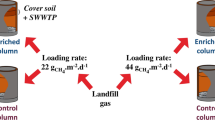
Abstract
Previous studies have demonstrated the effective utility of volcanic pumice soil to mitigate both high and low levels of methane (CH4) emissions through the activity of both γ-proteobacterial (type I) and α-proteobacterial (type II) aerobic methanotrophs. However, the limited availability of volcanic pumice soil necessitates the assessment of other farm soils and potentially suitable, economical and widely available biofilter materials. The potential biofilter materials, viz. farm soil (isolated from a dairy farm effluent pond bank area), pine biochar, garden waste compost and weathered pine bark mulch, were inoculated with a small amount of volcanic pumice soil. Simultaneously, a similar set-up of potential biofilter materials without inoculum was studied to understand the effect of the inoculum on the ability of these materials to oxidise CH4 and their effect on methanotroph growth and activity. These materials were incubated at 25 °C with periodic feeding of CH4, and flasks were aerated with air (O2) to support methanotroph growth and activity by maintaining aerobic conditions. The efficiency of CH4 removal was monitored over 6 months. All materials supported the growth and activity of methanotrophs. However, the efficiency of CH4 removal by all the materials tested fluctuated between no or low removal (0–40 %) and high removal phases (>90 %), indicating biological disturbances rather than physico-chemical changes. Among all the treatments, CH4 removal was consistently high (>80 %) in the inoculated farm soil and inoculated biochar, and these were more resilient to changes in the methanotroph community. The CH4 removal from inoculated farm soil and inoculated biochar was further enhanced (up to 99 %) by the addition of a nutrient solution. Our results showed that (i) farm soil and biochar can be used as a biofilter material by inoculating with an active methanotroph community, (ii) an abundant population of α-proteobacterial methanotrophs is essential for effective and stable CH4 removal and (iii) addition of nutrients enhances the growth and activity of methanotrophs in the biofilter materials. Further studies are underway to assess the feasibility of these materials at small plot and field scales.







Similar content being viewed by others
References
Albanna M, Fernandes L, Warith M (2007) Methane oxidation in landfill cover soil; the combined effects of moisture content, nutrient addition, and cover thickness. J Envrion Eng Sci 6:191–200. doi:10.1139/s06-047
Anette BEA, Kim B (1993) Effect of mineral nutrients on the kinetics of methane utilization by methanotrophs. Biodegradation 4:163–170
Asenjo JA, Suk JS (1986) Microbial conversion of methane into poly-β-hydroxybutyrate (PHB): growth and intracellular product accumulation in a type II methanotroph. J Ferment Technol 64:271–278. doi:10.1016/0385-6380(86)90118-4
Blakemore LC, Searle PL, Daly BK (1987) Methods for chemical analysis of soils, New Zealand Soil Bureau Scientific report, pp. 103
Chen Y, Dumont MG, Cébron A, Murrell JC (2007) Identification of active methanotrophs in a landfill cover soil through detection of expression of 16S rRNA and functional genes. Environ Microbiol 9:2855–2869. doi:10.1111/j.1462-2920.2007.01401.x
Dunbar J, Ticknor LO, Kuske CR (2000) Assessment of microbial diversity in four southwestern United States soils by 16S rRNA gene terminal restriction fragment analysis. Appl Environ Microbiol 66:2943–2950
Dunfield PF, Liesack W, Henckel T, Knowles R, Conrad R (1999) High-affinity methane oxidation by a soil enrichment culture containing a type II methanotroph. Appl Environ Microbiol 65:1009–1014
Gradwell MW (1972) Methods for physical analysis of soils. New Zealand Soil bureau, New Zealand
Graham DW, Chaudhary JA, Hanson RS, Arnold RG (1993) Factors affecting competition between type I and type II methanotrophs in two-organism, continuous-flow reactors. Microbial Ecol 25:1–17. doi:10.1007/bf00182126
Hanson RS, Hanson TE (1996) Methanotrophic bacteria. Microbiol Rev 60:439–471
Haubrichs R, Widmann R (2006) Evaluation of aerated biofilter systems for microbial methane oxidation of poor landfill gas. Waste Manag 26:408–416. doi:10.1016/j.wasman.2005.11.008
Henneberger R, Lüke C, Mosberger L, Schroth MH (2012) Structure and function of methanotrophic communities in a landfill-cover soil. FEMS Microbiol Ecol 81:52–65. doi:10.1111/j.1574-6941.2011.01278.x
Ho A, Kerckhof FM, Luke C, Reim A, Krause S, Boon N, Bodelier PLE (2013) Conceptualizing functional traits and ecological characteristics of methane-oxidizing bacteria as life strategies. Environ Microbiol 5:335–345. doi:10.1111/j.1758-2229.2012.00370.x
Jiang H, Chen Y, Jiang P, Zhang C, Smith TJ, Murrell JC, Xing X-H (2010) Methanotrophs: multifunctional bacteria with promising applications in environmental bioengineering. Biochem Eng J 49:277–288
Kolb S, Knief C, Stubner S, Conrad R (2003) Quantitative detection of methanotrophs in soil by novel pmoA-targeted real-time PCR assays. Appl Environ Microbiol 69:2423–2439. doi:10.1128/AEM.69.5.2423–2429.2003
Kumar, S., Stecher, G., and Tamura, K. (2015). MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Mol Biol Evol (submitted)
Kuo YC, Cheng SF, Liu PWG, Chiou HY, Kao CM (2012) Application of enhanced bioremediation for TCE contaminated groundwater: a pilot-scale study. Inst Chem E 41:364–371
Lee SW, Keeney DR, Lim D-H, Dispirito AA, Semrau JD (2006) Mixed pollutant degradation by methylosinus trichosporium OB3b expressing either soluble or particulate methane monooxygenase: can the tortoise beat the hare? Appl Environ Microbiol 72:7503–7509
Lee C, Lee S, Shin SG, Hwang S (2008) Real-time PCR determination of rRNA gene copy number: absolute and relative quantification assays with Escherichia coli. Appl Microbiol Biot:371–376
Menard C, Ramirez AA, Nikiema J, Heitz M (2012) Biofiltration of methane and trace gases from landfills: a review. Environ Rev 20:40–53. doi:10.1139/A11-022
Mohanty SR, Bodelier PLE, Floris V, Conrad R (2006) Differential effects of nitrogenous fertilizers on methane-consuming microbes in rice field and forest soils. Appl Environ Microbiol 72:1346–1354. doi:10.1128/AEM.72.2.1346-1354.2006
Nikiema J, Brzezinski R, Heitz M (2010) Influence of phosphorus, potassium, and copper on methane biofiltration performance. Can J Civil Eng 37:335–345. doi:10.1139/L09-145
Noll M, Frenzel P (2008). Selective stimulation of type I methanotrophs in a rice paddy soil by urea fertilization revealed by RNA-based stable isotope probing FEMS Microbiol Ecol 65:125–132
Pratt C, Walcroft AS, Tate KR, Ross DJ, Roy R, Reid MH, Veiga PW (2012a) In vitro methane removal by volcanic pumice soil biofilter columns over one year. J Environ Qual 41:80–87
Pratt C, Walcroft AS, Tate KR, Ross DJ, Roy R, Reid MH, Veiga PW (2012b) Biofiltration of methane emissions from a dairy farm effluent pond. Agric Ecosyst Environ 152:33–39
Pratt C, Deslippe J, Tate KR (2012c) Testing a biofilter cover design to mitigate dairy effluent pond methane emissions. Environ Sci Technol 47:526–532. doi:10.1021/es303225h
Scheutz C, Kjeldsen P, Bogner JE, De Visscher A, Gebert J, Hilger HA, Huber-Humer M, Spokas K (2009) Microbial methane oxidation processes and technologies for mitigation of landfill gas emissions. Waste Manag Res 27:409. doi:10.1177/0734242X09339325
Semrau JD, DiSpirito AA, Yoon S (2010) Methanotrophs and copper. FEMS Microbiol Rev 34:496–531
Syed R, Saggar S, Tate K, Rehm BHA (2016) Does acidification of a soil biofilter compromise its methane-oxidising capacity? Biol Fertil Soils 52:573–583. doi:10.1007/s00374-016-1103-y
Tate KR (2015) Soil methane oxidation and land-use change—from process to mitigation. Soil Biol Biochem 80:260–272
Tate KR, Ross DJ, Saggar S, Hedley CB, Dando J, Singh BK, Lambie SM (2007) Methane uptake in soils from Pinus radiata plantations, a reverting shrubland and adjacent pastures: effects of land-use change, and soil texture, water and mineral nitrogen. Soil Biol Biochem 39:1437–1449
Tate KR, Walcroft AS, Pratt C (2012) Varying atmospheric methane concentrations affect soil methane oxidation rates and methanotroph populations in pasture, an adjacent pine forest, and a landfill. Soil Biol Biochem 52:75–81
Acknowledgments
The authors thank Landcare Research for the Des Ross Memorial Scholarship to support the first author and for research facilities for this study, Peter Berben and Thilak Palmada for technical help, Neha Jha for the internal review, Anne Austin for the editing, Massey Genome Services for the sequencing and Massey University for providing access to the Roche light cycler machine for qPCR analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study was funded by Landcare Research, New Zealand (Des Ross Memorial Scholarship 2013–2016). Author(s) declares that they have no conflict of interest. This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Syed, R., Saggar, S., Tate, K. et al. Assessment of farm soil, biochar, compost and weathered pine mulch to mitigate methane emissions. Appl Microbiol Biotechnol 100, 9365–9379 (2016). https://doi.org/10.1007/s00253-016-7794-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-016-7794-z