Abstract
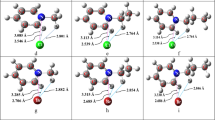
A special class of hydrogen-binding metal complexes are complexes able to bind hydrogen molecules at one or more of their ligand positions. One of such complexes, Fe(H)2(H2)(PEtPh2)3, was characterized experimentally and theoretically in previous works. Its specific properties were related to the asymmetry of the non-hydrogen ligands. Following this reasoning, attachment and dissociation of hydrogen molecule to and from the 5-coordinated and 6-coordinated complex were investigated theoretically. Relaxed and partially constrained potential-energy scans were performed and transition-states for these processes were investigated. Non-hydrogen ligand asymmetry seems to reflect on the different barrier energies for approach to and dissociation from the two dihydrogen ligand positions. Steric and environment effects are estimated comparing behavior for partially constrained and gas-phase models. On the basis of these findings, theoretically predicted pathways for single-step dihydrogen binding and dissociation processes are established, and means for experimental verification are proposed.








Similar content being viewed by others
References
Layzell D, Strem G, Caldwell G, Jamieson B, Pouliot M (2020) Powering the circular economy: the role of hydrogen. Circular Economy Leadership Coalition. September 27, 2020. https://circulareconomyleaders.ca/powering-the-circular-economy/
Liptak BG (2009) Post-oil energy technology the world’s first solar-hydrogen demonstration power plant. CRC Press, Boca Raton
Rifkin J (2003) The hydrogen economy: the creation of the worldwide energy web and the redistribution of power on earth, 1st edn. TarcherPerigee, New York
Hoffmann P (2012) Tomorrow’s Energy: Hydrogen, Fuel Cells, and the Prospects for a Cleaner Planet, revised and expanded. The MIT Press, Cambridge
International Association for Hydrogen Energy (2020) Founding Editor-in-Chief T. N. Veziroglu. Int J Hydrogen Energy Elsevier B.V@@
de Miranda PEV (2019). In: de Miranda PEV (ed) Science and engineering of hydrogen-based energy technologies. Elsevier, Amsterdam, p 2019
European Comission (2014) Hydrogen Europe and Hydrogen Europe Research Joint Undertaking, Fuel Cells and Hydrogen (November 14, 2020). https://www.fch.europa.eu/page/documents
Kubas GJ, Ryan RR, Swanson BI, Vergamini PJ, Wasserman HJ (1984) JACS 100(2):451–452
Kubas GJ (2009) J Organomet Chem 694:2648–2653
Brayshaw SK, Harrison A, McIndoe JS, Marken F, Raithby PR, Warren JE, Weller AS (2007) JACS 129:1793–1804
Yang YX, Singh RK, Webley PA (2008) Adsorption 14:265–274
Bushnell JE, Kemper PR, van Koppen P, Bowers MT (2001) J Phys Chem A 105:2216–2224
Otsuka K, Takenaka S (2004) J Jpn Petrol Inst 47(6):377–386
Weck PF, Kumar TJD, Kim E, Balakrishnan N (2007) J Chem Phys 126:094703
Durgun E, Ciraci S, Zhou W, Yildirim T (2006) arXiv:cond-mat/0609068v2 [cond-mat.other] 30 oct 2006
Dong Q, Tian WQ, Chen D-L, Sun C-C (2009) Int J Hydrogen Energ 34:5444–5448
Maseras F, Lledós A, Clot E, Eisenstein O (2000) Chem Rev 100:601–636
Kubas GJ (2005) Catalysis Lett 104:79–101
Dutta S (2011) C R Chimie 14:1029–1053
Weller AS, McIndoe JS (2007) Eur J Inorg Chem 2007:4411–4423
Wiedner ES, Chambers MB, Pitman CL, Bullock RM, Miller AJM, Appel AM (2016) Chem Rev 116:8655–8692
Crabtree RH (2016) Chem Rev 116(15):8750–8769
Valencia H, Gil A, Frapper G (2015) J Phys Chem C 10:5506–5522
Manadé M, Vines F, Gil A, Illas F (2018) Phys Chem Chem Phys 20(5):3819–3830
Di Liberto G, Cipriano LA, Pacchioni G (2021) J Am Chem Soc 143(48):20431–20441
Jeyakumar TC, Baskaran S, Sivasankar C (2018) J Chem Sci 130:57
Van der Sluys LS, Eckert J, Eisenstein O, Hall JH, Huffman JC, Jackson SA, Koetzle TF, Kubas GJ, Vergamini PJ, Caulton KG (1990) JACS 112:4831–4841
Došlić N, Gomzi V, Mališ M, Matanović I, Eckert J (2011) Inorg Chem 50(21):10740–10747
Gonzalez ME, Eckert J, Aquino AJA, Poirier B (2018) J Chem Phys 148(15):154303
Morris L, Hales JJ, Trudeau ML, Georgiev P, Embs JP, Eckert J, Kaltsoyannis N, Antonelli DM (2019) Energy Environ Sci 12:1580–1591
Allendorf MD et al (2018) Energy Environ Sci 11:2784–2812
Kubas GJ (2001) Metal dihydrogen and σ-bond complexes. Springer, Berlin
Kubas GJ (2007) Chem Rev 107(10):4152–4205
Nguyen MC, Lee H, Ihm J (2008) Solid State Comm 147:419–422
Aresta M, Giannoccaro P, Rossi M, Sacco A (1971) Inorg Chim Acta 5(1):115–118
Becke AD (1993) J Chem Phys 98:5648–5652
Rassolov VA, Pople JA, Ratner MA, Windus TL (1998) J Chem Phys 109:1223–1229
Rassolov VA, Ratner MA, Pople JA, Redfern PC, Curtiss LA (2001) J. Comp. Chem. 22:976–984
Check CE, Faust TO, Bailey JM, Wright BJ, Gilbert TM, Sunderlin LS (2001) J Phys Chem A 105(34):8111–8116
Dunning TH Jr, Hay PJ (1997). In: Schaefer HF III (ed) Methods of electronic structure theory, vol 2. Plenum Press, Berlin
Hay J, Wadt WR (1985) J Chem Phys 82:284
Abrecht DG, Fultz B (2012) J Phys Chem C Nanomater Interfaces 116(42):22245–22252
Devarajan D, Ess DH (2012) Inorg Chem 51:6367–6375
Barea G, Ujaque G, Maseras F, Lledos A (1996) J Mol Struc Theochem 371:59–68
Schaefer A, Huber C, Ahlrichs R (1994) J Chem Phys 100:5829–5835
Grimme S, Antony J, Ehrlich S, Krieg H (2010) J Chem Phys 132:154104
Tomasi J, Mennucci B, Cammi R (2005) Chem Rev 105:2999–3093
Frisch MJ et al (2013) Gaussian 09, Revision D01. Gaussian, Inc., Wallingford
Peng C, Schlegel HB (1993) Israel J Chem 33:449–454
Jansen HB, Ros P (1969) Chem Phys Lett 3:140–143
Liu B, McLean AD (1973) J Chem Phys 59:4557–4558
Boys SF, Bernardi F (1970) Mol Phys 19(4):553–566
Simon S, Duran M, Dannenberg JJ (1996) J Chem Phys 105:11024–11031
Dunning TH (2000) J Phys Chem A 104:9062
Liedl KR (1998) J Chem Phys 108:3199
Halkier A, Klopper W, Helgaker T, Jørgensen P, Taylor PR (1999) J Chem Phys 111:9157
Asturiol D, Duran M, Salvador P (2008) J Chem Phys 128:144108
Ren J, Musyoka NM, Langmi HW, Mathe M, Liao S (2017) Int J Hydrogen Energy 42(1):289–311
Rusman NAA, Dahari M (2016) Int J Hydrogen Energy 41(28):12108–12126
Bérubé V, Radtke G, Dresselhaus M, Chen G (2007) Int J Energy Res 31:637–663
Sandhya KS, Suresh CH (2012) Dalton Trans 41:11018–11025
Sandhya KS, Suresh CH (2014) Dalton Trans 43:12279–12287
Belkova NV, Epstein LM, Filippov OA, Shubina ES (2016) Chem Rev 116:8545–8587
Perutz RN, Procacci B (2016) Chem Rev 116:8506–8544
Runcevski T, Kapelewski MT, Torres-Gavosto RM, Tarver JD, Brown CM, Long JR (2016) Chem Commun 52:8251–8254
Acknowledgements
During this work, the Croatian National Grid (CRO-NGI) computational resources have been used.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gomzi, V. Dihydrogen attachment and dissociation reactions in Fe(H)2(H2)(PEtPh2)3: a DFT potential-energy scan. Theor Chem Acc 141, 12 (2022). https://doi.org/10.1007/s00214-022-02870-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-022-02870-0