Abstract
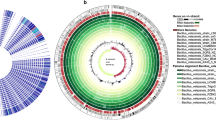
A new strain of Bacillus velezensis NDB was isolated from Xiangshan Harbor and antibacterial test revealed antibacterial activity of this strain against 12 major pathogenic bacteria. The whole genome of the bacterium was sequenced and found to consist of a 4,214,838 bp circular chromosome and a 7410 bp circular plasmid. Furthermore, it was predicted by AntiSMASH and BAGEL4 to have 12 clusters of secondary metabolism genes for the synthesis of the inhibitors, fengycin, bacillomycin, macrolactin H, bacillaene, and difficidin, and there were also five clusters encoding potentially novel antimicrobial substances, as well as three bacteriocin biosynthesis gene clusters of amylocyclicin, ComX1, and LCI. qRT-PCR revealed significant up-regulation of antimicrobial secondary metabolite synthesis genes after 24 h of antagonism with pathogenic bacteria. Furthermore, MALDI-TOF mass spectrometry revealed that it can secrete surfactin non-ribosomal peptide synthase and polyketide synthase to exert antibacterial effects. GC-MS was used to analyze methanol extract of B. velezensis NDB, a total of 68 compounds were identified and these metabolites include 16 amino acids, 17 acids, 3 amines, 11 sugars, 11 alcohols, 1 ester, and 9 other compounds which can inhibit pathogenic bacteria by initiating the antibiotic secretion pathway. A comparative genomic analysis of gene families showed that the specificity of B. velezensis NDB was mainly reflected in environmental adaptability. Overall, this research on B. velezensis NDB provides the basis for elucidating its biocontrol effect and promotes its future application as a probiotic.










Similar content being viewed by others
Data availability
The sequences have been deposited sited in the NCBI Sequence Read Archive Database under the accession number CP135184-CP135185. The authors declare that [the/all other] data supporting the findings of this study are available within the paper (and its supplementary information files). The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Al-Ali A, Deravel J, Krier F, Béchet M, Jacques P (2017) Biofilm formation is determinant in tomato rhizosphere colonization by Bacillus velezensis FZB42. Environ Sci Pollut Res Int 25:1–11. https://doi.org/10.1007/s11356-017-0469-1
Alexandra S, Ricardo SM, Máximo G, Michael A (2021) The role of surfactin production by Bacillus velezensis on colonization, biofilm formation on tomato root and leaf surfaces and subsequent protection (ISR) against botrytis cinerea. Microorganisms 9(11):2251–2251. https://doi.org/10.3390/microorganisms9112251
Arguelles Arias A, Ongena M, Devreese B, Terrak M, Joris B, Fickers P (2013) Characterization of amylolysin, a novel lantibiotic from Bacillus amyloliquefaciens GA1. PLoS ONE 8:e83037. https://doi.org/10.1371/journal.pone.0083037
Baharudin M, Ngalimat M, Mohd S, Balia Y, Karim M, Baharum SN, Sabri S (2021) Antimicrobial activities of Bacillus velezensis strains isolated from stingless bee products against methicillin-resistant Staphylococcus aureus. PLoS ONE 16(5):e0251514. https://doi.org/10.1371/journal.pone.0251514
Blumenscheit C, Jähne J, Schneider A, Blom J, Schweder T, Lasch P, Borriss R (2022) Genome sequence data of Bacillus velezensis BP12A and BT24. Data Brief 41:107978. https://doi.org/10.1016/j.dib.2022.107978
Cao L, Do T, Jame LA (2021) Mechanisms of action of ribosomally synthesized and posttranslationally modified peptides (RiPPs). J Indus Microbiol Biotechnol 48(3–4):kuab005. https://doi.org/10.1093/jimb/kuab005
Chen XH, Scholz R, Borriss M, Junge H, Gel G, Kunz S, Borriss R (2009) Difficidin and bacilysin produced by plant-associated Bacillus amyloliquefaciens are efficient in controlling fire blight disease. J Biotechnol 140:38–44. https://doi.org/10.1016/j.jbiotec.2008.10.015
Chen WC, Wei RS, Yu H (2015) Applications of a lipopeptide biosurfactant, surfactin, produced by microorganisms. Biochem Eng J 103:158–169. https://doi.org/10.1016/j.bej.2015.07.009
Chen X, Yang F, Bai C, Shi Q, Hu S, Tang X, Peng L, Ding H (2022) Bacillus velezensis strain GUMT319 reshapes soil microbiome biodiversity and increases grape yields. Biology 11(10):1486–1486. https://doi.org/10.3390/biology11101486
Chen X, Zhao H, Meng F, Zhou L, Lu Z, Lu Y (2023) Surfactin alleviated hyperglycaemia in mice with type 2 diabetes induced by a high-fat diet and streptozotocin. Food Sci Human Wellness 12(6):16. https://doi.org/10.1016/j.fshw.2023.03.012
Conesa A, Götz S (2008) Blast2GO: a comprehensive suite for functional analysis in plant genomics. Int J Plant Genom 2008:619832. https://doi.org/10.1155/2008/619832
Delcher AL, Harmon D, Kasif S, White O, Salzberg SL (1999) Improved microbial gene identification with Glimmer. Nucleic Acids Res 27:4636–4641. https://doi.org/10.1093/nar/27.23.4636
Drula E, Garron ML, Dogan S, Lombard V, Henrissat B, Terrapon N (2022) The carbohydrate-active enzyme database: functions and literature. Nucleic Acids Res 50:D571–D577. https://doi.org/10.1093/nar/gkab1045
Esmaeilishirazifard E, Dariush A, Moschos SA, Keshavarz T (2018) A novel antifungal property for the Bacillus licheniformis ComX pheromone and its possible role in inter-kingdom cross-talk. Appl Microbiol Biotechnol 102(12):5197–5208. https://doi.org/10.1007/s00253-018-9004-7
Gong WB, Chen ZL, Xia B, Lu G (2011) Solution structure of LCI, a novel antimicrobial peptide from Bacillus subtilis. Biochemistry 50:3621. https://doi.org/10.1021/bi200123w
Gowda MT, Meena BR, Krishnan N, Manjunath M, Sellaperumal C, Rai AB, Singh A, Manimurugan C, Patil J, Pandey KK (2022) Antimicrobial peptides producing native Bacillus spp. for the management of root-knot nematode Meloidogyne incognita infecting okra (Abelmoschus esculentus L. Moench). Biol Control Theor Appl Pest Manage 171:104951. https://doi.org/10.1016/j.biocontrol.2022.104951
Grant JR, Stothard P (2008) The CGView server: a comparative genomics tool for circular genomes. Nucleic Acids Res 36:W181-184. https://doi.org/10.1093/nar/gkn179
James G, Das BC, Jose S (2021) Bacillus as an aquaculture friendly microbe. Aquaculture Int 29:323–353. https://doi.org/10.1007/s10499-020-00630-0
Kresna IDM, Wuisan ZG, Pohl JM, Mettal U, Linares Otoya V, Gand M, Marner M, Otoya LL, Bohringer N, Vilcinskas A (2022) Genome-mining-guided discovery and characterization of the PKS-NRPS-hybrid polyoxyperuin produced by a marine-derived Streptomycete. J Nat Prod 85(4):888–898. https://doi.org/10.1021/acs.jnatprod.1c01018
Lasch C, Stierhof M, Estévez MR, Myronovskyi M, Luzhetskyy A (2020) Dudomycins: new secondary metabolites produced after heterologous expression of an Nrps cluster from streptomyces albus ssp. Chlorinus. https://doi.org/10.3390/microorganisms8111800
Li YL, Gu YL, Li J (2015) Biocontrol agent Bacillus amyloliquefaciens LJ02 induces systemic resistance against cucurbits powdery mildew. Front Microbiol 6:883. https://doi.org/10.3389/fmicb.2015.00883
Li J, Wu ZB, Zhang Z, Zha JW, Ling F (2019) Effects of potential probiotic Bacillus velezensis K2 on growth, immunity and resistance to Vibrio harveyi infection of hybrid grouper (Epinephelus lanceolatus × E. fuscoguttatus). Fish Shellfish Immunol 93:1047–1055. https://doi.org/10.1016/j.fsi.2019.08.047
Liu G, Kong Y, Fan Y, Geng C, Peng D, Sun M (2017) Whole-genome sequencing of Bacillus velezensis LS69, a strain with a broad inhibitory spectrum against pathogenic bacteria. J Biotechnol 249:20–24. https://doi.org/10.1016/j.jbiotec.2017.03.018
Liu Y, Lu J, Sun J, Lu F, Bie X, Lu Z (2019) Membrane disruption and DNA binding of Fusarium graminearum cell induced by C16-Fengycin a produced by Bacillus amyloliquefaciens. Food Control 102:206–213. https://doi.org/10.1016/j.foodcont.2019.03.031
Lu J, Zhao Y, Hu R, Cheng Y, Qin J, Yang J, Fang Y, Lu M, Wang S (2022) Screening and characteristics of marine Bacillus velezensis Z-1 protease and its application of enzymatic hydrolysis of mussels to prepare antioxidant active substances. Molecules 27(19):6570. https://doi.org/10.3390/molecules27196570
Ming T, Han J, Li Y, Lu C, Qiu D (2018) A metabolomics and proteomics study of the Lactobacillus plantarum in the grass carp fermentation. BMC Microbiol 6(27):344–350. https://doi.org/10.1186/s12866-018-1354-x
Moriya YI, Okuda M, Shujiro Y, Akiyasu C, Minoru (2007) KAAS: an automatic genome annotation and pathway reconstruction server. Nucleic Acids Res 35:W182–W185. https://doi.org/10.1093/nar/gkm321
Oxford J, Kozlov R (2013) Antibiotic resistance - a call to arms for primary healthcare providers. Int J Clin Pract 67:1–3. https://doi.org/10.1038/s41591-020-01201-9
Paulo BSS, de Oliveira R, Luciana G (2019) Recent advances in combinatorial biosynthesis of polyketides: perspectives and challenges. Quimica Nova 42:71–83. https://doi.org/10.2147/DDDT.S63023
Powell S, Forslund K, Szklarczyk D, Trachana K, Roth A, Huerta-Cepas J, Gabaldón T, Rattei T, Creevey C, Kuhn M, Jensen LJ, von Mering C, Bork P (2014) eggNOG v4.0: nested orthology inference across 3686 organisms. Nucleic Acids Res 42:D231-239. https://doi.org/10.1093/nar/gkt1253
Puan SL, Erriah P, Baharudin M, Yahaya N, Kamil M, Sabri S (2023) Antimicrobial peptides from Bacillus spp. and strategies to enhance their yield. Appl Microbiol Biotechnol 107(18):5569–5593. https://doi.org/10.1007/s00253-023-12651-9
Rabbee MFB, Kwang-Hyun, (2020) Antimicrobial activities of lipopeptides and polyketides of Bacillus velezensis for agricultural applications. Molecules 25:4973. https://doi.org/10.3390/molecules25214973
Rabbee M, Ali M, Choi J, Hwang B, Jeong S, Baek KH (2019) Bacillus velezensis: a valuable member of bioactive molecules within plant microbiomes. Molecules 24(6):1046. https://doi.org/10.3390/molecules24061046
Ranjan A, Rajput VD, Prazdnova EV, Gurnani M, Bhardwaj P, Sharma S, Sushkova S, Mandzhieva SS, Minkina T, Sudan J, Zargar SM, Chauhan A, Jindal T (2023) Nature’s antimicrobial arsenal: non-ribosomal peptides from PGPB for plant pathogen biocontrol. Fermentation 9(7):597. https://doi.org/10.3390/microorganisms9030614
Ruiz-García C, Béjar V, Martínez-Checa F, Llamas I, Quesada E (2005) Bacillus velezensis sp. nov., a surfactant-producing bacterium isolated from the river Vélez in Málaga, southern Spain. Int J Syst Evol Microbiol 55:191–195. https://doi.org/10.1099/ijs.0.63310-0
Ruiz-Sanchez MN, Mejía B, Alejandra SD (2016) Antifungal activity and molecular identification of native strains of bacillus subtilis. Agrociencia 50:133–148. https://doi.org/10.1111/efp.12309
Samia SA, Marwa SK, Matia S, Sara AA, Soraya AS (2020) Novel research on nanocellulose production by a marine Bacillus velezensis strain SMR: a comparative study. Sci Rep 10(1):10–13. https://doi.org/10.1038/s41598-020-70857-7
Sam-On MFS, Mustafa S, Mohd Hashim A, Yusof MT, Zulkifly S, Malek AZA, Roslan MAH, Mohd Asrore MS (2023) Mining the genome of Bacillus velezensis FS26 for probiotic markers and secondary metabolites with antimicrobial properties against aquaculture pathogens. Microb Pathog 181:106161. https://doi.org/10.1016/j.micpath.2023.106161
Senchenkov VY, Lyakhovchenko NS, Nikishin IA, Myagkov DA, Chepurina AA, Polivtseva VN, Abashina TN, Delegan YA, Nikulicheva TB, Nikulin IS, Bogun AG, Solomentsev VI, Solyanikova IP (2023) Whole-genome sequencing and biotechnological potential assessment of two bacterial strains isolated from poultry farms in Belgorod, Russia. Microorganisms 11(9):2235. https://doi.org/10.3390/microorganisms11092235
Seng KL, CarolinaM AU, Rikky W, Caroline C, Anthony W, Sandra K, Kavita K (2018) Genome sequence of Bacillus velezensis SGAir0473, isolated from tropical air collected in Singapore. Genome Announc 6(27):205–211. https://doi.org/10.1128/genomea.00642-18
Shafi J, Tian H, Ji MS (2017) Bacillus species as versatile weapons for plant pathogens: a review. Biotechnol Biotechnol Equip 31(3):446–459. https://doi.org/10.1080/13102818.2017.1286950
Singh M, Chaudhary S, Sareen D (2017) Non-ribosomal peptide synthetases: Identifying the cryptic gene clusters and decoding the natural product. J Biosci 42:1–13. https://doi.org/10.1007/s12038-017-9663-z
Sonowal S, Ahmed R, Chikkaputtaiah C, Basar E, Velmurugan N (2022) A comprehensive characterization of culturable endophytic bacteria of Paris polyphylla and their potential role in microalgal growth in co-culture. Applied Soil Ecology 174:104410. https://doi.org/10.1016/j.apsoil.2022.104410
Sumi CD, Yang BW, Yeo IC (2015) Antimicrobial peptides of the genus Bacillus: a new era for antibiotics. Can J Microbiol 61(2):93–103. https://doi.org/10.1139/cjm-2014-0613
Tran DM, Huynh TU, Nguyen TH, Do TO, Nguyen QV, Nguyen AD (2022) Molecular analysis of genes involved in chitin degradation from the chitinolytic bacterium Bacillus velezensis. Antonie Van Leeuwenhoek 115:215–231. https://doi.org/10.1007/s10482-021-01697-2
Vignesh M, Shankar SRM, MubarakAli D, Hari BNV (2022) A novel rhizospheric bacterium: Bacillus velezensis NKMV-3 as a biocontrol agent against alternaria leaf blight in tomato. Appl Biochem Biotechnol 194:1–17. https://doi.org/10.1007/s12010-021-03684-9
Wang B, Peng H, Wu W, Yang B, Chen Y, Xu F, Peng Y, Qin Y, Fu P, Lu J (2021) Genomic insights into biocontrol potential of Bacillus stercoris LJBS06. Biotech 11:458. https://doi.org/10.1007/s13205-021-03000-6
Wang KX, Xu WH, Chen ZN, Hu JL, Luo SQ, Wang ZG (2022) Complete genome sequence of Bacillus velezensis WB, an isolate from the watermelon rhizosphere: genomic insights into its antifungal effects. J Global Antimicrob Resist 30(1):442–444. https://doi.org/10.1016/j.jgar.2022.05.010
Xia J, Psychogios N, Young N, Wishart DS (2009) MetaboAnalyst: a web server for metabolomic data analysis and interpretation. Nucleic Acids Res 37:W652-660. https://doi.org/10.1093/nar/gkp356
Yi Y, Zhang Z, Zhao F, Liu H, Wang G (2018) Probiotic potential of Bacillus velezensis JW: antimicrobial activity against fish pathogenic bacteria and immune enhancement effects on Carassius auratus. Fish Shellfish Immunol 78:322–330. https://doi.org/10.1016/j.fsi.2018.04.055
Funding
This work was sponsored by the One Health Interdisciplinary Research Project, Ningbo University; the Ningbo Science and Technology Correspondent Team Project; the Natural Science Foundation of Zhejiang Province (LGC22C190002); and the KC Wong Magna Fund at Ningbo University.
Author information
Authors and Affiliations
Contributions
W.Z. performed the conceptualization, data curation, formal analysis, and writing-original draft; Z.W., W.Z., Z.Z., L.Y., L.S., W.Q., and E.S. performed validation and visualization; H.J., Z.J., X.J., and Y.X. performed supervision, validation, and visualization; and Z.Z. designed the experiments, contributed to experimental materials, writed the original draft and revised the paper; S.X., and W.R. designed the experiments, contributed to experimental materials, and revised the paper. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Communicated by Yusuf Akhter.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
203_2024_3861_MOESM1_ESM.docx
Supplementary file1 Table S1 Antibacterial results of B. velezensis NDB strains screened by the disk-diffusion assay. Table S2 Prediction of secondary metabolites of B. velezensis NDB genome. Table S3 Prediction results of B. velezensis NDB bacteriocins. Supplementary Table 4 Primers used in this study. Table S5 Concentration of intracellular metabolites of B. velezensis NDB. (DOCX 34 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Z., Zhang, W., Wang, Z. et al. Analysis of antimicrobial biological activity of a marine Bacillus velezensis NDB. Arch Microbiol 206, 131 (2024). https://doi.org/10.1007/s00203-024-03861-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-024-03861-4