Abstract
Rationale and objectives
Nicotine and d-amphetamine can strengthen reinforcing effects of unconditioned visual stimuli. We investigated whether these reinforcement-enhancing effects reflect a slowing of stimulus habituation and depend on food restriction.
Methods
Adult male rats pressed an active lever to illuminate a cue light during daily 60-min sessions. Depending on the experiment, rats were challenged with fixed or varying doses of d-amphetamine (0.25–2 mg/kg IP) and nicotine (0.025–0.2 mg/kg SC) or with the tobacco constituent norharman (0.03–10 μg/kg IV). Experiment 1 tested for possible reinforcement-enhancing effects of d-amphetamine and norharman. Experiment 2 investigated whether nicotine and amphetamine inhibited the spontaneous within-session decline in lever pressing. Experiment 3 assessed the effects of food restriction.
Results
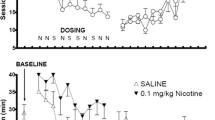
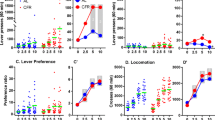
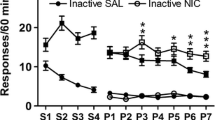
Amphetamine (0.25–1 mg/kg) and nicotine (0.1 mg/kg) increased active lever pressing specifically (two- to threefold increase). The highest doses of nicotine and amphetamine also affected inactive lever responding (increase and decrease, respectively). With the visual reinforcer omitted, responding was largely extinguished. Neither drug appeared to slow habituation, as assessed by the within-session decline in lever pressing, and reinforcement-enhancing effects still occurred if the drugs were given after this decline had occurred. Food restriction enhanced the reinforcement-enhancing effect of amphetamine but not that of nicotine.
Conclusions
Responding remained goal-directed after several weeks of testing. Low doses of d-amphetamine and nicotine produced reinforcement enhancement even in free-feeding subjects, independent of the spontaneous within-session decline in responding. Reinforcement enhancement by amphetamine, but not nicotine, was enhanced by concurrent subchronic food restriction.




Similar content being viewed by others
References
Barrett ST, Bevins RA (2013) Nicotine enhances operant responding for qualitatively distinct reinforcers under maintenance and extinction conditions. Pharmacol Biochem Behav 114-115:9–15. https://doi.org/10.1016/j.pbb.2013.10.012
Barrett ST, Odum AL (2011) The effects of repeated exposure on the reward-enhancing effects of nicotine. Behav Pharmacol 22(4):283–290. https://doi.org/10.1097/FBP.0b013e3283473c25
Berlyne DE, Salapatek PH, Gelman RS, Zener SL (1964) Is light increment really rewarding to the rat? J Comp Physiol Psychol 58(1):148–151. https://doi.org/10.1037/h0047385
Cabeza de Vaca S, Carr KD (1998) Food restriction enhances the central rewarding effect of abused drugs. J Neurosci 18(18):7502–7510
Cadoni C, Solinas M, Valentini V, Di Chiara G (2003) Selective psychostimulant sensitization by food restriction: differential changes in accumbens shell and core dopamine. Eur J Neurosci 18(8):2326–2334. https://doi.org/10.1046/j.1460-9568.2003.02941.x
Caggiula AR, Donny EC, White AR, Chaudhri N, Booth S, Gharib MA, Hoffman A, Perkins KA, Sved AF (2002) Environmental stimuli promote the acquisition of nicotine self-administration in rats. Psychopharmacology 163(2):230–237. https://doi.org/10.1007/s00213-002-1156-5
Cain ME, Green TA, Bardo MT (2006) Environmental enrichment decreases responding for visual novelty. Behav Process 73(3):360–366. https://doi.org/10.1016/j.beproc.2006.08.007
Carr KD (2002) Augmentation of drug reward by chronic food restriction: behavioral evidence and underlying mechanisms. Physiol Behav 76(3):353–364. https://doi.org/10.1016/S0031-9384(02)00759-X
Chaudhri N, Caggiula AR, Donny EC, Booth S, Gharib MA, Craven LA, Allen SS, Sved AF, Perkins KA (2005) Sex differences in the contribution of nicotine and nonpharmacological stimuli to nicotine self-administration in rats. Psychopharmacology 180(2):258–266. https://doi.org/10.1007/s00213-005-2152-3
Clarke PBS (1984) Chronic central nicotinic blockade after a single administration of the bisquaternary ganglion-blocking drug chlorisondamine. Br J Pharmacol 83(2):527–535. https://doi.org/10.1111/j.1476-5381.1984.tb16517.x
Clarke PBS (1987) Nicotine and smoking: a perspective from animal studies. Psychopharmacology 92(2):135–143
Clarke PBS, Kumar R (1984) Effects of nicotine and d-amphetamine on intracranial self-stimulation in a shuttle box test in rats. Psychopharmacology 84(1):109–114. https://doi.org/10.1007/BF00432037
Collins GT, Woods JH (2009) Influence of conditioned reinforcement on the response-maintaining effects of quinpirole in rats. Behav Pharmacol 20(5-6):492–504. https://doi.org/10.1097/FBP.0b013e328330ad9b
Constantin A, Clarke PBS (2017) Reinforcement enhancement by nicotine in adult rats: behavioral selectivity and relation to mode of delivery and blood nicotine levels. Psychopharmacology (Berl). https://doi.org/10.1007/s00213-017-4778-3
Dickinson A, Nicholas DJ, Adams D (1982) The effect of the instrumental training contingency on susceptibility to reinforcer devaluation. Q J Exp Psychol 35:35–51
Dieu Y, Seillier A, Majchrzak M, Marchand A, Di Scala G (2005) Systemic or intra-accumbens injection of D-amphetamine delays habituation to a tone stimulus in rats. Behav Pharmacol 16(1):35–42. https://doi.org/10.1097/00008877-200502000-00004
Esposito RU, Perry W, Kornetsky C (1980) Effects of d-amphetamine and naloxone on brain stimulation reward. Psychopharmacology 69(2):187–191. https://doi.org/10.1007/BF00427648
File SE (1975) Effects of parachlorophenylalanine and amphetamine on habituation of orienting. Pharmacol Biochem Behav 3(6):979–983. https://doi.org/10.1016/0091-3057(75)90005-2
Gancarz AM, San George MA, Ashrafioun L, Richards JB (2011) Locomotor activity in a novel environment predicts both responding for a visual stimulus and self-administration of a low dose of methamphetamine in rats. Behav Process 86(2):295–304. https://doi.org/10.1016/j.beproc.2010.12.013
Ghosheh O, Dwoskin LP, Li WK, Crooks PA (1999) Residence times and half-lives of nicotine metabolites in rat brain after acute peripheral administration of [2′-(14)C]nicotine. Drug Metab Dispos 27(12):1448–1455
Grilly DM, Loveland A (2001) What is a “low dose” of d-amphetamine for inducing behavioral effects in laboratory rats? Psychopharmacology 153(2):155–169. https://doi.org/10.1007/s002130000580
Harris AC, Stepanov I, Pentel PR, LeSage MG (2012) Delivery of nicotine in an extract of a smokeless tobacco product reduces its reinforcement-attenuating and discriminative stimulus effects in rats. Psychopharmacology 220(3):565–576. https://doi.org/10.1007/s00213-011-2514-y
Harrison AA, Gasparini F, Markou A (2002) Nicotine potentiation of brain stimulation reward reversed by DHbetaE and SCH 23390, but not by eticlopride, LY 314582 or MPEP in rats. Psychopharmacology 160(1):56–66. https://doi.org/10.1007/s00213-001-0953-6
Inoue K, Zorrilla EP, Tabarin A, Valdez GR, Iwasaki S, Kiriike N, Koob GF (2004) Reduction of anxiety after restricted feeding in the rat: implication for eating disorders. Biol Psychiatry 55(11):1075–1081. https://doi.org/10.1016/j.biopsych.2004.01.026
Keller KL, Vollrath-Smith FR, Jafari M, Ikemoto S (2014) Synergistic interaction between caloric restriction and amphetamine in food-unrelated approach behavior of rats. Psychopharmacology 231(5):825–840. https://doi.org/10.1007/s00213-013-3300-9
Kiernan CC (1965) Modification of the effects of amphetamine sulphate by past experience in the hooded rat. Psychopharmacologia 8(1):23–31. https://doi.org/10.1007/BF00405358
Liebman JM, Butcher LL (1974) Comparative involvement of dopamine and noradrenaline in rate-free self-stimulation in substania nigra, lateral hypothalamus, and mesencephalic central gray. Naunyn Schmiedeberg’s Arch Pharmacol 284(2):167–194. https://doi.org/10.1007/BF00501121
Lloyd DR, Gancarz AM, Ashrafioun L, Kausch MA, Richards JB (2012a) Habituation and the reinforcing effectiveness of visual stimuli. Behav Process 91(2):184–191. https://doi.org/10.1016/j.beproc.2012.07.007
Lloyd DR, Hausknecht KA, Richards JB (2014) Nicotine and methamphetamine disrupt habituation of sensory reinforcer effectiveness in male rats. Exp Clin Psychopharmacol 22(2):166–175. https://doi.org/10.1037/a0034741
Lloyd DR, Kausch MA, Gancarz AM, Beyley LJ, Richards JB (2012b) Effects of novelty and methamphetamine on conditioned and sensory reinforcement. Behav Brain Res 234(2):312–322. https://doi.org/10.1016/j.bbr.2012.07.012
Ludbrook J (1998) Multiple comparison procedures updated. Clin Exp Pharmacol Physiol 25(12):1032–1037. https://doi.org/10.1111/j.1440-1681.1998.tb02179.x
Peng XX, Lister A, Rabinowitsch A, Kolaric R, Cabeza de Vaca S, Ziff EB, Carr KD (2015) Episodic sucrose intake during food restriction increases synaptic abundance of AMPA receptors in nucleus accumbens and augments intake of sucrose following restoration of ad libitum feeding. Neuroscience 295:58–71. https://doi.org/10.1016/j.neuroscience.2015.03.025
Perkins KA, Karelitz JL, Boldry MC (2017) Nicotine acutely enhances reinforcement from non-drug rewards in humans. Front Psychiatry 8:article 65. https://doi.org/10.3389/fpsyt.2017.00065
Phillips AG, Fibiger HC (1990) Role of reward and enhancement of conditioned reward in persistence of responding for cocaine. Behav Pharmacol 1(4):269–282. https://doi.org/10.1097/00008877-199000140-00002
Pratt JA, Stolerman IP, Garcha HS, Giardini V, Feyerabend C (1983) Discriminative stimulus properties of nicotine: further evidence for mediation at a cholinergic receptor. Psychopharmacology 81(1):54–60. https://doi.org/10.1007/BF00439274
Robbins TW, Watson BA, Gaskin M, Ennis C (1983) Contrasting interactions of pipradrol, d-amphetamine, cocaine, cocaine analogues, apomorphine and other drugs with conditioned reinforcement. Psychopharmacology 80(2):113–119. https://doi.org/10.1007/BF00427952
Rommelspacher H, Meier-Henco M, Smolka M, Kloft C (2002) The levels of norharman are high enough after smoking to affect monoamineoxidase B in platelets. Eur J Pharmacol 441(1-2):115–125. https://doi.org/10.1016/S0014-2999(02)01452-8
Rupprecht LE, Smith TT, Schassburger RL, Buffalari DM, Sved AF, Donny EC (2015) Behavioral mechanisms underlying nicotine reinforcement. Curr Top Behav Neurosci 24:19–53. https://doi.org/10.1007/978-3-319-13482-6_2
Shin R, Cao J, Webb SM, Ikemoto S (2010) Amphetamine administration into the ventral striatum facilitates behavioral interaction with unconditioned visual signals in rats. PLoS One 5(1):e8741. https://doi.org/10.1371/journal.pone.0008741
Simola N, Frau L, Plumitallo A, Morelli M (2014) Direct and long-lasting effects elicited by repeated drug administration on 50-kHz ultrasonic vocalizations are regulated differently: implications for the study of the affective properties of drugs of abuse. Int J Neuropsychopharmacol 17(03):429–441. https://doi.org/10.1017/S1461145713001235
Sood P, Cole S, Fraier D, Young AM (2009) Evaluation of metaquant microdialysis for measurement of absolute concentrations of amphetamine and dopamine in brain: a viable method for assessing pharmacokinetic profile of drugs in the brain. J Neurosci Methods 185(1):39–44. https://doi.org/10.1016/j.jneumeth.2009.09.004
Sorge RE, Pierre VJ, Clarke PB (2009) Facilitation of intravenous nicotine self-administration in rats by a motivationally neutral sensory stimulus. Psychopharmacology 207(2):191–200. https://doi.org/10.1007/s00213-009-1647-8
Stewart J (1960) Reinforcing effects of light as a function of intensity and reinforcement schedule. J Comp Physiol Psychol 53(2):187–193. https://doi.org/10.1037/h0047315
Veltri T, Taroyan N, Overton PG (2017) Nicotine enhances an auditory event-related potential component which is inversely related to habituation. J Psychopharmacol 31(7):861–872. https://doi.org/10.1177/0269881117695860
Vollrath-Smith FR, Shin R, Ikemoto S (2012) Synergistic interaction between baclofen administration into the median raphe nucleus and inconsequential visual stimuli on investigatory behavior of rats. Psychopharmacology 220(1):15–25. https://doi.org/10.1007/s00213-011-2450-x
Winterbauer NE, Balleine BW (2007) The influence of amphetamine on sensory and conditioned reinforcement: evidence for the re-selection hypothesis of dopamine function. Front Integr Neurosci 1:9. https://doi.org/10.3389/neuro.07.009.2007
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All experiments comply with the current laws of Canada.
Conflict of interest
P.B.S.C. is a member of the Center for Studies in Behavioral Neurobiology at Concordia University, Montreal. The authors have no financial relationship with the organizations that sponsored this research.
Electronic supplementary material
Supplementary Figure 1
Effect of food restriction on body weights in Experiment 3. Each rat was tested under ad libitum (AL) and food-restricted (FR) conditions, in a crossover design. Initially, 14 rats were tested under ad libitum feeding, and the remaining 16 rats were tested under food restriction. After a 10-day recovery period of ad libitum feeding and no testing, the feeding conditions were reversed. Y-axes show mean ± SEM (n = 14 and 16 rats per group). Note that most SEM bars are contained within the accompanying symbol. (JPEG 69 kb)
Rights and permissions
About this article
Cite this article
Wright, J.M., Ren, S., Constantin, A. et al. Enhancement of a visual reinforcer by d-amphetamine and nicotine in adult rats: relation to habituation and food restriction. Psychopharmacology 235, 803–814 (2018). https://doi.org/10.1007/s00213-017-4796-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-017-4796-1