Abstract
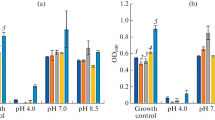
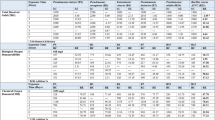
Oxytetracycline (OTC), is a widely used veterinary antibiotic for treatment and prophylaxis in aquaculture. As an emerging pollutant, OTC in the environment exerts selective pressure on aquatic organisms causing proliferation of antibiotic resistant genes. In the present study, an OTC tolerant isolate labelled as pw2 was selected among the 11 OTC tolerant isolates, isolated from the aquaculture effluent, for investigating its OTC degrading potential. The cell morphology, biochemical characteristics, and 16S ribosomal RNA (rRNA) gene sequence of the isolated strain indicated that it belonged to the genus Planococcus. The OTC removal percentage was estimated through measuring its residual concentration in the culture medium with high performance liquid chromatography. The strain exhibited maximum removal efficiency of 90.62%, with initial OTC concentration of 10 µg/ml. The optimum degrading conditions were 35 °C and pH 7. The degradation rate of OTC with (biotic) and without strain pw2 (abiotic) was 3.253 and 1.149 mg/l/d, respectively. The half-life was recorded to be 2.13 d in the presence of strain pw2, in contrast to 6.03 days recorded without strain pw2. The total (biotic + abiotic) OTC degradation efficiency was 75.74, 83.93, 90.62, and 86.47% for the initial OTC concentrations of 1 to 25 µg/ml, respectively. Addition of carbon and nitrogen did not influence the OTC removal which indicates Planococcus sp. pw2 use OTC as sole energy source. Thus, Planococcus sp. pw2 plays a vital role in reducing the OTC concentration in the environment, offering a promising method for treatment of aquaculture effluent containing OTC.





Similar content being viewed by others
References
Ahmed T, Baidya S, Sharma BC et al (2013) Identification of drug-resistant bacteria among export quality shrimp samples in Bangladesh. Asian J Microbiol Biotechnol Environ Exp Sci 15:655–660. https://doi.org/10.1093/ajcp/45.4_ts.493
Altinok I (2017) Adverse effects of mariculture activities and practices on marine environment. Oceanogr Fish Open Access J. https://doi.org/10.19080/ofoaj.2017.04.555630
Arikan O, Mulbry W, Ingram D, Millner P (2009a) Minimally managed composting of beef manure at the pilot scale: Effect of manure pile construction on pile temperature profiles and on the fate of oxytetracycline and chlortetracycline. Biores Technol 100:4447–4453. https://doi.org/10.1016/j.biortech.2008.12.063
Arikan OA, Mulbry W, Rice C (2009b) Management of antibiotic residues from agricultural sources: use of composting to reduce chlortetracycline residues in beef manure from treated animals. J Hazard Mater 164:483–489. https://doi.org/10.1016/j.jhazmat.2008.08.019
Belkacem S, Bouafia S, Chabani M (2017) Study of oxytetracycline degradation by means of anodic oxidation process using platinized titanium (Ti/Pt) anode and modeling by artificial neural networks. Process Saf Environ Prot 111:170–179. https://doi.org/10.1016/j.psep.2017.07.007
Berglund B, Khan GA, Weisner SEB et al (2014) Efficient removal of antibiotics in surface-flow constructed wetlands, with no observed impact on antibiotic resistance genes. Sci Total Environ 476–477:29–37. https://doi.org/10.1016/j.scitotenv.2013.12.128
Brambilla G, Patrizii M, De Filippis SP et al (2007) Oxytetracycline as environmental contaminant in arable lands. Anal Chim Acta 586:326–329. https://doi.org/10.1016/j.aca.2006.11.019
Cabello FC (2006) Heavy use of prophylactic antibiotics in aquaculture: a growing problem for human and animal health and for the environment. Environ Microbiol 8:1137–1144
Chen W, Liu W, Pan N et al (2013) Oxytetracycline on functions and structure of soil microbial community. J Soil Sci Plant Nutr 13:967–975. https://doi.org/10.4067/S0718-95162013005000076
Choi YJ, Kim LH, Zoh KD (2016) Removal characteristics and mechanism of antibiotics using constructed wetlands. Ecol Eng 91:85–92. https://doi.org/10.1016/j.ecoleng.2016.01.058
De Liguoro M, Cibin V, Capolongo F et al (2003) Use of oxytetracycline and tylosin in intensive calf farming: Evaluation of transfer to manure and soil. Chemosphere 52:203–212. https://doi.org/10.1016/S0045-6535(03)00284-4
Dharmadasa PS, Liyanage GY, Manage PM (2017) Degradation of microcystin LR, oxytetracycline and amphicillin by four native bacteria species. J Trop for Environ. https://doi.org/10.31357/jtfe.v7i2.3305
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32:1792–1797. https://doi.org/10.1093/nar/gkh340
Edwards U, Rogall T, Blöcker H et al (1989) Isolation and direct complete nucleotide determination of entire genes. Characterization of a gene coding for 16S ribosomal RNA. Nucleic Acids Res 17:7843–7853. https://doi.org/10.1093/nar/17.19.7843
Felsenstein J (1985) Phylogenies and the comparative method. Am Nat 125:1–15. https://doi.org/10.1086/284325
Ghosh S, Sadowsky MJ, Roberts MC et al (2009) Sphingobacterium sp. strain PM2-P1–29 harbours a functional tet(X) gene encoding for the degradation of tetracycline. J Appl Microbiol. https://doi.org/10.1111/j.1365-2672.2008.04101.x
Gullberg E, Cao S, Berg OG et al (2011) Selection of resistant bacteria at very low antibiotic concentrations. PLoS Pathog. https://doi.org/10.1371/journal.ppat.1002158
Guo T, Lou C, Zhai W et al (2018) Increased occurrence of heavy metals, antibiotics and resistance genes in surface soil after long-term application of manure. Sci Total Environ 635:995–1003. https://doi.org/10.1016/j.scitotenv.2018.04.194
Halling-Sørensen B, Sengeløv G, Ingerslev F, Jensen LB (2003) Reduced antimicrobial potencies of oxytetracycline, tylosin, sulfadiazin, streptomycin, ciprofloxacin, and olaquindox due to environmental processes. Arch Environ Contam Toxicol 44:7–16. https://doi.org/10.1007/s00244-002-1234-z
Harrabi M, Alexandrino DAM, Aloulou F et al (2019) Biodegradation of oxytetracycline and enrofloxacin by autochthonous microbial communities from estuarine sediments. Sci Total Environ 648:962–972. https://doi.org/10.1016/j.scitotenv.2018.08.193
Hirsch R, Ternes TA, Haberer K et al (1998) Determination of antibiotics in different water compartments via liquid chromatography-electrospray tandem mass spectrometry. J Chromatogr A. https://doi.org/10.1016/S0021-9673(98)00335-5
Ingerslev F, Toräng L, Loke ML et al (2001) Primary biodegradation of veterinary antibiotics in aerobic and anaerobic surface water simulation systems. Chemosphere 44:865–872. https://doi.org/10.1016/S0045-6535(00)00479-3
Jabeen H, Iqbal S, Anwar S, Parales RE (2015) Optimization of profenofos degradation by a novel bacterial consortium PBAC using response surface methodology. Int Biodeterior Biodegrad 100:89–97. https://doi.org/10.1016/j.ibiod.2015.02.022
Jukes TH, Cantor CR (1969) Evolution of protein molecules BT—mammalian protein metabolism. Mammalian protein metabolism. Academic Press, New York, pp 21–132
Leng Y, Bao J, Chang G et al (2016) Biotransformation of tetracycline by a novel bacterial strain Stenotrophomonas maltophilia DT1. J Hazard Mater 318:125–133. https://doi.org/10.1016/j.jhazmat.2016.06.053
Li S, Zhang S, Ye C et al (2017) Biofilm processes in treating mariculture wastewater may be a reservoir of antibiotic resistance genes. Mar Pollut Bull 118:289–296. https://doi.org/10.1016/j.marpolbul.2017.03.003
Li Zj, Qi Wn, Feng Y et al (2019) Degradation mechanisms of oxytetracycline in the environment. J Integr Agric 18:1953–1960
Liu Y, Chang H, Li Z et al (2017) Biodegradation of gentamicin by bacterial consortia AMQD4 in synthetic medium and raw gentamicin sewage. Sci Rep. https://doi.org/10.1038/s41598-017-11529-x
Magdaleno A, Carusso S, Moretton J (2017) Toxicity and genotoxicity of three antimicrobials commonly used in veterinary medicine. Bull Environ Contam Toxicol 99:315–320. https://doi.org/10.1007/s00128-017-2091-9
Maki T, Hasegawa H, Kitami H et al (2006) Bacterial degradation of antibiotic residues in marine fish farm sediments of Uranouchi Bay and phylogenetic analysis of antibiotic-degrading bacteria using 16S rDNA sequences. Fish Sci. https://doi.org/10.1111/j.1444-2906.2006.01222.x
Migliore L, Fiori M, Spadoni A, Galli E (2012) Biodegradation of oxytetracycline by Pleurotus ostreatus mycelium: a mycoremediation technique. J Hazard Mater 215–216:227–232. https://doi.org/10.1016/j.jhazmat.2012.02.056
Moghadam MS, Ebrahimipour G, Abtahi B, Ghassempour A (2013) Isolation, identification and optimization of phenanthrene degrading bacteria from the coastal sediments of Nayband Bay. Jundishapur J Microbiol. https://doi.org/10.5812/jjm.13816
Park H (2012) Reduction of antibiotics using microorganisms containing glutathione S-transferases under immobilized conditions. Environ Toxicol Pharmacol 34:345–350. https://doi.org/10.1016/j.etap.2012.05.011
Ramesh M, Thilagavathi T, Rathika R, Poopal RK (2018) Antioxidant status, biochemical, and hematological responses in a cultivable fish Cirrhinus mrigala exposed to an aquaculture antibiotic Sulfamethazine. Aquaculture 491:10–19. https://doi.org/10.1016/j.aquaculture.2018.02.046
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Santos L, Ramos F (2016) Analytical strategies for the detection and quantification of antibiotic residues in aquaculture fishes: a review. Trends Food Sci Technol 52:16
Shao S, Hu Y, Cheng J, Chen Y (2019) Action of oxytetracycline (OTC) degrading bacterium and its application in Moving Bed Biofilm Reactor (MBBR) for aquaculture wastewater pre-treatment. Ecotoxicol Environ Saf. https://doi.org/10.1016/j.ecoenv.2019.01.040
Shen DS, Tao XQ, Shentu JL, Wang MZ (2014) Residues of veterinary antibiotics in pig feeds and manures in Zhejiang province. Adv Mater Res 1010–1012:301–304
Suda T, Hata T, Kawai S et al (2012) Treatment of tetracycline antibiotics by laccase in the presence of 1-hydroxybenzotriazole. Biores Technol 103:498–501. https://doi.org/10.1016/j.biortech.2011.10.041
Tran HT, Pham TV, Ngo HPT et al (2014) Crystallization and preliminary X-ray crystallographic analysis of the XoGroEL chaperonin from Xanthomonas oryzae pv. oryzae. Acta Crystallogr Sect f: Struct Biol Commun 70:604–607. https://doi.org/10.1107/S2053230X14006591
Vlková E, Rada V, Popelářová P et al (2006) Antimicrobial susceptibility of bifidobacteria isolated from gastrointestinal tract of calves. Livest Sci 105:253–259. https://doi.org/10.1016/j.livsci.2006.04.011
Wen X, Jia Y, Li J (2010) Enzymatic degradation of tetracycline and oxytetracycline by crude manganese peroxidase prepared from Phanerochaete chrysosporium. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2010.01.005
Xie X, Zhou Q, Lin D et al (2011) Toxic effect of tetracycline exposure on growth, antioxidative and genetic indices of wheat (Triticum aestivum L.). Environ Sci Pollut Res 18:566–575. https://doi.org/10.1007/s11356-010-0398-8
Xiong W, Sun Y, Zhang T et al (2015) Antibiotics, antibiotic resistance genes, and bacterial community composition in fresh water aquaculture environment in China. Microb Ecol 70:425–432. https://doi.org/10.1007/s00248-015-0583-x
Yin H, Yang C, Jia Y et al (2018) Dual removal of phosphate and ammonium from high concentrations of aquaculture wastewaters using an efficient two-stage infiltration system. Sci Total Environ 635:936–946. https://doi.org/10.1016/j.scitotenv.2018.04.218
Acknowledgements
S. Suruttaiyan would like to thank Bharathiar University, Tamilnadu, India for granting financial assistance in the form of University Research Fellowship.
Author information
Authors and Affiliations
Contributions
SS and SK carried out the experiments. PD designed the project and prepared the manuscript with LP. SS reviewed the manuscript.
Corresponding authors
Additional information
Communicated by Yusuf Akhter.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Suruttaiyan, S., Duraisamy, P., Krishnaraj, S. et al. Isolation, characterization and degradation performance of oxytetracycline degrading bacterium Planococcus sp. strain pw2. Arch Microbiol 204, 122 (2022). https://doi.org/10.1007/s00203-021-02732-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-021-02732-6