Abstract
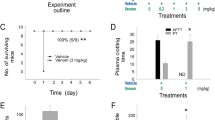
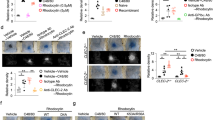
Venomous snakebites cause clinical manifestations that range from local to systemic and are considered a significant global health challenge. Persistent or refractory thrombocytopenia has been frequently reported in snakebite patients, especially in cases caused by viperidae snakes. Viper envenomation-induced thrombocytopenia may persist in the absence of significant consumption coagulopathy even after the antivenom treatment, yet the mechanism remains largely unknown. Our study aims to investigate the mechanism and discover novel therapeutic targets for coagulopathy-independent thrombocytopenia caused by viper envenomation. Here we found that patients bitten by Protobothrops mucrosquamatus and Trimeresurus stejnegeri, rather than Naja. atra may develop antivenom-resistant and coagulopathy-independent thrombocytopenia. Crude venoms and the derived C-type lectin-like proteins from these vipers significantly increased platelet surface expression of neuraminidase and platelet desialylation, therefore led to platelet ingestion by both macrophages and hepatocytes in vitro, and drastically decreased peripheral platelet counts in vivo. Our study is the first to demonstrate that desialylation-mediated platelet clearance is a novel mechanism of viper envenomation-induced refractory thrombocytopenia and C-type lectin-like proteins derived from the viper venoms contribute to snake venom-induced thrombocytopenia. The results of this study suggest the inhibition of platelet desialylation as a novel therapeutic strategy against viper venom-induced refractory thrombocytopenia.






Similar content being viewed by others
References
Alirol E, Sharma SK, Bawaskar HS, Kuch U, Chappuis F (2010) Snake bite in South Asia: a review. Plos Negl Trop Dis 4(1):e603. https://doi.org/10.1371/journal.pntd.0000603
Aslam R, Speck ER, Kim M et al (2006) Platelet Toll-like receptor expression modulates lipopolysaccharide-induced thrombocytopenia and tumor necrosis factor-α production in vivo. Blood 107(2):637–641
Bolt HM (2021) New aspects in snake venom toxicology. Arch Toxicol. https://doi.org/10.1007/s00204-021-03066-4
Chen YW, Chen MH, Chen YC et al (2009) Differences in clinical profiles of patients with Protobothrops mucrosquamatus and Viridovipera stejnegeri envenoming in Taiwan. Am J Trop Med Hyg 80(1):28–32
Chen ZM, Wu JB, Zhang Y et al (2011) A novel platelet glycoprotein Ib-binding protein with human platelet aggregation-inhibiting activity from Trimeresurus jerdonii venom. Toxicon 57(5):672–679. https://doi.org/10.1016/j.toxicon.2011.01.010
Cheng C-L, Mao Y-C, Liu P-Y, Chiang L-C, Liao S-C, Yang C-C (2017) Deinagkistrodon acutus envenomation: a report of three cases. J Venom Anim Toxins Incl Trop Dis 23(1):20
Clemetson KJ (2010) Snaclecs (snake C-type lectins) that inhibit or activate platelets by binding to receptors. Toxicon 56(7):1236–1246. https://doi.org/10.1016/j.toxicon.2010.03.011
de França FS, Gabrili JJM, Mathieu L, Burgher F, Blomet J, Tambourgi DV (2021) Bothrops lanceolatus snake (Fer-de-lance) venom triggers inflammatory mediators’ storm in human blood. Arch Toxicol 95(3):1129–1138
Deppermann C, Kratofil RM, Peiseler M, et al. (2020) Macrophage galactose lectin is critical for Kupffer cells to clear aged platelets. J Exp Med 217(4)
Eble JA (2019) Structurally Robust and Functionally Highly Versatile—C-Type Lectin (-Related) Proteins in Snake Venoms. Toxins 11(3):136
Fang Y, He X, Zhang P et al (2019) In Vitro and In Vivo Antimalarial Activity of LZ1, a Peptide Derived from Snake Cathelicidin. Toxins 11(7) doi:https://doi.org/10.3390/toxins11070379
Franchini M, Veneri D, Lippi G (2017) Thrombocytopenia and infections. Expert Rev Hematol 10(1):99–106
Gold BS, Barish RA, Rudman MS (2004) Refractory thrombocytopenia despite treatment for rattlesnake envenomation. N Engl J Med 350(18):1912–1913; discussion 1912–1913. https://doi.org/10.1056/NEJM200404293501824
Grozovsky R, Begonja AJ, Liu K et al (2015) The Ashwell-Morell receptor regulates hepatic thrombopoietin production via JAK2-STAT3 signaling. Nat Med 21(1):47–54
Gutierrez JM, Calvete JJ, Habib AG, Harrison RA, Williams DJ, Warrell DA (2017) Snakebite envenoming. Nat Rev Dis Primers. https://doi.org/10.1038/Nrdp.2017.63
Gutiérrez JM, Albulescu L-O, Clare RH et al (2021) The Search for Natural and Synthetic Inhibitors That Would Complement Antivenoms as Therapeutics for Snakebite Envenoming. Toxins 13(7):451
Heemskerk J, Mattheij N, Cosemans J (2013) Platelet-based coagulation: different populations, different functions. J Thromb Haemost 11(1):2–16
Herrera C, Rucavado A, Warrell DA, Gutiérrez JM (2013) Systemic effects induced by the venom of the snake Bothrops caribbaeus in a murine model. Toxicon 63:19–31
Hoffmeister KM, Felbinger TW, Falet H et al (2003a) The clearance mechanism of chilled blood platelets. Cell 112(1):87–97. https://doi.org/10.1016/s0092-8674(02)01253-9
Hoffmeister KM, Josefsson EC, Isaac NA, Clausen H, Hartwig JH, Stossel TP (2003b) Glycosylation restores survival of chilled blood platelets. Science 301(5639):1531–1534. https://doi.org/10.1126/science.1085322
Isbister GK (2010) Snakebite Doesn’t Cause Disseminated Intravascular Coagulation: Coagulopathy and Thrombotic Microangiopathy in Snake Envenoming. Semin Thromb Hemost 36(4):444–451. https://doi.org/10.1055/s-0030-1254053
Jansen A, Josefsson EC, Rumjantseva V et al (2012) Desialylation accelerates platelet clearance after refrigeration and initiates GPIbα metalloproteinase-mediated cleavage in mice. Blood 119(5):1263–1273
Jiang Y, Li Y, Lee W et al (2011) Venom gland transcriptomes of two elapid snakes (Bungarus multicinctus and Naja atra) and evolution of toxin genes. BMC Genomics 12:1. https://doi.org/10.1186/1471-2164-12-1
Josefsson EC, Gebhard HH, Stossel TP, Hartwig JH, Hoffmeister KM (2005) The macrophage αMβ2 integrin αM lectin domain mediates the phagocytosis of chilled platelets. J Biol Chem 280(18):18025–18032
Karakas D, Xu M, Ni H (2021) GPIbα is the driving force of hepatic thrombopoietin generation. Research and Practice in Thrombosis and Haemostasis 5(4):e12506
Latinovic Z, Leonardi A, Sribar J et al (2016) Venomics of Vipera berus berus to explain differences in pathology elicited by Vipera ammodytes ammodytes envenomation: Therapeutic implications. J Proteomics 146:34–47. https://doi.org/10.1016/j.jprot.2016.06.020
Lavonas EJ, Schaeffer TH, Kokko J, Mlynarchek SL, Bogdan GM (2009) Crotaline Fab antivenom appears to be effective in cases of severe North American pit viper envenomation: an integrative review. BMC Emerg Med 9:13. https://doi.org/10.1186/1471-227X-9-13
Lazarovici M, Lelkes, (2019) From Snake Venom’s Disintegrins and C-Type Lectins to Anti-Platelet Drugs. Toxins 11(5):303
Lei X, Reheman A, Hou Y et al (2014) Anfibatide, a novel GPIb complex antagonist, inhibits platelet adhesion and thrombus formation in vitro and in vivo in murine models of thrombosis. Thromb Haemost 112(02):279–289
Li J, Callum JL, Lin Y, Zhou Y, Zhu G, Ni H (2014) Severe platelet desialylation in a patient with glycoprotein Ib/IX antibody-mediated immune thrombocytopenia and fatal pulmonary hemorrhage. Haematologica 99(4):e61–e63. https://doi.org/10.3324/haematol.2013.102897
Li J, Van Der Wal DE, Zhu G et al (2015) Desialylation is a mechanism of Fc-independent platelet clearance and a therapeutic target in immune thrombocytopenia. Nat Commun 6:7737
Li M-f, Li X-l, Fan K-l et al (2017) Platelet desialylation is a novel mechanism and a therapeutic target in thrombocytopenia during sepsis: an open-label, multicenter, randomized controlled trial. J Hematol Oncol 10(1):1–10
Li J, Sullivan JA, Ni H (2018) Pathophysiology of immune thrombocytopenia. Curr Opin Hematol 25(5):373–381
Li C, Li J, Ni H (2020) Crosstalk Between Platelets and Microbial Pathogens. Frontiers in Immunology 11
Li BX, Dai X, Xu XR et al (2021) In vitro assessment and phase I randomized clinical trial of anfibatide a snake venom derived anti-thrombotic agent targeting human platelet GPIbα. Sci Rep 11(1):1–17
Lin C-C, Chen Y-C, Goh ZNL et al (2020) Wound infections of snakebites from the venomous Protobothrops mucrosquamatus and Viridovipera stejnegeri in taiwan: Bacteriology, antibiotic susceptibility, and predicting the need for antibiotics—A bite study. Toxins 12(9):575
Liu C-C, Lin C-C, Hsiao Y-C, Wang P-J, Yu J-S (2018) Proteomic characterization of six Taiwanese snake venoms: Identification of species-specific proteins and development of a SISCAPA-MRM assay for cobra venom factors. J Proteomics 187:59–68
Long C, Liu M, Tian H, et al. (2020) Potential Role of Platelet-Activating C-Type Lectin-Like Proteins in Viper Envenomation Induced Thrombotic Microangiopathy Symptom. Toxins (Basel) 12(12) doi:https://doi.org/10.3390/toxins12120749
Mackeigan DT, Ni T, Shen C, Stratton TW, Ni H (2020) Updated Understanding of Platelets in Thrombosis and Hemostasis: The Roles of Integrin PSI Domains and their Potential as Therapeutic Targets.
Maduwage K, Isbister GK (2014) Current Treatment for Venom-Induced Consumption Coagulopathy Resulting from Snakebite. Plos Neglected Tropical Diseases 8(10) doi:ARTN e322010.1371/journal.pntd.0003220
McKenzie CG, Guo L, Freedman J, Semple JW (2013) Cellular immune dysfunction in immune thrombocytopenia (ITP). Br J Haematol 163(1):10–23
Monzon Manzano E, Alvarez Roman MT, Justo Sanz R et al (2020) Platelet and immune characteristics of immune thrombocytopaenia patients non-responsive to therapy reveal severe immune dysregulation. Br J Haematol 189(5):943–953. https://doi.org/10.1111/bjh.16459
Ni H, Freedman J (2003) Platelets in hemostasis and thrombosis: role of integrins and their ligands. Transfus Apheres Sci 28(3):257–264
Norris PA, Segel GB, Burack WR, et al. (2021) FcγRI and FcγRIII on splenic macrophages mediate phagocytosis of anti-glycoprotein IIb/IIIa autoantibody-opsonized platelets in immune thrombocytopenia. haematologica 106(1)
Odeleye AA, Presley AE, Passwater ME, Mintz PD (2004) Report of two cases: Rattlesnake venom-induced thrombocytopenia. Ann Clin Lab Sci 34(4):467–470
Offerman SR, Barry JD, Schneir A, Clark RF (2003) Biphasic rattlesnake venom-induced thrombocytopenia. J Emerg Med 24(3):289–293. https://doi.org/10.1016/s0736-4679(02)00763-1
Ogawa T, Chijiwa T, Oda-Ueda N, Ohno M (2005) Molecular diversity and accelerated evolution of C-type lectin-like proteins from snake venom. Toxicon 45(1):1–14. https://doi.org/10.1016/j.toxicon.2004.07.028
Peng ML, Lu WQ, Beviglia L, Niewiarowski S, Kirby EP (1993) Echicetin - a Snake-Venom Protein That Inhibits Binding of Vonwillebrand-Factor and Alboaggregins to Platelet Glycoprotein-Ib. Blood 81(9):2321–2328
Qiu J, Liu X, Li X et al (2016) CD8+ T cells induce platelet clearance in the liver via platelet desialylation in immune thrombocytopenia. Sci Rep 6(1):1–12
Quach ME, Chen W, Li R (2018) Mechanisms of platelet clearance and translation to improve platelet storage. Blood 131(14):1512–1521. https://doi.org/10.1182/blood-2017-08-743229
Reheman A, Yang H, Zhu G et al (2009) Plasma fibronectin depletion enhances platelet aggregation and thrombus formation in mice lacking fibrinogen and von Willebrand factor Blood. J Am Soc Hematol 113(8):1809–1817
Revilla N, Corral J, Miñano A, et al. (2018) Multirefractory primary immune thrombocytopenia; targeting the decreased sialic acid content. Platelets:1–9
Rucavado A, Soto M, Kamiguti AS et al (2001) Characterization of aspercetin, a platelet aggregating component from the venom of the snake Bothrops asper which induces thrombocytopenia and potentiates metalloproteinase-induced hemorrhage. Thromb Haemost 85(4):710–715
Rumjantseva V, Grewal PK, Wandall HH et al (2009) Dual roles for hepatic lectin receptors in the clearance of chilled platelets. Nat Med 15(11):1273–1280
Sanhajariya S, Isbister GK, Duffull SB (2020) The Influence of the Different Disposition Characteristics of Snake Toxins on the Pharmacokinetics of Snake Venom. Toxins 12(3):188
Shao L, Wu Y, Zhou H et al (2015) Successful treatment with oseltamivir phosphate in a patient with chronic immune thrombocytopenia positive for anti-GPIb/IX autoantibody. Platelets 26(5):495–497
Shen C, Liu M, Tian H et al (2020a) Conformation-Specific Blockade of alphaIIbbeta3 by a Non-RGD Peptide to Inhibit Platelet Activation without Causing Significant Bleeding and Thrombocytopenia. Thromb Haemost 120(10):1432–1441. https://doi.org/10.1055/s-0040-1714215
Shen C, Liu M, Xu R et al (2020b) The 14–3-3zeta-c-Src-integrin-beta3 complex is vital for platelet activation. Blood 136(8):974–988. https://doi.org/10.1182/blood.2019002314
Tao L, Zeng Q, Li J et al (2017) Platelet desialylation correlates with efficacy of first-line therapies for immune thrombocytopenia. J Hematol Oncol 10(1):1–4
Tasoulis T, Isbister GK (2017) A Review and Database of Snake Venom Proteomes. Toxins 9(9) doi:Artn 29010.3390/Toxins9090290
Tian H, Liu M, Li J et al (2020) Snake C-Type Lectins Potentially Contribute to the Prey Immobilization in Protobothrops mucrosquamatus and Trimeresurus stejnegeri Venoms. Toxins 12(2):105
Villalta M, Pla D, Yang SL et al (2012) Snake venomics and antivenomics of Protobothrops mucrosquamatus and Viridovipera stejnegeri from Taiwan: keys to understand the variable immune response in horses. J Proteomics 75(18):5628–5645. https://doi.org/10.1016/j.jprot.2012.08.008
Waheed H, Moin F, S, I Choudhary M, (2017) Snake venom: from deadly toxins to life-saving therapeutics. Curr Med Chem 24(17):1874–1891
Wang Y, Reheman A, Spring CM et al (2014) Plasma fibronectin supports hemostasis and regulates thrombosis. J Clin Investig 124(10):4281–4293
Warrell DA (2010) Snake bite. Lancet 375(9708):77–88. https://doi.org/10.1016/S0140-6736(09)61754-2
Webster ML, Sayeh E, Crow M et al (2006) Relative efficacy of intravenous immunoglobulin G in ameliorating thrombocytopenia induced by antiplatelet GPIIbIIIa versus GPIbα antibodies. Blood 108(3):943–946
Wedasingha S, Isbister G, Silva A (2020) Bedside Coagulation Tests in Diagnosing Venom-Induced Consumption Coagulopathy in Snakebite. Toxins (Basel) 12(9). https://doi.org/10.3390/toxins12090583
Xu XR, Zhang D, Oswald BE et al (2016) Platelets are versatile cells: New discoveries in hemostasis, thrombosis, immune responses, tumor metastasis and beyond. Crit Rev Clin Lab Sci 53(6):409–430
Xu M, Li J, Neves MAD et al (2018) GPIbα is required for platelet-mediated hepatic thrombopoietin generation. Blood 132(6):622–634
Yamashita KM, Alves AF, Barbaro KC, Santoro ML (2014) Bothrops jararaca venom metalloproteinases are essential for coagulopathy and increase plasma tissue factor levels during envenomation. PLoS Negl Trop Dis 8(5):e2814
Yang H, Reheman A, Chen P et al (2006) Fibrinogen and von Willebrand factor-independent platelet aggregation in vitro and in vivo. J Thromb Haemost 4(10):2230–2237
Zeng Q, Zhu L, Tao L et al (2012) Relative efficacy of steroid therapy in immune thrombocytopenia mediated by anti-platelet GPIIbIIIa versus GPIbα antibodies. Am J Hematol 87(2):206–208
Acknowledgements
This work was supported by grants from the Heart and Stroke Foundation of Ontario, Canada, the Canadian Institutes of Health Research Foundation [389035] and Health Commission of Guangxi Zhuang autonomous region [Z20200287].
Author information
Authors and Affiliations
Contributions
C.S. and M.L. performed experiments, created figures, analyzed data. J.L. and Y.D. provided medical records of snakebite patients. D.T.M, Z.C., P.C., D.K., J.L., P.A.A.N., C.L. and R.L. provided valuable suggestions and contributed to paper writing. C.S. wrote the manuscript. H.N. supervised the research and wrote the paper.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflict of interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shen, C., Liu, M., Mackeigan, D.T. et al. Viper venoms drive the macrophages and hepatocytes to sequester and clear platelets: novel mechanism and therapeutic strategy for venom-induced thrombocytopenia. Arch Toxicol 95, 3589–3599 (2021). https://doi.org/10.1007/s00204-021-03154-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-021-03154-5