Abstract
Rationale
Given that the pregnane neurosteroid, 5α-pregnan-3α-ol-20-one (3α,5α-THP), is increased following behavioral challenges (e.g., mating), and that there is behavioral-induced biosynthesis of 3α,5α-THP in midbrain and mesocorticolimbic structures, 3α,5α-THP likely has a role in homeostasis and motivated reproduction and reproduction-related behaviors (e.g., affect, affiliation). The role of pregnane xenobiotic receptor (PXR), involved in cholesterol metabolism, for these effects is of continued interest.
Objectives
We hypothesized that there would be differences in brain levels of 3α,5α-THP following varied behavioral experiences, an effect abrogated by knockdown of PXR in the midbrain.
Methods
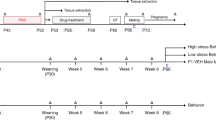
Proestrous rats were infused with PXR antisense oligonucleotides (AS-ODNs) or vehicle to the ventral tegmental area before different behavioral manipulations and assessments. Endpoints were expression levels of PXR in the midbrain, 3α,5α-THP, and ovarian steroids (estradiol, progesterone, dihydroprogesterone) in the midbrain, striatum, hippocampus, hypothalamus, prefrontal cortex, and plasma.
Results
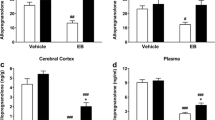
Across experiments, knocking down PXR reduced PXR expression and 3α,5α-THP levels in the midbrain and hippocampus. There were differences in terms of the behavioral manipulations, such that paced mating had the most robust effects to increase 3α,5α-THP levels and reduce open field exploration and social interaction. An additional question that was addressed is whether brain-derived neurotrophic factor (BDNF) is a downstream factor for regulating effects of behavioral-induced 3α,5α-THP biosynthesis. Rats infused with PXR AS-ODNs had lower levels of BDNF in the hippocampus.
Conclusion
Thus, PXR may be a regulator of mating-induced 3α,5α-THP formation and behavioral changes and neural plasticity, such as BDNF.






Similar content being viewed by others
References
Agís-Balboa RC, Pinna G, Pibiri F, Kadriu B, Costa E, Guidotti A (2007) Down-regulation of neurosteroid biosynthesis in corticolimbic circuits mediates social isolation-induced behavior in mice. Proc Natl Acad Sci U S A 104:18736–18741
Aguirre CC, Baudry M (2009) Progesterone reverses 17beta-estradiol-mediated neuroprotection and BDNF induction in cultured hippocampal slices. Eur J Neurosci 29:447–454
Aguirre C, Jayaraman A, Pike C, Baudry M (2010) Progesterone inhibits estrogen-mediated neuroprotection against excitotoxicity by down-regulating estrogen receptor-beta. J Neurochem 115:1277–1287
Akwa Y, Purdy RH, Koob GF, Britton KT (1999) The amygdala mediates the anxiolytic-like effect of the neurosteroid allopregnanolone in rat. Behav Brain Res 106:119–125
Angelucci F, Aloe L, Iannitelli A, Gruber SH, Mathé AA (2005) Effect of chronic olanzapine treatment on nerve growth factor and brain-derived neurotrophic factor in the rat brain. Eur Neuropsychopharmacol 15:311–317
Arzate DM, Portillo W, Corona R, Paredes RG (2012) Repeated paced mating promotes the arrival of more newborn neurons in the main and accessory olfactory bulbs of adult female rats. Neuroscience. doi:10.1016/j.neuroscience.2012.12.014
Barbaccia ML, Roscetti G, Trabucchi M, Mostallino MC, Concas A, Purdy RH, Biggio G (1996) Time-dependent changes in rat brain neuroactive steroid concentrations and GABAA receptor function after acute stress. Neuroendocrinology 63:166–172
Barde YA, Edgar D, Thoenen H (1982) Purification of a new neurotrophic factor from mammalian brain. EMBO J 1(5):49–53
Bath KG, Chuang J, Spencer-Segal JL, Amso D, Altemus M, McEwen BS, Lee FS (2012) Variant brain-derived neurotrophic factor (Valine66Methionine) polymorphism contributes to developmental and estrous stage-specific expression of anxiety-like behavior in female mice. Biol Psychiatry 72:499–504
Bauer B, Hartz AM, Fricker G, Miller DS (2004) Pregnane X receptor up-regulation of P-glycoprotein expression and transport function at the blood-brain barrier. Mol Pharmacol 66:413–419
Bauer B, Yang X, Hartz AM, Olson ER, Zhao R, Kalvass JC, Pollack GM, Miller DS (2006) In vivo activation of human pregnane X receptor tightens the blood-brain barrier to methadone through P-glycoprotein up-regulation. Mol Pharmacol 70:1212–9
Bimonte-Nelson HA, Granholm AC, Nelson ME, Moore AB (2008) Patterns of neurotrophin protein levels in male and female Fischer 344 rats from adulthood to senescence: how young is “young” and how old is “old”? Exp Aging Res 34:13–26
Bimonte-Nelson HA, Nelson ME, Granholm AC (2004) Progesterone counteracts estrogen-induced increases in neurotrophins in the aged female rat brain. Neuroreport 15:2659–2663
Bortolato M, Devoto P, Roncada P, Frau R, Flore G, Saba P, Pistritto G, Soggiu A, Pisanu S, Zappala A, Ristaldi MS, Tattoli M, Cuomo V, Marrosu F, Barbaccia ML (2011) Isolation rearing-induced reduction of brain 5α-reductase expression: relevance to dopaminergic impairments. Neuropharmacology 60:1301–1308
Brambilla F, Mellado C, Alciati A, Pisu MG, Purdy RH, Zanone S, Perini G, Serra M, Biggio G (2005) Plasma concentrations of anxiolytic neuroactive steroids in men with panic disorder. Psychiatry Res 135:185–190
Brunton PJ, Russell JA (2011) Neuroendocrine control of maternal stress responses and fetal programming by stress in pregnancy. Prog Neuropsychopharmacol Biol Psychiatry 35:1178–1191
Camacho FJ, García-Horsman P, Paredes RG (2009) Hormonal and testing conditions for the induction of conditioned place preference by pacedmating. Horm Behav 56:410–415
Causing CG, Gloster A, Aloyz R, Bamji SX, Chang E, Fawcett J, Kuchel G, Miller FD (1997) Synaptic innervation density is regulated by neuron-derived BDNF. Neuron 18:257–267
Corona R, Larriva-Sahd J, Paredes RG (2011) Paced-mating increases the number of adult new born cells in the internal cellular (granular) layer of the accessory olfactory bulb. PLoS One 6:e19380
Dong E, Matsumoto K, Uzunova V, Sugaya I, Takahata H, Nomura H, Watanabe H, Costa E, Guidotti A (2001) Brain 5alpha-dihydroprogesterone and allopregnanolone synthesis in a mouse model of protracted social isolation. Proc Natl Acad Sci U S A 98:2849–2854
Duman RS, Monteggia LM (2006) A neurotrophic model for stress-related mood disorders. Biol Psychiatry 59:1116–1127
Dussault I, Forman BM (2002) The nuclear receptor PXR: a master regulator of “homeland” defense. Crit Rev Eukaryot Gene Expr 12:53–64
Engler-Chiurazzi E, Tsang C, Nonnenmacher S, Liang WS, Corneveaux JJ, Prokai L, Huentelman MJ, Bimonte-Nelson HA (2011) Tonic premarin dose-dependently enhances memory, affects neurotrophin protein levels and alters gene expression in middle-aged rats. Neurobiol Aging 32:680–697
Francis GA, Fayard E, Picard F, Auwerx J (2002) Nuclear receptors and the control of metabolism. Annu Rev Physiol 65:261–311
Franklin TB, Perrot-Sinal TS (2006) Sex and ovarian steroids modulate brain-derived neurotrophic factor (BDNF) protein levels in rat hippocampus under stressful and non-stressful conditions. Psychoneuroendocrinology 31:38–48
Frye CA (2011) Novel substrates for, and sources of, progestogens for reproduction. J Neuroendocrinol 23:961–973
Frye CA, Erskine MS (1990) Influence of time of mating and paced copulation on induction of pseudopregnancy in cyclic female rats. J Reprod Fertil 90:375–385
Frye CA, Bayon LE, Pursnani NK, Purdy RH (1998) The neurosteroids, progesterone and 3alpha,5alpha-THP, enhance sexual motivation, receptivity, and proceptivity in female rats. Brain Res 808:72–83
Frye CA, Koonce CJ, Walf AA (2014) Role of pregnane xenobiotic receptor in the midbrain ventral tegmental area for estradiol- and 3α,5α-THP-facilitated lordosis of female rats. Psychopharmacology (in press)
Frye CA, Koonce CJ, Walf AA, Rusconi JC (2013a) Motivated behaviors and levels of 3α,5α-THP in the midbrain are attenuated by knocking down expression of pregnane xenobiotic receptor in the midbrain ventral tegmental area of proestrous rats. J Med Sex 10:1692–1706
Frye CA, Koonce CJ, Walf AA (2013b) Progesterone, compared to medroxyprogesterone acetate, to C57BL/6, but not 5α-reductase mutant, mice enhances object recognition and placement memory and is associated with higher BDNF levels in the hippocampus and cortex. Neurosci Lett 551:53–57
Frye CA, Rhodes ME (2005) Estrogen-priming can enhance progesterone's anti-seizure effects in part by increasing hippocampal levels of allopregnanolone. Pharmacol Biochem Behav 81:907–16
Frye CA, Walf AA (2004) Estrogen and/or progesterone administered systemically or to the amygdala can have anxiety-, fear-, and pain-reducing effects in ovariectomized rats. Behav Neurosci 118:306–313
Frye CA, Walf AA, Paris JJ (2010) Conjugated equine estrogen, with medroxyprogesterone acetate, enhances formation of 5α-reduced progestogens and reduces anxiety-like behavior of middle-aged rats. Behav Pharmacol 21:530–539
García-Horsman SP, Agmo A, Paredes RG (2008) Infusions of naloxone into the medial preoptic area, ventromedial nucleus of the hypothalamus, and amygdala block conditioned place preference induced by paced mating behavior. Horm Behav 54:709–716
Geick A, Eichelbaum M, Burk O (2001) Nuclear receptor response elements mediate induction of intestinal MDR1 by rifampin. J Biol Chem 276:14581–14587
Genazzani AR, Stomati M, Bernardi F, Luisi S, Casarosa E, Puccetti S, Genazzani AD, Palumbo M, Luisi M (2004) Conjugated equine estrogens reverse the effects of aging on central and peripheral allopregnanolone and beta-endorphin levels in female rats. Fertil Steril 81:757–766
Gibbs RB (1998) Levels of trkA and BDNF mRNA, but not NGF mRNA, fluctuate across the estrous cycle and increase in response to acute hormone replacement. Brain Res 787:259–268
González SL, Labombarda F, González Deniselle MC, Guennoun R, Schumacher M, De Nicola AF (2004) Progesterone up-regulates neuronal brain-derived neurotrophic factor expression in the injured spinal cord. Neuroscience 125:605–614
Gonzalez Deniselle MC, Garay L, Gonzalez S, Saravia F, Labombarda F, Guennoun R, Schumacher M, De Nicola AF (2007) Progesterone modulates brain-derived neurotrophic factor and choline acetyltransferase in degenerating Wobbler motoneurons. Exp Neurol 203:406–414
González-Flores O, Camacho FJ, Domínguez-Salazar E, Ramírez-Orduna JM, Beyer C, Paredes RG (2004) Progestins and place preference conditioning after paced mating. Horm Behav 46:151–157
Holder MK, Mong JA (2010) Methamphetamine enhances paced mating behaviors and neuroplasticity in the medial amygdala of female rats. Horm Behav 58:519–525
Jodhka PK, Kaur P, Underwood W, Lydon JP, Singh M (2009) The differences in neuroprotective efficacy of progesterone and medroxyprogesterone acetate correlate with their effects on brain-derived neurotrophic factor expression. Endocrinology 150:3162–3168
Klein AB, Williamson R, Santini MA, Clemmensen C, Ettrup A, Rios M, Knudsen GM, Aznar S (2011) Blood BDNF concentrations reflect brain-tissue BDNF levels across species. Int J Neuropsychopharmacol 14:347–353
Kliewer SA, Goodwin B, Willson TM (2002) The nuclear pregnane X receptor: a key regulator of xenobiotic metabolism. Endocr Rev 23:687–702
Lamba V, Yasuda K, Lamba JK, Assem M, Davila J, Strom S, Schuetz EG (2004) PXR (NR1I2): splice variants in human tissues, including brain, and dentification of neurosteroids and nicotine as PXR activators. Toxicol Appl Pharmacol 199:251–265.10
Lieberwirth C, Liu Y, Jia X, Wang Z (2012) Social isolation impairs adult neurogenesis in the limbic system and alters behaviors in female prairie voles. Horm Behav 62:357–366
Lieberwirth C, Wang Z (2012) The social environment and neurogenesis in the adult mammalian brain. Front Hum Neurosci 6:118
Llidó A, Mòdol L, Darbra S, Pallarès M (2013) Interaction between neonatal allopregnanolone administration and early maternal separation: effects on adolescent and adult behaviors in male rat. Horm Behav S0018-506X(13):00037-8
Lorenz L, Dang J, Misiak M, Tameh Abolfazl A, Beyer C, Kipp M (2009) Combined 17beta-oestradiol and progesterone treatment prevents neuronal cell injury in cortical but not midbrain neurones or neuroblastoma cells. J Neuroendocrinol 21:841–849
Ma X, Idle JR, Gonzalez FJ (2008) The pregnane X receptor: from bench to bedside. Expert Opin Drug Metab Toxicol 4:895–908
Marini S, Nannelli A, Sodini D, Dragoni S, Valoti M, Longo V, Gervasi PG (2007) Expression, microsomal and mitochondrial activities of cytochrome P450 enzymes in brain regions from control and phenobarbital-treated rabbits. Life Sci 80:910–917
Matsumoto K, Uzunova V, Pinna G, Taki K, Uzunov DP, Watanabe H, Mienville JM, Guidotti A, Costa E (1999) Permissive role of brain allopregnanolone content in the regulation of pentobarbital-induced righting reflex loss. Neuropharmacology 38:955–963
Mellon SH, Gong W, Schonemann MD (2008) Endogenous and synthetic neurosteroids in treatment of Niemann-Pick type C disease. Brain Res Rev 57:410–420
Meyer M, Gonzalez Deniselle MC, Gargiulo-Monachelli G, Garay LI, Schumacher M, Guennoun R, De Nicola AF (2012) Progesterone effects on neuronal brain-derived neurotrophic factor and glial cells during progression of Wobbler mouse neurodegeneration. Neuroscience 201:267–279
Mòdol L, Darbra S, Pallarès M (2011) Neurosteroids infusion into the CA1 hippocampal region on exploration, anxiety-like behaviour and aversive learning. Behav Brain Res 222:223–229
Naert G, Maurice T, Tapia-Arancibia L, Givalois L (2007) Neuroactive steroids modulate HPA axis activity and cerebral brain-derived neurotrophic factor (BDNF) protein levels in adult male rats. Psychoneuroendocrinology 32:1062–1078
National Research Council (US) Committee on Guidelines for the Use of Animals in Neuroscience and Behavioral Research (2003) Guidelines for the Care and Use of Mammals in Neuroscience and Behavioral Research. Washington (DC): National Academies Press (US)
Nelson M, Pinna G (2011) S-Norfluoxetine microinfused into the basolateral amygdala increases allopregnanolone levels and reduces aggression in socially isolated mice. Neuropharmacology 60:1154–1159
Nilsen J, Brinton RD (2002) Impact of progestins on estrogen-induced neuroprotection: synergy by progesterone and 19-norprogesterone and antagonism by medroxyprogesterone acetate. Endocrinology 143:205–212
Nin MS, Martinez LA, Pibiri F, Nelson M, Pinna G (2011) Neurosteroids reduce social isolation-induced behavioral deficits: a proposed link with neurosteroid-mediated upregulation of BDNF expression. Front Endocrinol 2:73
Nyuyki KD, Waldherr M, Baeuml S, Neumann ID (2011) Yes, I am ready now: differential effects of paced versus unpaced mating on anxiety and central oxytocin release in female rats. PLoS One 6:e23599
Ott M, Fricker G, Bauer B (2009) Pregnane X receptor (PXR) regulates P-glycoprotein at the blood–brain barrier: functional similarities between pig and human PXR. J Pharmacol Exp Ther 329:141–149
Paris JJ, Brunton PJ, Russell JA, Frye CA (2011) Immune stress in late pregnant rats decreases length of gestation and fecundity, and alters later cognitive and affective behaviour of surviving pre-adolescent offspring. Stress 14:652–664
Paxinos G, Watson C (1986) The rat brain. Academic Press, New York
Pibiri F, Nelson M, Guidotti A, Costa E, Pinna G (2008) Decreased corticolimbic allopregnanolone expression during social isolation enhances contextual fear: a model relevant for posttraumatic stress disorder. Proc Natl Acad Sci U S A 105:5567–5572
Pinna G, Agis-Balboa RC, Pibiri F, Nelson M, Guidotti A, Costa E (2008) Neurosteroid biosynthesis regulates sexually dimorphic fear and aggressive behavior in mice. Neurochem Res 33:1990–2007
Pinna G, Rasmusson AM (2012) Up-regulation of neurosteroid biosynthesis as a pharmacological strategy to improve behavioural deficits in a putative mouse model of post-traumatic stress disorder. J Neuroendocrinol 24:102–116
Pluchino N, Russo M, Santoro AN, Litta P, Cela V, Genazzani AR (2013) Steroids hormones and BDNF. Neuroscience 3(239):271–279
Porcu P, Mostallino MC, Sogliano C, Santoru F, Berretti R, Concas A (2012) Long-term administration with levonorgestrel decreases allopregnanolone levels and alters GABA(A) receptor subunit expression and anxiety-like behavior. Pharmacol Biochem Behav 102:366–372
Pruginin-Bluger M, Shelton DL, Kalcheim C (1997) A paracrine effect for neuron-derived BDNF in development of dorsal root ganglia: stimulation of Schwann cell myelin protein expression by glial cells. Mech Dev 61:99–111
Purdy RH, Morrow AL, Moore PH Jr, Paul SM (1991) Stress-induced elevations of gamma-aminobutyric acid type A receptor-active steroids in the rat brain. Proc Natl Acad Sci U S A 88(10):4553–4557
Santoru F, Berretti R, Locci A, Porcu P, Concas A (2014) Decreased allopregnanolone induced by hormonal contraceptives is associated with a reduction in social behavior and sexual motivation in female rats. Psyhcopharmacology, under consideration
Serra M, Pisu MG, Floris I, Floris S, Cannas E, Mossa A, Trapani G, Latrofa A, Purdy RH, Biggio G (2004) Social isolation increases the response of peripheral benzodiazepine receptors in the rat. Neurochem Int 45:141–148
Singh M, Su C (2013) Progesterone, brain-derived neurotrophic factor and neuroprotection. Neuroscience 239:84–91
Vallée M, Rivera JD, Koob GF, Purdy RH, Fitzgerald RL (2000) Quantification of neurosteroids in rat plasma and brain following swim stress and allopregnanolone administration using negative chemical ionization gas chromatography/mass spectrometry. Anal Biochem 287:153–166
Xu DX, Wei W, Sun MF, Wei LZ, Wang JP (2005) Melatonin attenuates lipopolysaccharide-induced down-regulation of pregnane X receptor and its target gene CYP3A in mouse liver. J Pineal Res 38:27–34
Yao J, Chen S, Cadenas E, Brinton RD (2011) Estrogen protection against mitochondrial toxin-induced cell death in hippocampal neurons: antagonism by progesterone. Brain Res 16(1379):2–10
Zimmerberg B, Blaskey LG (1998) Prenatal stress effects are partially ameliorated by prenatal administration of the neurosteroidallopregnanolone. Pharmacol Biochem Behav 59:819–827
Zhang B, Xie W, Krasowski MD (2008) PXR: a xenobiotic receptor of diverse function implicated in pharmacogenetics. Pharmacogenomics 9:1695–1709
Acknowledgments
Research herein was supported by grants from the National Institute of Mental Health (MH0676980; RMH067698B). Technical assistance, provided by Drs. Paris and Rusconi, and Julianne Power, Anthony Santarelli, Eric Selke, Jennifer Torgersen, and Zhenhong Zhao, is greatly appreciated.
Conflict of interest
All authors report that they have no conflicts of interest (financial or otherwise) that would bias them to the outcome of these experiments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Frye, C.A., Koonce, C.J. & Walf, A.A. Involvement of pregnane xenobiotic receptor in mating-induced allopregnanolone formation in the midbrain and hippocampus and brain-derived neurotrophic factor in the hippocampus among female rats. Psychopharmacology 231, 3375–3390 (2014). https://doi.org/10.1007/s00213-014-3569-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-014-3569-3