Abstract
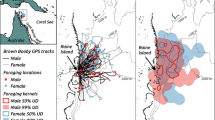
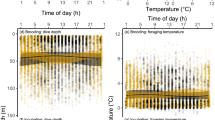
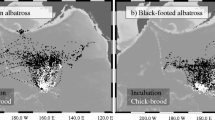
Sexual segregation in foraging habitat occurs in many marine predators and is usually attributed to competitive exclusion, different parental roles of each sex or niche specialisation associated with sexual size dimorphism. However, relatively few studies have attempted to understand the patterns and underlying drivers of local-scale sexual segregation in marine predators. We studied habitat use, diet and feeding ecology of female and male wandering albatrosses Diomedea exulans, fitted with GPS and stomach-temperature loggers during the chick-rearing period (austral winter) at South Georgia in 2009. During this period, when oceanographic conditions were anomalous and prey availability was low in waters near the breeding colony, the tracked wandering albatrosses showed high consistency in their foraging areas at a large spatial scale, and both males and females targeted sub-Antarctic and subtropical waters. Despite consistency in large-scale habitat use, males and females showed different foraging behaviours in response to oceanographic conditions at a smaller scale. Males appeared to be more opportunistic, scavenging for offal or non-target fish discarded by fishing vessels in less productive, oceanic waters. They exhibited sinuous movements, feeding mostly on large prey and consuming similar amounts of food during the outbound and return parts of the foraging trip. In contrast, females targeted natural productivity hotspots, and fed on a wide variety of fish and cephalopods. They commuted directly to these areas; most prey were ingested on the outbound part of the trip, and they often started their return after ingesting large prey at the farthest point from the colony. Together, these results indicate that sexual segregation in core foraging areas of wandering albatrosses is driven by sex-specific habitat selection due to the low availability of prey in local Antarctic waters. This segregation results in different feeding behaviour at local scales which may be explained by differing breeding roles and degree of parental investment by each sex, with females investing more than males in reproduction. Further investigations are necessary to confirm the existence of this pattern through time under contrasting environmental conditions and to identify the drivers responsible for local-scale sexual segregation in wandering albatrosses.



Similar content being viewed by others
References
Åkesson S, Weimerskirch H (2014) Evidence for sex-segregated ocean distributions of first-winter wandering albatrosses at Crozet Islands. PLoS One 9:e86779. https://doi.org/10.1371/journal.pone.0086779
Araújo MB, Pearson RG, Thuiller W, Erhard M (2005) Validation of species-climate impact models under climate change. Glob Change Biol 11:1504–1513. https://doi.org/10.1111/j.1365-2486.2005.01000.x
Barbet-Massin M, Jiguet F, Albert CH, Thuiller W (2012) Selecting pseudo-absences for species distribution models: how, where and how many? Methods Ecol Evol 3:327–338. https://doi.org/10.1111/j.2041-210X.2011.00172.x
Broennimann O, Fitzpatrick MC, Pearman PB et al (2012) Measuring ecological niche overlap from occurrence and spatial environmental data. Glob Ecol Biogeogr 21:481–497. https://doi.org/10.1111/j.1466-8238.2011.00698.x
Calenge C (2006) The package “adehabitat” for the R software: a tool for the analysis of space and habitat use by animals. Ecol Model 197:516–519. https://doi.org/10.1016/j.ecolmodel.2006.03.017
Catry P, Phillips RA, Phalan B et al (2004) Foraging strategies of grey-headed albatrosses Thalassarche chrysostoma: integration of movements, activity and feeding events. Mar Ecol Prog Ser 280:261–273. https://doi.org/10.3354/meps280261
Cherel Y, Xavier JC, de Grissac S et al (2017) Feeding ecology, isotopic niche, and ingestion of fishery-related items of the wandering albatross Diomedea exulans at Kerguelen and Crozet Islands. Mar Ecol Prog Ser 565:197–215. https://doi.org/10.3354/meps11994
Cleasby IR, Wakefield ED, Bodey TW et al (2015) Sexual segregation in a wide-ranging marine predator is a consequence of habitat selection. Mar Ecol Prog Ser 518:1–12. https://doi.org/10.3354/meps11112
Croxall JP, Prince PA (1994) Dead or alive, night or day—how do albatrosses catch squid. Antarct Sci 6:155–162. https://doi.org/10.1017/S0954102094000246
do Amara KB, Alvares DJ, Heinzelmann L et al (2015) Ecological niche modeling of Stenella dolphins (Cetartiodactyla: Delphinidae) in the southwestern Atlantic Ocean. J Exp Mar Biol Ecol 472:166–179. https://doi.org/10.1016/j.jembe.2015.07.013
Dormann CF, Elith J, Bacher S et al (2013) Collinearity: a review of methods to deal with it and a simulation study evaluating their performance. Ecography 36:27–46. https://doi.org/10.1111/j.1600-0587.2012.07348.x
Elith J, Graham C, Anderson R et al (2006) Novel methods improve prediction of species’ distributions from occurrence data. Ecography 29:129–151. https://doi.org/10.1111/j.2006.0906-7590.04596.x
Elith J, Phillips SJ, Hastie T et al (2011) A statistical explanation of MaxEnt for ecologists. Divers Distrib 17:43–57. https://doi.org/10.1111/j.1472-4642.2010.00725.x
Engler R, Guisan A, Rechsteiner L (2004) An improved approach for predicting the distribution of rare and endangered species from occurrence and pseudo-absence data. J Appl Ecol 41:263–274. https://doi.org/10.1111/j.0021-8901.2004.00881.x
Fauchald P, Tveraa T (2003) Using first-passage time in the analysis of area-restricted search and habitat selection. Ecology 84:282–288. https://doi.org/10.1890/0012-9658(2003)084[0282:ufptit]2.0.co;2
Fielding S, Watkins JL, Collins MA et al (2012) Acoustic determination of the distribution of fish and krill across the Scotia Sea in spring 2006, summer 2008 and autumn 2009. Deep Sea Res Part II 59–60:173–188. https://doi.org/10.1016/j.dsr2.2011.08.002
Fielding S, Watkins JL, Trathan PN et al (2014) Interannual variability in Antarctic krill (Euphausia superba) density at South Georgia, Southern Ocean: 1997–2013. ICES J Mar Sci 71:2578–2588. https://doi.org/10.1093/icesjms/fsu104
Froy H, Lewis S, Catry P et al (2015) Age-related variation in foraging behaviour in the wandering albatross at South Georgia: no evidence for senescence. PLoS One 10:e0116415. https://doi.org/10.1371/journal.pone.0116415
Graham MH (2003) Confronting multicollinearity in ecological regression. Ecology 84:2809–2815. https://doi.org/10.1890/02-3114
Granadeiro JP, Phillips RA, Brickle P, Catry P (2011) Albatrosses following fishing vessels: how badly hooked are they on an easy meal? PLoS One 6:e17467. https://doi.org/10.1371/journal.pone.0017467
Grémillet D, Prudor A, le Maho Y, Weimerskirch H (2012) Vultures of the seas: hyperacidic stomachs in wandering albatrosses as an adaptation to dispersed food resources, including fishery wastes. PLoS One 7:e37834. https://doi.org/10.1371/journal.pone.0037834
Halvorsen R, Mazzoni S, Dirksen JW et al (2016) How important are choice of model selection method and spatial autocorrelation of presence data for distribution modelling by MaxEnt? Ecol Model 328:108–118. https://doi.org/10.1016/j.ecolmodel.2016.02.021
Hill S, Belchier M, Collins MA et al (2009) Multiple indicators suggest a strong ecosystem anomaly at South Georgia in 2009. WG-EMM-09/23
Jenouvrier S, Péron C, Weimerskirch H (2015) Extreme climate events and individual heterogeneity shape life-history traits and population dynamics. Ecol Monogr 85:605–624. https://doi.org/10.1890/14-1834.1
Jiménez S, Domingo A, Brazeiro A et al (2016) Sex-related variation in the vulnerability of wandering albatrosses to pelagic longline fleets. Anim Conserv 19:281–295. https://doi.org/10.1111/acv.12245
Jodice PG, Roby DD, Hatch SA et al (2002) Does food availability affect energy expenditure rates of nesting seabirds? A supplemental-feeding experiment with Black-legged Kittiwakes (Rissa tridactyla). Can J Zool 80:214–222. https://doi.org/10.1139/z01-221
Jodice P, Roby DD, Suryan RM et al (2006) Increased energy expenditure by a seabird in response to higher food abundance. Mar Ecol Prog Ser 306:283–293. https://doi.org/10.3354/meps306283
Krüger L, Paiva VH, Petry MV, Ramos JA (2016) Strange lights in the night: using abnormal peaks of light in geolocator data to infer interaction of seabirds with nocturnal fishing vessels. Polar Biol 40:221–226. https://doi.org/10.1007/s00300-016-1933-y
Lobo JM, Jiménez-Valverde A, Real R (2008) AUC: a misleading measure of the performance of predictive distribution models. Glob Ecol Biogeogr 17:145–151. https://doi.org/10.1111/j.1466-8238.2007.00358.x
Louzao M, Pinaud D, Péron C et al (2011) Conserving pelagic habitats: seascape modelling of an oceanic top predator. J Appl Ecol 48:121–132. https://doi.org/10.1111/j.1365-2664.2010.01910.x
Mancini PL, Bond AL, Hobson KA et al (2013) Foraging segregation in tropical and polar seabirds: testing the intersexual competition hypothesis. J Exp Mar Biol Ecol 449:186–193. https://doi.org/10.1016/j.jembe.2013.09.011
Meredith MP, Murphy EJ, Hawker EJ et al (2008) On the interannual variability of ocean temperatures around South Georgia, Southern Ocean: forcing by El Niño/Southern Oscillation and the Southern Annular Mode. Deep Sea Res Part II 55:2007–2022. https://doi.org/10.1016/j.dsr2.2008.05.020
Murphy EJ, Watkins JL, Trathan PN et al (2007) Spatial and temporal operation of the Scotia Sea ecosystem: a review of large-scale links in a krill centred food web. Philos Trans R Soc B Biol Sci 362:113–148. https://doi.org/10.1098/rstb.2006.1957
Naimi B, Skidmore AK, Groen TA, Hamm NAS (2011) Spatial autocorrelation in predictors reduces the impact of positional uncertainty in occurrence data on species distribution modelling. J Biogeogr 38:1497–1509. https://doi.org/10.1111/j.1365-2699.2011.02523.x
Nel DC, Ryan PG, Nel JL et al (2002) Foraging interactions between wandering albatrosses Diomedea exulans breeding on Marion Island and long-line fisheries in the southern Indian Ocean. Ibis 144:E141–E154. https://doi.org/10.1046/j.1474-919X.2002.00092.x
Nevitt GA, Losekoot M, Weimerskirch H (2008) Evidence for olfactory search in wandering albatross, Diomedea exulans. Proc Natl Acad Sci USA 105:4576–4581. https://doi.org/10.1073/pnas.0709047105
Paiva VH, Geraldes P, Ramírez I et al (2010a) Foraging plasticity in a pelagic seabird species along a marine productivity gradient. Mar Ecol Prog Ser 398:259–274. https://doi.org/10.3354/meps08319
Paiva VH, Geraldes P, Ramírez I et al (2010b) How area restricted search of a pelagic seabird changes while performing a dual foraging strategy. Oikos 119:1423–1434. https://doi.org/10.1111/j.1600-0706.2010.18294.x
Paiva VH, Geraldes P, Marques V et al (2013) Effects of environmental variability on different trophic levels of the North Atlantic food web. Mar Ecol Prog Ser 477:15–28. https://doi.org/10.3354/meps10180
Paiva VH, Geraldes P, Rodrigues I et al (2015) The foraging ecology of the endangered cape verde shearwater, a sentinel species for marine conservation off West Africa. PLoS One 10:e0139390. https://doi.org/10.1371/journal.pone.0139390
Paiva VH, Pereira J, Ceia FR, Ramos JA (2017) Environmentally driven sexual segregation in a marine top predator. Sci Rep UK 7:358. https://doi.org/10.1038/s41598-017-02854-2
Pearson RG, Thuiller W, Araújo MB et al (2006) Model-based uncertainty in species range prediction. J Biogeogr 33:1704–1711. https://doi.org/10.1111/j.1365-2699.2006.01460.x
Pereira JM, Paiva VH, Xavier JC (2017) Using seabirds to map the distribution of elusive pelagic cephalopod species. Mar Ecol Prog Ser 567:257–262. https://doi.org/10.3354/meps12020
Phalan B, Phillips RA, Silk J et al (2007) Foraging behaviour of four albatross species by night and day. Mar Ecol Prog Ser 340:271–286. https://doi.org/10.3354/meps340271
Phillips RA, Silk JRD, Phalan B et al (2004) Seasonal sexual segregation in two Thalassarche albatross species: competitive exclusion, reproductive role specialization or foraging niche divergence? Proc R Soc B Biol Sci 271:1283–1291. https://doi.org/10.1098/rspb.2004.2718
Phillips SJ, Anderson RP, Schapire RE (2006) Maximum entropy modeling of species geographic distributions. Ecol Model 190:231–259. https://doi.org/10.1016/j.ecolmodel.2005.03.026
Phillips RA, McGill RAR, Dawson DA, Bearhop S (2011) Sexual segregation in distribution, diet and trophic level of seabirds: insights from stable isotope analysis. Mar Biol 158:2199–2208. https://doi.org/10.1007/s00227-011-1725-4
Pinaud D, Weimerskirch H (2005) Scale-dependent habitat use in a long-ranging central place predator. J Anim Ecol 74:852–863. https://doi.org/10.1111/j.1365-2656.2005.00984.x
Pinet P, Jaquemet S, Phillips RA, Le Corre M (2012) Sex-specific foraging strategies throughout the breeding season in a tropical, sexually monomorphic small petrel. Anim Behav 83:979–989. https://doi.org/10.1016/j.anbehav.2012.01.019
R Development Core Team (2016) R: a language and environment for statistical computing, Vienna
Ramos JA, Isabel Fagundes A, Xavier JC et al (2015) A switch in the Atlantic Oscillation correlates with inter-annual changes in foraging location and food habits of Macaronesian shearwaters (Puffinus baroli) nesting on two islands of the sub-tropical Atlantic Ocean. Deep Sea Res Part I 104:60–71. https://doi.org/10.1016/j.dsr.2015.07.001
Sachs G, Traugott J, Nesterova AP et al (2012) Flying at no mechanical energy cost: disclosing the secret of wandering albatrosses. PLoS One 7:e41449. https://doi.org/10.1371/journal.pone.0041449
Seco J, Roberts J, Ceia FR et al (2016) Distribution, habitat and trophic ecology of Antarctic squid Kondakovia longimana and Moroteuthis knipovitchi: inferences from predators and stable isotopes. Polar Biol 39:167–175. https://doi.org/10.1007/s00300-015-1675-2
Shaffer SA, Weimerskirch H, Costa DP (2001) Functional significance of sexual dimorphism in wandering albatrosses, Diomedea exulans. Funct Ecol 15:203–210. https://doi.org/10.1046/j.1365-2435.2001.00514.x
Shaffer SA, Costa DP, Weimerskirch H (2003) Foraging effort in relation to the constraints of reproduction in free-ranging albatrosses. Funct Ecol 17:66–74. https://doi.org/10.1046/j.1365-2435.2003.00705.x
Tancell C, Phillips RA, Xavier JC et al (2013) Comparison of methods for determining key marine areas from tracking data. Mar Biol 160:15–26. https://doi.org/10.1007/s00227-012-2050-2
Wakefield ED, Bodey TW, Bearhop S et al (2013) Space partitioning without territoriality in gannets. Science 341:68–70. https://doi.org/10.1126/science.1236077
Warren DL, Seifert SN (2011) Ecological niche modeling in Maxent: the importance of model complexity and the performance of model selection criteria. Ecol Appl 21:335–342
Warren DL, Glor RE, Turelli M (2008) Environmental niche equivalency versus conservatism: quantitative approaches to niche evolution. Evolution 62:2868–2883. https://doi.org/10.1111/j.1558-5646.2008.00482.x
Warren DL, Glor RE, Turelli M (2010) ENMTools: a toolbox for comparative studies of environmental niche models. Ecography 35:1160–1611. https://doi.org/10.1111/j.1600-0587.2009.06142.x
Weimerskirch H, Lys P (2000) Seasonal changes in the provisioning behaviour and mass of male and female wandering albatrosses in relation to the growth of their chick. Polar Biol 23:733–744. https://doi.org/10.1007/s003000000144
Weimerskirch H, Wilson RP (1992) When do wandering albatrosses Diomedea exulans forage? Mar Ecol Prog Ser 86:297–300. https://doi.org/10.3354/meps086297
Weimerskirch H, Salamolard M, Sarrazin F, Jouventin P (1993) Foraging strategy of wandering albatrosses through the breeding-season—a study using satellite telemetry. Auk 110:325–342
Weimerskirch H, Doncaster CP, Cuenot-Chaillet F (1994) Pelagic seabirds and the marine environment: foraging patterns of wandering albatrosses in relation to prey availability and distribution. Proc R Soc B Biol Sci 255:91–97. https://doi.org/10.1098/rspb.1994.0013
Weimerskirch H, Cherel Y, Cuenot-Chaillet F, Ridoux V (1997a) Alternative foraging strategies and resource allocation by male and female wandering albatrosses. Ecology 78:2051–2063. https://doi.org/10.2307/2265944
Weimerskirch H, Wilson RP, Lys P (1997b) Activity pattern of foraging in the wandering albatross: a marine predator with two modes of prey searching. Mar Ecol Prog Ser 151:245–254. https://doi.org/10.3354/meps151245
Weimerskirch H, Barbraud C, Lys P (2000) Sex differences in parental investment and chick growth in wandering albatrosses: fitness consequences. Ecology 81:309–318. https://doi.org/10.2307/177428
Weimerskirch H, Pinaud D, Pawlowski F, Bost C (2007) Does prey capture induce area-restricted search? A fine-scale study using GPS in a marine predator, the wandering albatross. Am Nat 170:734. https://doi.org/10.2307/4541144
Weimerskirch H, Louzao M, de Grissac S, Delord K (2012) Changes in wind pattern alter albatross distribution and life-history traits. Science 335:211–214. https://doi.org/10.1126/science.1210270
Wilson RP, Cooper J, Plötz J (1992) Can we determine when marine endotherms feed? A case study with seabirds. J Exp Biol 167:267–275
Wilson RP, Pütz K, Grémillet D et al (1995) Realiability of stomach temperature changes in determining feeding characteristics of seabirds. J Exp Biol 198:1115–1135
Xavier JC, Cherel Y (2009) Cephalopod beak guide for the Southern Ocean. British Antarctic Survey, Cambridge
Xavier JC, Croxall JP (2005) Sexual differences in foraging behaviour and diets: a case of study of wandering albatrosses. In: Ruckstuhl KE, Neuhaus P (eds) Sexual segregation in vertebrates: ecology of the two sexes. Cambridge University Press, Cambridge, pp 74–91
Xavier JC, Croxall JP (2007) Predator-prey interactions: why do larger albatrosses eat bigger squid? J Zool 271:408–417. https://doi.org/10.1111/j.1469-7998.2006.00224.x
Xavier JC, Rodhouse PG, Trathan PN, Wood AG (1999) A geographical information system (GIS) atlas of cephalopod distribution in the southern ocean. Antarct Sci 11:61–62. https://doi.org/10.1017/S0954102099000097
Xavier J, Croxall J, Trathan P, Rodhouse P (2003a) Inter-annual variation in the cephalopod component of the diet of the wandering albatross, Diomedea exulans, breeding at Bird Island, South Georgia. Mar Biol 142:611–622. https://doi.org/10.1007/s00227-002-0962-y
Xavier JC, Croxall JP, Trathan PN, Wood AG (2003b) Feeding strategies and diets of breeding grey-headed and wandering albatrosses at South Georgia. Mar Biol 143:221–232. https://doi.org/10.1007/s00227-003-1049-0
Xavier JC, Trathan PN, Croxall JP et al (2004) Foraging ecology and interactions with fisheries of wandering albatrosses (Diomedea exulans) breeding at South Georgia. Fish Oceanogr 13:324–344. https://doi.org/10.1111/j.1365-2419.2004.00298.x
Xavier JC, Tarling AG, Croxall JP (2006) Determining prey distribution patterns from stomach-contents of satellite-tracked high-predators of the Southern Ocean. Ecography 29:260–272. https://doi.org/10.1111/j.2006.0906-7590.04525.x
Xavier JC, Louzao M, Thorpe SE et al (2013) Seasonal changes in the diet and feeding behaviour of a top predator indicate a flexible response to deteriorating oceanographic conditions. Mar Biol 160:1597–1606. https://doi.org/10.1007/s00227-013-2212-x
Xavier JC, Raymond B, Jones DC, Griffiths H (2016) Biogeography of cephalopods in the southern ocean using habitat suitability prediction models. Ecosystems 19:220–247. https://doi.org/10.1007/s10021-015-9926-1
Xavier JC, Trathan PN, Ceia FR et al (2017) Sexual and individual foraging segregation in gentoo penguins Pygoscelis papua from the Southern Ocean during an abnormal winter. PLoS One 12:e0174850. https://doi.org/10.1371/journal.pone.0174850
Zuur AF, Ieno EN, Elphick CS (2009) A protocol for data exploration to avoid common statistical problems. Methods Ecol Evol 1:3–14. https://doi.org/10.1111/j.2041-210X.2009.00001.x
Zwolinski JP, Oliveira PB, Quintino V, Stratoudakis Y (2010) Sardine potential habitat and environmental forcing off western Portugal. ICES J Mar Sci 67:1553–1564. https://doi.org/10.1093/icesjms/fsq068
Acknowledgements
We thank Jaime Ramos for very useful comments on earlier drafts of the manuscript. This study is a contribution to SCAR AnT-ERA (Antarctic Thresholds—Ecosystem Resilience and Adaptation) program, ICED (Integrating climate and Ecosystem Dynamics), BAS-CEPH program and to the PROPOLAR (Programa Polar Português). This study benefited from the strategic program of MARE, financed by FCT (MARE- UID/MAR/04292/2013). JCX was supported by the Investigator FCT program (IF/00616/2013) and VHP by a FCT fellowship (SFRH/BPD/85024/2012).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare they have no conflict of interests.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. The animal procedures used in this study were reviewed and approved by the Joint British Antarctic Survey–Cambridge University Animal Welfare and Ethical Review Committee. Permits to operate were issued by the Government of South Georgia and the South Sandwich Islands.
Additional information
Responsible Editor: Y. Cherel.
Reviewed by Undisclosed experts.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pereira, J.M., Paiva, V.H., Phillips, R.A. et al. The devil is in the detail: small-scale sexual segregation despite large-scale spatial overlap in the wandering albatross. Mar Biol 165, 55 (2018). https://doi.org/10.1007/s00227-018-3316-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00227-018-3316-0