Abstract
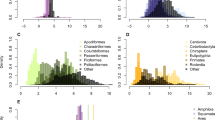
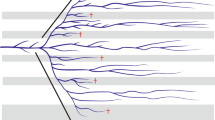
During the past 50 years, the molecular clock has become one of the main tools for providing a time scale for the history of life. In the era of robust molecular evolutionary analysis, clock calibration is still one of the most basic steps needing attention. When fossil records are limited, well-dated geological events are the main resource for calibration. However, biogeographic calibrations have often been used in a simplistic manner, for example assuming simultaneous vicariant divergence of multiple sister lineages. Here, we propose a novel iterative calibration approach to define the most appropriate calibration date by seeking congruence between the dates assigned to multiple allopatric divergences and the geological history. Exploring patterns of molecular divergence in 16 trans-Bering sister clades of echinoderms, we demonstrate that the iterative calibration is predominantly advantageous when using complex geological or climatological events—such as the opening/reclosure of the Bering Strait—providing a powerful tool for clock dating that can be applied to other biogeographic calibration systems and further taxa. Using Bayesian analysis, we observed that evolutionary rate variability in the COI-5P gene is generally distributed in a clock-like fashion for Northern echinoderms. The results reveal a large range of genetic divergences, consistent with multiple pulses of trans-Bering migrations. A resulting rate of 2.8% pairwise Kimura-2-parameter sequence divergence per million years is suggested for the COI-5P gene in Northern echinoderms. Given that molecular rates may vary across latitudes and taxa, this study provides a new context for dating the evolutionary history of Arctic marine life.







Similar content being viewed by others
References
Addison JA, Pogson GH (2009) Multiple gene genealogies reveal asymmetrical hybridization and introgression among strongylocentrotid sea urchins. Mol Ecol 18:1239–1251. https://doi.org/10.1111/j.1365-294X.2009.04094.x
Arndt A, Marquez C, Lambert P, Smith MJ (1996) Molecular phylogeny of Eastern Pacific sea cucumbers (Echinodermata: Holothuroidea) based on mitochondrial DNA sequence. Mol Phylogenet Evol 6:425–437. https://doi.org/10.1006/mpev.1996.0091
Athey T (2013) Assessing errors in DNA barcode sequence records. Master thesis, University of Guelph
Bacon CD, Silvestro D, Jaramillo C et al (2015) Biological evidence supports an early and complex emergence of the Isthmus of Panama. Proc Natl Acad Sci 112:E3153–E3153. https://doi.org/10.1073/pnas.1509107112
Bargelloni L, Ritchie PA, Patarnello T et al (1994) Molecular evolution at subzero temperatures: mitochondrial and nuclear phylogenies of fishes from Antarctica (suborder Notothenioidei), and the evolution of antifreeze glycopeptides. Mol Biol Evol 11:854–863
Bastrop R, Blank M (2006) Multiple invasions - a polychaete genus enters the Baltic Sea. Biol Invasions 8:1195–1200. https://doi.org/10.1007/s10530-005-6186-6
Bleidorn C, Kruse I, Albrecht S, Bartolomaeus T (2006) Mitochondrial sequence data expose the putative cosmopolitan polychaete Scoloplos armiger (Annelida, Orbiniidae) as a species complex. BMC Evol Biol 6:47. https://doi.org/10.1186/1471-2148-6-47
Bleiweiss R (1998a) Relative-rate tests and biological causes of molecular evolution in hummingbirds. Mol Biol Evol 15:481–491
Bleiweiss R (1998b) Slow rate of molecular evolution in high-elevation hummingbirds. Proc Natl Acad Sci USA 95:612–616. https://doi.org/10.1073/pnas.95.2.612
Boissin E, Stöhr S, Chenuil A (2011) Did vicariance and adaptation drive cryptic speciation and evolution of brooding in Ophioderma longicauda (Echinodermata: Ophiuroidea), a common Atlanto-Mediterranean ophiuroid? Mol Ecol 2:4737–4755. https://doi.org/10.1111/j.1365-294X.2011.05309.x
Bribiesca-Contreras G, Solís-Marín FA, Laguarda-Figueras A, Zaldívar-Riverón A (2013) Identification of echinoderms (Echinodermata) from an anchialine cave in Cozumel Island, Mexico, using DNA barcodes. Mol Ecol Resour 13:1137–1145. https://doi.org/10.1111/1755-0998.12098
Briggs JC (1970) A faunal history of the North Atlantic Ocean. Syst Zool 19:19–34. https://doi.org/10.2307/2412025
Bromham L (2002) Molecular clocks in reptiles: life history influences rate of molecular evolution. Mol Biol Evol 19:302–309. https://doi.org/10.1093/oxfordjournals.molbev.a004083
Carr CM (2010) The Polychaeta of Canada: exploring diversity and distribution patterns using DNA barcodes. Master thesis, University of Guelph
Carr CM, Hardy SM, Brown TM et al (2011) A tri-oceanic perspective: DNA barcoding reveals geographic structure and cryptic diversity in Canadian polychaetes. PLoS ONE 6:e22232. https://doi.org/10.1371/journal.pone.0022232
Coates AG, Obando JA (1996) The geologic evolution of the Central American Isthmus. In: Jackson JBC, Budd AF, Coates AG (eds) Evolution and environment in tropical America. The University of Chicago Press, Chicago, pp 21–56
Collins LS, Coates AG, Berggren WA et al (1996a) The late Miocene Panama isthmian strait. Geology 24:687–690
Collins TM, Frazer K, Brown WM (1996b) Evolutionary history of Northern Hemisphere Nucella (Gastropoda, Muricidae): molecular, morphological, ecological, and paleontological evidence. Evolution 50:2287–2304
Coppard SE, Zigler KS, Lessios H (2013) Phylogeography of the sand dollar genus Mellita: cryptic speciation along the coasts of the Americas. Mol Phylogenet Evol 69:1033–1042. https://doi.org/10.1016/j.ympev.2013.05.028
Dodson JJ, Tremblay S, Colombani F et al (2007) Trans-Arctic dispersals and the evolution of a circumpolar marine fish species complex, the capelin (Mallotus villosus). Mol Ecol 16:5030–5043. https://doi.org/10.1111/j.1365-294X.2007.03559.x
Drummond AJ, Bouckaert RR (2015) Bayesian evolutionary analysis with BEAST. Cambridge University Press, Cambridge
Drummond AJ, Ho SYW, Phillips MJ, Rambaut A (2006) Relaxed phylogenetics and dating with confidence. PLoS Biol 4:699–710. https://doi.org/10.1371/journal.pbio.0040088
Drummond AJ, Suchard MA, Xie D, Rambaut A (2012) Bayesian phylogenetics with BEAUti and the BEAST 1.7. Mol Biol Evol 29:1969–1973. https://doi.org/10.1093/molbev/mss075
Dunton K (1992) Arctic biogeography: the paradox of the marine benthic fauna and flora. Trends Ecol Evol 7:183–189. https://doi.org/10.1016/0169-5347(92)90070-R
Duque-Caro H (1990) Neogene stratigraphy, paleoceanography and paleobiogeography in northwest South America and the evolution of the Panama seaway. Palaeogeogr Palaeoclimatol Palaeoecol 77:203–234. https://doi.org/10.1016/0031-0182(90)90178-A
Durham J, MacNeil F (1967) Cenozoic migrations of marine invertebrates through the Bering Strait region. In: Hopkins D (ed) The Bering land bridge. Stanford University Press, Stanford, pp 326–349
Einarsson T, Hopkins DM, Doell RR (1967) The stratigraphy of Tjornes, North Iceland and the history of the Bering land bridge. In: Hopkins D (ed) The Bering land bridge. Stanford University Press, Stanford California, pp 312–325
Feuda R, Smith AB (2015) Phylogenetic signal dissection identifies the root of starfishes. PLoS ONE 10:e0123331. https://doi.org/10.1371/journal.pone.0123331
Fleischer RC, McIntosh CE, Tarr CL (1998) Evolution on a volcanic conveyor belt: using phylogeographic reconstructions and K-Ar-based ages of the Hawaiian Islands to estimate molecular evolutionary rates. Mol Ecol 7:533–545. https://doi.org/10.1046/j.1365-294x.1998.00364.x
Foltz DW, Nguyen AT, Kiger JR, Mah CL (2008) Pleistocene speciation of sister taxa in a North Pacific clade of brooding sea stars (Leptasterias). Mar Biol 154:593–602. https://doi.org/10.1007/s00227-008-0952-9
Gernhard T (2008) The conditioned reconstructed process. J Theor Biol 253:769–778. https://doi.org/10.1016/j.jtbi.2008.04.005
Gillooly JF, Allen AP, West GB, Brown JH (2005) The rate of DNA evolution: effects of body size and temperature on the molecular clock. Proc Natl Acad Sci USA 102:140–145. https://doi.org/10.1073/pnas.0407735101
Gladenkov AY, Gladenkov YB (2004) Onset of connections between the Pacific and Arctic Oceans through the Bering Strait in the Neogene. Stratigr Geol Correl 12:175–187
Gladenkov A, Oleinik A, Marincovich LJ, Barinov K (2002) A refined age for the earlier opening of Bering Strait. Paleogeogr Paleclimatol Paleoecol 183:321–328
Gregory TR (2008) Understanding evolutionary trees. Evol Educ Outreach 1:121–137. https://doi.org/10.1007/s12052-008-0035-x
Haq B, Hardenbol J, Vail P (1987) Chronology of fluctuating sea levels since the Triassic. Science 235:1156–1167. https://doi.org/10.1126/science.235.4793.1156
Hardy SM, Carr CM, Hardman M et al (2011) Biodiversity and phylogeography of Arctic marine fauna: insights from molecular tools. Mar Biodivers 41:195–210. https://doi.org/10.1007/s12526-010-0056-x
Harper FM, Hart MW (2007) Morphological and phylogenetic evidence for hybridization and introgression in a sea star secondary contact zone. Invertebr Biol 126:373–384. https://doi.org/10.1111/j.1744-7410.2007.00107.x
Harper FM, Addison JA, Hart MW (2007) Introgression versus immigration in hybridizing high-dispersal echinoderms. Evolution 61:2410–2418. https://doi.org/10.1111/j.1558-5646.2007.00200.x
Harris SA (2005) Thermal history of the Arctic Ocean environs adjacent to North America during the last 3.5 Ma and a possible mechanism for the cause of the cold events (major glaciations and permafrost events). Prog Phys Geogr 29:218–238. https://doi.org/10.1191/0309133305pp444ra
Hart MW, Byrne M, Smith MJ (1997) Molecular phylogenetic analysis of life-history evolution in asterinid starfish. Evolution 51:1848–1861
Haug GH, Sigman DM, Tiedemann R et al (1999) Onset of permanent startification in the subarctic Pacific Ocean. Nature 401:779–782
Hebert PDN, Cywinska A, Ball SL, DeWaard JR (2003a) Biological identifications through DNA barcodes. Proc R Soc Lond B Biol Sci 270:313–321. https://doi.org/10.1098/rspb.2002.2218
Hebert PDN, Ratnasingham S, DeWaard JR (2003b) Barcoding animal life: cytochrome c oxidase subunit 1 divergences among closely related species. Proc R Soc Lond B Biol Sci 270 Suppl:S96–S99. https://doi.org/10.1098/rsbl.2003.0025
Held C (2001) No evidence for slow-down of molecular substitution rates at subzero temperatures in Antarctic serolid isopods (Crustacea, Isopoda, Serolidae). Polar Biol 24:497–501. https://doi.org/10.1007/s003000100245
Herman Y, Hopkins DM (1980) Arctic oceanic climate in Late Cenozoic time. Science 209:557–562
Ho SYW, Duchêne S (2014) Molecular-clock methods for estimating evolutionary rates and timescales. Mol Ecol 23:5947–5965. https://doi.org/10.1111/mec.12953
Ho SYW, Larson G (2006) Molecular clocks: when times are a-changin’. Trends Genet 22:79–83. https://doi.org/10.1016/j.tig.2005.11.006
Ho SYW, Phillips MJ (2009) Accounting for calibration uncertainty in phylogenetic estimation of evolutionary divergence times. Syst Biol 58:367–380. https://doi.org/10.1093/sysbio/syp035
Ho SYW, Phillips MJ, Cooper A, Drummond AJ (2005) Time dependency of molecular rate estimates and systematic overestimation of recent divergence times. Mol Biol Evol 22:1561–1568. https://doi.org/10.1093/molbev/msi145
Ho SYW, Lanfear R, Bromham L et al (2011) Time-dependent rates of molecular evolution. Mol Ecol 20:3087–3101. https://doi.org/10.1111/j.1365-294X.2011.05178.x
Ho SYW, Duchêne S, Molak M, Shapiro B (2015) Time-dependent estimates of molecular evolutionary rates: evidence and causes. Mol Ecol 24:6007–6012. https://doi.org/10.1111/mec.13450
Hoareau TB, Boissin E, Paulay G, Bruggemann JH (2013) The Southwestern Indian Ocean as a potential marine evolutionary hotspot: perspectives from comparative phylogeography of reef brittle-stars. J Biogeogr 40:2167–2179. https://doi.org/10.1111/jbi.12155
Hopkins D (1967) Quaternary marine trangressions in Alaska. In: Hopkins DM (ed) The Bering land bridge. Stanford University Press, Stanford, pp 47–90
Horikawa K, Martin EE, Basak C et al (2015) Pliocene cooling enhanced by flow of low-salinity Bering Sea water to the Arctic Ocean. Nat Commun 6:7587. https://doi.org/10.1038/ncomms8587
Hu A, Meehl GA, Han W (2007) Role of the Bering Strait in the thermohaline circulation and abrupt climate change. Geophys Res Lett 34:1–6. https://doi.org/10.1029/2006GL028906
Hurt C, Anker A, Knowlton N (2009) A multilocus test of simultaneous divergence across the Isthmus of Panama using snapping shrimp in the genus Alpheus. Evolution 63:514–530. https://doi.org/10.1111/j.1558-5646.2008.00566.x
Jordan DS (1908) The law of geminate species. Am Nat 42:73–80
Kamarudin KR, Hashim R, Usup G (2010) Phylogeny of sea cucumber (Echinodermata: Holothuroidea) as inferred from 16 s mitochondrial rRNA gene sequences. Sains Malays 39:209–218
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120. https://doi.org/10.1007/BF01731581
Knowlton N, Weigt LA (1998) New dates and new rates for divergence across the Isthmus of Panama. Proc R Soc Lond B Biol Sci 265:2257–2263
Kober KM, Bernardi G (2013) Phylogenomics of strongylocentrotid sea urchins. BMC Evol Biol 13:88. https://doi.org/10.1186/1471-2148-13-88
Landis MJ (2016) Biogeographic dating of speciation times using paleogeographically informed processes. Syst Biol 66:128–144. https://doi.org/10.1093/sysbio/syw040
Layton KKS, Corstorphine EA, Hebert PDN (2016) Exploring Canadian echinoderm diversity through DNA barcodes. PLoS ONE 1–16. https://doi.org/10.1371/journal.pone.0166118
Lee YH (2003) Molecular phylogenies and divergence times of sea urchin species of Strongylocentrotidae, Echinoida. Mol Biol Evol 20:1211–1221. https://doi.org/10.1093/molbev/msg125
Leigh EG, O’Dea A, Vermeij GJ (2014) Historical biogeography of the Isthmus of Panama. Biol Rev 89:148–172. https://doi.org/10.1111/brv.12048
Lessios H (1979) Use of Panamanian sea urchins to test the molecular clock. Nature 280:599–601
Lessios H (2008) The great American schism: divergence of marine organisms after the rise of the central American Isthmus. Annu Rev Ecol Evol Syst 39:63–91. https://doi.org/10.1146/annurev.ecolsys.38.091206.095815
Lessios H, Pearse J (1996) Hybridization and introgression between Indo-Pacific species of Diadema. Mar Biol 126:715–723. https://doi.org/10.1007/BF00351338
Lessios H, Kessing BD, Pearse JS (2001) Population structure and speciation in tropical seas: global phylogeography of the sea urchin Diadema. Evolution 55:955–975. https://doi.org/10.1111/j.0014-3820.2001.tb00613.x
Lessios H, Lockhart S, Collin R et al (2012) Phylogeography and bindin evolution in Arbacia, a sea urchin genus with an unusual distribution. Mol Ecol 21:130–144. https://doi.org/10.1111/j.1365-294X.2011.05303.x
Littlewood DTJ, Smith AB, Clough KA, Emson RH (1997) The interrelationships of the echinoderm classes: morphological and molecular evidence. Biol J Linn Soc 61:409–438. https://doi.org/10.1111/j.1095-8312.1997.tb01799.x
Luttikhuizen PC, Drent J, Baker AJ (2003) Disjunct distribution of highly diverged mitochondrial lineage clade and population subdivision in a marine bivalve with pelagic larval dispersal. Mol Ecol 12:2215–2229. https://doi.org/10.1046/j.1365-294X.2003.01872.x
Mah CL, Blake DB (2012) Global diversity and phylogeny of the Asteroidea (Echinodermata). PLoS ONE 7:e35644. https://doi.org/10.1371/journal.pone.0035644
Mah C, Foltz D (2011) Molecular phylogeny of the Forcipulatacea (Asteroidea: Echinodermata): systematics and biogeography. Zool J Linn Soc 162:646–660. https://doi.org/10.1111/j.1096-3642.2010.00688.x
Marincovich LJ (1993) Danian mollusks from the Prince Creek formation Northern Alaska and implications for Arctic Ocean paleogeography. Paleontol Soc Mem 35:1–35. https://doi.org/10.2307/1315585
Marincovich LJ (2000) Central American paleogeography controlled Pliocene Arctic Ocean molluscan migrations. Geology 28:551–554
Marincovich LJ, Gladenkov A (1999) Evidence for an early opening of the Bering Strait. Nature 397:149–151. https://doi.org/10.1038/16446
Marincovich L, Gladenkov AY (2001) New evidence for the age of Bering Strait. Quat Sci Rev 20:329–335. https://doi.org/10.1016/S0277-3791(00)00113-X
Marko PB, Moran AL (2002) Correlated evolutionary divergence of egg size and a mitochondrial protein across the Isthmus of Panama. Evolution 56:1303–1309
Martin AP, Palumbi SR (1993) Body size, metabolic rate, generation time, and the molecular clock. Proc Natl Acad Sci 90:4087–4091. https://doi.org/10.1073/pnas.90.9.4087
Martin AP, Naylor GJ, Palumbi SR (1992) Rates of mitochondrial DNA evolution in sharks are slow compared with mammals. Nature 357:153–155. https://doi.org/10.1038/357153a0
Maslin MA, Haug G, Sarnthein M, Tiedemann R (1996) The progressive intensification of Northern Hemisphere glaciation as seen from the North Pacific. Geol Rundschau 85:452–465. https://doi.org/10.1007/BF02369002
Matthiessen J, Knies J, Vogt C, Stein R (2009) Pliocene palaeoceanography of the Arctic Ocean and subarctic seas. Philos Trans R Soc Lond A 367:21–48. https://doi.org/10.1098/rsta.2008.0203
McCartney M, Keller G, Lessios H (2000) Dispersal barriers in tropical oceans and speciation in Atlantic and eastern Pacific sea urchins of the genus Echinometra. Mol Ecol 9:1391–1400. https://doi.org/10.1046/j.1365-294X.2000.01022.x
Mendes FK, Hahn MW (2016) Gene tree discordance causes apparent substitution rate variation. Syst Biol 65:711–721. https://doi.org/10.1093/sysbio/syw018
Miura O, Torchin ME, Bermingham E (2010) Molecular phylogenetics reveals differential divergence of coastal snails separated by the Isthmus of Panama. Mol Phylogenet Evol 56:40–48. https://doi.org/10.1016/j.ympev.2010.04.012
Mooers AO, Harvey PH (1994) Metabolic rate, generation time, and the rate of molecular evolution in birds. Mol Phylogenet Evol 3:344–350. https://doi.org/10.1006/mpev.1994.1040
O’Dea A, Lessios HA, Coates AG et al (2016) Formation of the Isthmus of Panama. Sci Adv 2:1–12. https://doi.org/10.1126/sciadv.1600883
Ogg J, Ogg G, Gradstein F (2016) A concise geological time scale. Elsevier, Amsterdam
Palumbi SR, Kessing BD (1991) Population biology of the Trans-Arctic exchange: MtDNA sequence similarity between Pacific and Atlantic sea urchins. Evolution 45:1790–1805
Perseke M, Bernhard D, Fritzsch G et al (2010) Mitochondrial genome evolution in Ophiuroidea, Echinoidea, and Holothuroidea: insights in phylogenetic relationships of Echinodermata. Mol Phylogenet Evol 56:201–211. https://doi.org/10.1016/j.ympev.2010.01.035
Polyakova YI (2001) Late Cenozoic evolution of Northern Eurasian marginal seas based on the diatom record. Polarforschung 69:211–220
Radulovici AE, Archambault P, Dufresne F (2010) DNA barcodes for marine biodiversity. Moving fast forward? Diversity 2:450–472. https://doi.org/10.3390/d2040450
Rambaut A, Suchard MA, Xie D, Drummond AJ (2014) Tracer v1.6. Available from http://beast.bio.ed.ac.uk/Tracer
Ratnasingham S, Hebert PDN (2007) BOLD: the barcode of life data system: barcoding. Mol Ecol Notes 7:355–364. https://doi.org/10.1111/j.1471-8286.2007.01678.x
Ratnasingham S, Hebert PDN (2013) A DNA-based registry for all animal species: the Barcode Index Number (BIN) system. PLoS One 8:e66213. https://doi.org/10.1371/journal.pone.0066213
Robinson M, Gouy M, Gautier C, Mouchiroud D (1998) Sensitivity of the relative-rate test to taxonomic sampling. Mol Biol Evol 15:1091–1098
Rohde K (1992) Latitudinal gradients in species diversity: the search for the primary cause. Oikos 65:514–527. https://doi.org/10.2307/3545569
Saitou N, Nei M (1987) The neighbour-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evo 4:406–425
Schrader H-J, Bjorklund K, Manum S et al (1976) Cenozoic biostratigraphy, physical stratigraphy and paleooceanography in the Norwegian-Greenland Sea, DSDP Leg 38 paleontological synthesis. In: Talwan M, Al E (eds) Initial reports deep sea drilling project. U.S. Goverment Printing Office, Washington DC, pp 1197–1211
Shackleton NJ, Opdyke ND (1977) Oxygen isotope and palaeomagnetic evidence for early Northern Hemisphere glaciation. Nature 270:216–219. https://doi.org/10.1038/270216a0
Shaffer G, Bendtsen J (1994) Role of the Bering Strait in controlling North Atlantic ocean circulation and climate. Nature 367:354–357. https://doi.org/10.1038/367354a0
Tajima F (1993) Simple methods for testing the molecular evolutionary clock hypothesis. Genetics 135:599–607
Tamura K, Stecher G, Peterson D et al (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729. https://doi.org/10.1093/molbev/mst197
Telford MJ, Lowe CJ, Cameron CB et al (2014) Phylogenomic analysis of echinoderm class relationships supports Asterozoa. Proc R Soc B Biol Sci 281:20140479. https://doi.org/10.1098/rspb.2014.0479
Vermeij GJ (1989) Geographical restriction as a guide to the causes of extinction: the case of the cold Northern Oceans during the Neogene. Paleobiology 15:335–356
Vermeij GJ (1991) Anatomy of an invasion: the trans-Arctic interchange. Paleobiology 17:281–307
Vogler C, Benzie J, Lessios H et al (2008) A threat to coral reefs multiplied? Four species of crown-of-thorns starfish. Biol Lett 4:696–699. https://doi.org/10.1098/rsbl.2008.0454
Ward RD, Holmes BH, O’Hara TD (2008) DNA barcoding discriminates echinoderm species. Mol Ecol Resour 8:1202–1211. https://doi.org/10.1111/j.1755-0998.2008.02332.x
Wares JP, Cunningham CW (2001) Phylogeography and historical ecology of the North Atlantic intertidal. Evolution 55:2455–2469. https://doi.org/10.1111/j.0014-3820.2001.tb00760.x
Weir JT, Schluter D (2008) Calibrating the avian molecular clock. Mol Ecol 17:2321–2328. https://doi.org/10.1111/j.1365-294X.2008.03742.x
Williams ST, Reid DG (2004) Speciation and diversity on tropical rocky shores: a global phylogeny of snails of the genus Echinolittorina. Evolution 58:2227–2251. https://doi.org/10.1111/j.0014-3820.2004.tb01600.x
Wright S, Keeling J, Gillman L (2006) The road from Santa Rosalia: a faster tempo of evolution in tropical climates. Proc Natl Acad Sci USA 103:7718–7722. https://doi.org/10.1073/pnas.0510383103
Yang Z, Rannala B (2006) Bayesian estimation of species divergence times under a molecular clock using multiple fossil calibrations with soft bounds. Mol Biol Evol 23:212–226. https://doi.org/10.1093/molbev/msj024
Zuckerkandl E, Pauling L (1965) Evolutionary divergence and convergence in proteins. In: Bryson V, Vogel H (eds) Evolving genes and proteins. Academic Press, New York, pp 97–166
Acknowledgements
This work was supported by a graduate scholarship from Consejo Nacional de Ciencia y Tecnología (315757 to TLQ) and by the Natural Sciences and Engineering Research Council of Canada (Discovery Grant 2010-386591 and 2016-06199 to SJA). We also acknowledge the Ontario Ministry of Research Innovation for providing funding to Paul Hebert for the development of BOLD, which was the main resource of data for this project. Special thanks to Teresa J. Crease, Elizabeth G. Boulding, Jonathan D.S. Witt, and Mari Kekkonen for discussion and their valuable feedback on earlier versions of this manuscript. We sincerely thank two anonymous reviewers for their helpful and constructive comments. Finally, we want to acknowledge all the researchers who have made public COI barcode sequences from echinoderms.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Loeza-Quintana, T., Adamowicz, S.J. Iterative Calibration: A Novel Approach for Calibrating the Molecular Clock Using Complex Geological Events. J Mol Evol 86, 118–137 (2018). https://doi.org/10.1007/s00239-018-9831-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00239-018-9831-2