Abstract
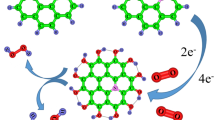
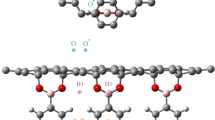
We utilized density functional theory (DFT) study to understand the adsorption mechanism of lithium oxides (LixO2) onto N-doped graphene during oxygen reduction reaction (ORR) for lithium–air batteries. We systematically proposed two possible ORR pathways and examined various adsorption configurations in each system, including for the O2 and Li ORR reactants and the LiO2 and Li2O2 ORR products. The doping of the N atom into graphene was calculated to enhance the adsorption of O2, but to attenuate the adsorption of Li, because of the repulsion between the electron-rich N-doped graphene and the electron-donating Li atom, and the attraction of this N-doped graphene for electronegative O2. Nevertheless, since the adsorption of Li onto N-doped graphene (−1.001 to −0.503 eV) was still stronger than the adsorption of O2 (−0.280 to −0.215 eV), Li should bind N-doped graphene first. Moreover, N-doped graphene was calculated to bind LiO2 (−0.588 eV) more strongly than was pristine graphene (−0.450 eV). Additionally, the Li2O2 configuration that yielded the most stable adsorption on N-doped graphene was calculated to yield an adsorption energy of −0.642 eV, which is more favorable than that for pristine graphene (−0.630 eV). Overall, N-doped graphene was found to strengthen the adsorption of lithium oxides (LixO2) and increase charge transfer to substantial levels.







Similar content being viewed by others
References
Girishkumar G, McCloskey B, Luntz AC, Swanson S, Wilcke W (2010) J Phys Chem Lett 1:2193
Kraytsberg A, Ein-Eli Y (2011) J Power Sources 196:886
Franco AA, Xue KH (2013) Ecs J Solid State Sci Technol 2:M3084
Allen MJ, Tung VC, Kaner RB (2010) Chem Rev 110:132
Xiao J, Mei DH, Li XL, Xu W, Wang DY, Graff GL, Bennett WD, Nie ZM, Saraf LV, Aksay IA, Liu J, Zhang JG (2011) Nano Lett 11:5071
Yoo E, Zhou HS (2011) ACS Nano 5:3020
Wang ZL, Xu D, Xu JJ, Zhang LL, Zhang XB (2012) Adv Funct Mater 22:3699
Yoo E, Zhou HS (2014) RSC Adv 4:13119
Ren XD, Zhu JZ, Du FM, Liu JJ, Zhang WQ (2014) J Phys Chem C 118:22412
Wang S, Dong SM, Wang J, Zhang LX, Han PX, Zhang CJ, Wang XG, Zhang KJ, Lan ZG, Cui GL (2012) J Mater Chem 22:21051
Li YL, Wang JJ, Li XF, Geng DS, Banis MN, Li RY, Sun XL (2012) Electrochem Commun 18:12
Lin ZY, Waller GH, Liu Y, Liu ML, Wong CP (2013) Carbon 53:130
Li Q, Cao RG, Cho J, Wu G (2014) PCCP 16:13568
Debart A, Bao J, Armstrong G, Bruce PG (2007) J Power Sources 174:1177
Lu YC, Xu ZC, Gasteiger HA, Chen S, Hamad-Schifferli K, Shao-Horn Y (2010) J Am Chem Soc 132:12170
Lu YC, Gasteiger HA, Parent MC, Chiloyan V, Shao-Horn Y (2010) Electrochem Solid State Lett 13:A69
Choi R, Jung J, Kim G, Song K, Kim YI, Jung SC, Han YK, Song H, Kang YM (2014) Energy Environ Sci 7:1362
Su DW, Kim HS, Kim WS, Wang GX (2013) J Power Sources 244:488
Wang YG, Zhou HS (2010) J Power Sources 195:358
Cheng H, Scott K (2010) J Power Sources 195:1370
Debart A, Paterson AJ, Bao J, Bruce PG (2008) Angewandte Chemie-Int Edn 47:4521
Minowa H, Hayashi M, Hayashi K, Kobayashi R, Takahashi K (2013) J Power Sources 244:17
Chen Y, Zhang Q, Zhang Z, Zhou X, Zhong Y, Yang M, Xie Z, Wei J, Zhou Z (2015) J Mater Chem A 3:17874
Zhang Z, Bao J, He C, Chen Y, Wei J, Zhou Z (2014) Adv Funct Mater 24:6826
Jing Y, Zhou Z (2015) ACS Catal 5:4309
Wei DC, Liu YQ, Wang Y, Zhang HL, Huang LP, Yu G (2009) Nano Lett 9:1752
Guo BD, Liu QA, Chen ED, Zhu HW, Fang LA, Gong JR (2010) Nano Lett 10:4975
Sheng ZH, Shao L, Chen JJ, Bao WJ, Wang FB, Xia XH (2011) ACS Nano 5:4350
Lin YC, Lin CY, Chiu PW (2010) Appl Phys Lett 96:133110
Kresse G, Furthmuller J (1996) Phys Rev B 54:11169
Kresse G, Furthmuller J (1996) Comp Mater Sci 6:15
Blochl PE (1994) Phys Rev B 50:17953
Perdew JP, Burke K, Ernzerhof M (1996) Phys Rev Lett 77:3865
Perdew JP, Burke K, Wang Y (1996) Phys Rev B 54:16533
Park H, Noh SH, Lee JH, Lee WJ, Jaung JY, Lee SG, Han TH (2015) Sci Rep 5:14163
Koh W, Lee JH, Lee SG, Choi JI, Jang SS (2015) RSC Adv 5:32819
Moon HS, Lee JH, Kwon S, Kim IT, Lee SG (2015) Carbon Lett 16:116
Koh W, Choi JI, Lee SG, Lee WR, Jang SS (2011) Carbon 49:286
Koh W, Choi JI, Jeong E, Lee SG, Jang SS (2014) Curr Appl Phys 14:1748
Kwon S, Lee SG (2015) Carbon Lett 16:198
Koh W, Choi JI, Donaher K, Lee SG, Jang SS (2011) ACS Appl Mater Inter 3:1186
Koh W, Moon HS, Lee SG, Choi JI, Jang SS (2015) ChemPhysChem 16:789
Yu Y-X (2013) PCCP 15:16819
Grimme S, Antony J, Ehrlich S, Krieg H (2010) J Chem Phys 132:154104
Manz TA, Sholl DS (2010) J Chem Theory Comput 6:2455
Lee JH, Kang SG, Moon HS, Park H, Kim IT, Lee SG (2015) Appl Surf Sci 351:193
Wu DH, Li YF, Zhou Z (2011) Theor Chem Acc 130:209
Rani P, Jindal VK (2013) RSC Adv 3:802
Acknowledgments
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (2014R1A1A1004096 and 2015R1C1A1A02036472). This work was financially supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIP) through GCRC-SOP (No. 2011-0030013).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, J.H., Kang, S.G., Kim, I.T. et al. Adsorption mechanisms of lithium oxides (LixO2) on N-doped graphene: a density functional theory study with implications for lithium–air batteries. Theor Chem Acc 135, 50 (2016). https://doi.org/10.1007/s00214-016-1805-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00214-016-1805-0