2018b) Core microbiomes for sustainable agroecosystems. Nat Plants 4:247–257. https://doi.org/10.1038/s41477-018-0139-4
Wang TT, Hao YW, Zhu MZ, Yu ST, Ran W, Xue C, Ling N, Shen QR (2019) Characterizing differences in microbial community composition and function between Fusarium wilt diseased and healthy soils under watermelon cultivation. Plant Soil 438(1–2):421–433. https://doi.org/10.1007/s11104-019-04037-6
Watanabe K, Matsui M, Honjo H, Becker JO, Fukui R (2011) Effects of soil pH on rhizoctonia damping-off of sugar beet and disease suppression induced by soil amendment with crop residues. Plant Soil 347:255–268. https://doi.org/10.1007/s11104-011-0843-6
Wei Z, Gu YA, Friman V, Kowalchuk G, Xu YC, Shen QR, Jousset A (2019) Initial soil microbiome composition and functioning predetermine future plant health. Sci Adv 5(9):eaaw0759. https://doi.org/10.1126/sciadv.aaw0759
Xiong W, Li R, Ren Y, Liu C, Zhao QY, Wu HS, Jousset A, Shen QR (2017) Distinct roles for soil fungal and bacterial communities associated with the suppression of vanilla Fusarium wilt disease. Soil Biol Biochem 107:198–207. https://doi.org/10.1016/j.soilbio.2017.01.010
Yeoh YK, Dennis PG, Paungfoo-Lonhienne C, Weber L, Brackin R, Ragan MA, Schmidt S, Hugenholtz P (2017) Evolutionary conservation of a core root microbiome across plant phyla along a tropical soil chronosequence. Nat Commun 8:215. https://doi.org/10.1038/s41467-017-00262-8
Zhao J, Zhou X, Jiang AQ, Fan JZ, Lan T, Zhang JB, Cai ZC (2018) Distinct impacts of reductive soil disinfestation and chemical soil disinfestation on soil fungal communities and memberships. Appl Microbiol Biot 102(17):7623–7634. https://doi.org/10.1007/s00253-018-9107-1
Zhao J, Liu SZ, Zhou X, Xia Q, Liu X, Zhang SR, Zhang JB, Cai ZC, Huang XQ (2020) Reductive soil disinfestation incorporated with organic residue combination significantly improves soil microbial activity and functional diversity than sole residue incorporation. Appl Microbiol Biot 104(7):7573–7588. https://doi.org/10.1007/s00253-020-10778-7
Zheng YK, Miao CP, Chen HH, Huang FF, Xia YM, Chen YW, Zhao LX (2017) Endophytic fungi harbored in Panax notoginseng: diversity and potential as biological control agents against host plant pathogens of root-rot disease. J Ginseng Res 41(3):353–360. https://doi.org/10.1016/j.jgr.2016.07.005
Zhou X, Li CY, Liu LL, Zhao J, Zhang JB, Cai ZC, Huang XQ (2019) Control of Fusarium wilt of lisianthus by reassembling the microbial community in infested soil through reductive soil disinfestation. Microbiol Res 220:1–11. https://doi.org/10.1016/j.micres.2018.12.001
Ziadi N, Tran TS (2007) Mehlich 3-extractable elements. In: Carter MR, Gregorich EG (eds) Soil sampling and methods of analysis, 2nd edn. CRC Press, Boca Raton, pp 81–88
Funding
This work was financially supported by the Key-Area Research and Development Program of Guangdong Province (2020B0202010006), the National Natural Science Foundation of China (Grant No. 42090065, 41771281), the China Postdoctoral Science Foundation (2021M691625), Reserve Talent Project of Young and Middle-aged Academic and Technical Leaders of Yunnan Province (202005AC160043), the Priority Academic Program Development (PAPD) of Jiangsu Higher Education Institutions, and the Science and Technology Research Project of Education Department of Jiangxi Province (No. GJJ190870).
Author information
Authors and Affiliations
Contributions
L.L.L., J.Z., Z.C.C., C.C.D., and X.Q.H. conceived designed research. L.L.L., Y.Y.Y., and H.X.D. conducted experiments. L.L.L. analyzed data and wrote the manuscript. All authors read and approved the manuscript. In addition, we acknowledge TopEdit LLC for the linguistic editing and proofreading during the preparation of this manuscript.
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, L., Yan, Y., Ding, H. et al. The fungal community outperforms the bacterial community in predicting plant health status. Appl Microbiol Biotechnol 105, 6499–6513 (2021). https://doi.org/10.1007/s00253-021-11486-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-021-11486-6