Abstract
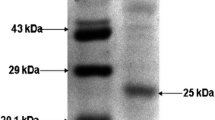
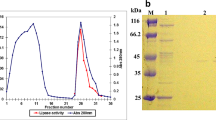
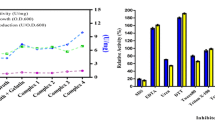
A halotolerant bacterial strain isolated and identified as Bacillus gibsonii was used for extracellular lipase production. The bacterial strain was able to grow up to 1200 mM salt concentration and showed maximum growth at 600 mM NaCl concentration. The present study includes production of extracellular lipase enzyme and characterization of partially purified lipase with respect to its kinetic and thermodynamic behaviour. Maximum lipase activity was observed at 60 °C under alkaline pH (9.0) condition. The kinetic parameters such as Vmax, Km and Kcat were calculated as 158.73 U/mL, 0.539 mM and 483.93 min−1 at 60 °C, respectively, suggested thermostable nature of the enzyme. The thermal inactivation energy [Ea(d)] was calculated as 66.98 kJ/mol. The values of Gibb’s free energy (86.31 kJ/mol), enthalpy (64.26 kJ/mol) and entropy (− 66.21 × 10–3 kJ/mol/K) for the enzyme inactivation obtained at 60 °C corroborated the assumption that 60 °C was the optimum temperature. Further, the deactivation rate constant (kd) values calculated at 60 °C and 80 °C were found to be 0.0907 and 0.182 min−1, respectively, which suggested that enzyme was more stable at 60 °C and it was partly inactivated at 80 °C.









Similar content being viewed by others
References
Abdel-Fattah YR, Gaballa AA (2008) Identification and over-expression of a thermostable lipase from Geobacillus thermoleovorans Toshki in Escherichia coli. Microbio res 163:13–20. https://doi.org/10.1016/j.micres.2006.02.004
Adams DM, Brawley TG (1981) Factors influencing the heat resistance of a heat-resistant lipase of Pseudomonas. J Food Sci 46:673–676. https://doi.org/10.1111/j.1365-2621.1981.tb15321.x
Ariahu CC, Ogunsua AO (2000) Thermal degradation kinetics of thiamine in periwinkle based formulated low acidity foods. Int J Food Sci Technol 35:315–321. https://doi.org/10.1046/j.1365-2621.2000.00366.x
Balan A, Ibrahim D, Abdul Rahim R, Ahmad Rashid FA (2012) Purification and characterization of a thermostable lipase from Geobacillus thermodenitrificans IBRL-nra. Enzyme Res. https://doi.org/10.1155/2012/987523
Bora L, Kalita MC (2008) Production of thermostable alkaline lipase on vegetable oils from a thermophilic Bacillus sp. DH4, characterization and its potential applications as detergent additive. J Chem Technol Biotechnol 83:688–693. https://doi.org/10.1002/jctb.1853
Ertuğrul S, Dönmez G, Takaç S (2007) Isolation of lipase producing Bacillus sp. from olive mill wastewater and improving its enzyme activity. J Hazard Mater 149:720–724. https://doi.org/10.1016/j.jhazmat.2007.04.034
Gandhi NN (1997) Applications of lipase. J Am Oil Chem Soc 74:621–634. https://doi.org/10.1007/s11746-997-0194-x
Goswami D, De S, Basu JK (2012) Effects of process variables and additives on mustard oil hydrolysis by porcine pancreas lipase. Braz J Chem Eng 29:449–460. https://doi.org/10.1590/S0104-66322012000300002
Griebeler N, Polloni AE, Remonatto D, Arbter F, Vardanega R, Cechet JL, Di Luccio M, de Oliveira D, Treichel H, Cansian RL, Rigo E (2011) Isolation and screening of lipase-producing fungi with hydrolytic activity. Food Bioproc Tech 4:578–586. https://doi.org/10.1007/s11947-008-0176-5
Griffiths MW, Phillips JD, Muir DD (1981) Thermostability of proteases and lipases from a number of species of psychrotrophic bacteria of dairy origin. J Appl Microbiol 50:289–303. https://doi.org/10.1111/j.1365-2672.1981.tb00894.x
Hasan F, Shah AA, Hameed A (2006) Industrial applications of microbial lipases. Enzyme Microb Technol 39:235–251. https://doi.org/10.1016/j.enzmictec.2005.10.016
Hiol A, Jonzo MD, Druet D, Comeau L (1999) Production, purification and characterization of an extracellular lipase from Mucor hiemalis f. hiemalis. Enzyme Microb Technol 25:80–87. https://doi.org/10.1016/S0141-0229(99)00009-5
Houde A, Kademi A, Leblanc D (2004) Lipases and their industrial applications. Appl Biochem Biotechnol 118:155–170
Jaenicke R (1991) Protein stability and molecular adaptation to extreme conditions. In: Christen P, Hofmann E (eds) EJB Reviews, vol 1991. Springer, Heidelberg
Kambourova M, Kirilova N, Mandeva R, Derekova A (2003) Purification and properties of thermostable lipase from a thermophilic Bacillus stearothermophilus MC 7. J. Mol. Catal. B Enzym 22:307–313. https://doi.org/10.1016/S1381-1177(03)00045-6
Kim HK, Sung MH, Kim HM, Oh TK (1994) Occurrence of thermostable lipase in thermophilic Bacillus sp. strain 398. Biosci Biotechnol Biochem 58:961–962. https://doi.org/10.1271/bbb.58.961
Kim HK, Park SY, Lee JK, Oh TK (1998) Gene cloning and characterization of thermostable lipase from Bacillus stearothermophilus L1. Biosci Biotechnol Biochem 62:66–71. https://doi.org/10.1271/bbb.62.66
Kim MH, Kim HK, Lee JK, Park SY, Oh TK (2000) Thermostable lipase of Bacillus stearothermophilus: high-level production, purification, and calcium-dependent thermostability. Biosci Biotechnol Biochem 64:280–286. https://doi.org/10.1271/bbb.64.280
Kristjansson MM, Kinsella JE (1991) Protein and enzyme stability: structural, thermodynamic, and experimental aspects. Adv Food Nutr Res 35:237–316. https://doi.org/10.1016/S1043-4526(08)60066-2
Kumar R, Sharma A, Kumar A, Singh D (2012) Lipase from Bacillus pumilus RK31: production, purification and some properties. World Appl Sci J 16:940–948
Kumari A, Gupta R (2012) Extracellular expression and characterization of thermostable lipases, LIP8, LIP14 and LIP18, from Yarrowia lipolytica. Biotechnol Lett 34:1733–1739. https://doi.org/10.1007/s10529-012-0958-8
Lee DW, Koh YS, Kim KJ, Kim BC, Choi HJ, Kim DS, Suhartono MT, Pyun YR (1999) Isolation and characterization of a thermophilic lipase from Bacillus thermoleovorans ID-1. FEMS Microbiol Lett. 179:393–400. https://doi.org/10.1111/j.1574-6968.1999.tb08754.x
Li X, Yu HY (2014) Characterization of an organic solvent-tolerant lipase from Haloarcula sp. G41 and its application for biodiesel production. Folia Microbiol 59:455–463. https://doi.org/10.1007/s12223-014-0320-8
Li H, Zhang X (2005) Characterization of thermostable lipase from thermophilic Geobacillus sp. TW1. Protein Expr Purif 42:153–159. https://doi.org/10.1016/j.pep.2005.03.011
Lineweaver H, Burk D (1934) The determination of enzyme dissociation constants. J Am Chem Soc 56(3):658–666. https://doi.org/10.1021/ja01318a036
Lowry RR, Tinsley IJ (1976) Rapid colorimetric determination of free fatty acids. J Am Oil Chem Soc 53:470–472. https://doi.org/10.1007/BF02636814
Nawani N, Kaur J (2000) Purification, characterization and thermostability of lipase from a thermophilic Bacillus sp. J33. Mol Cell Biochem 206:91–96. https://doi.org/10.1023/A:1007047328301
Noormohamadi R, Tabandeh F, Shariati P, Otadi M (2013) Characterization of a lipase from a newly isolated Pseudomonas sp. Iran J Microbiol 5:422
Olusesan AT, Azura LK, Forghani B, Bakar FA, Mohamed AK, Radu S, Manap MY, Saari N (2011) Purification, characterization and thermal inactivation kinetics of a non-regioselective thermostable lipase from a genotypically identified extremophilic Bacillus subtilis NS 8. N Biotechnol 28:738–745. https://doi.org/10.1016/j.nbt.2011.01.002
Ortega N, De Diego S, Perez-Mateos M, Busto MD (2004) Kinetic properties and thermal behaviour of polygalacturonase used in fruit juice clarification. Food chem 88:209–217. https://doi.org/10.1016/j.foodchem.2004.01.035
Owusu RK, Makhzoum A, Knapp JS (1992) Heat inactivation of lipase from psychrotrophic Pseudomonas fluorescens P38: activation parameters and enzyme stability at low or ultra-high temperatures. Food chem 44:261–268. https://doi.org/10.1016/0308-8146(92)90048-7
Prajapati V, Patel H, Trivedi U, Patel K (2014) Kinetic and thermodynamic characterization of lipase produced by Cellulomonas flavigena UNP3. J Basic Microbiol 54:976–983. https://doi.org/10.1002/jobm.201300065
Rahman RN, Baharum SN, Basri M, Salleh AB (2005) High-yield purification of an organic solvent-tolerant lipase from Pseudomonas sp. strain S5. Anal Biochem 341:267–274. https://doi.org/10.1016/j.ab.2005.03.006
Royter M, Schmidt M, Elend C, Höbenreich H, Schäfer T, Bornscheuer UT, Antranikian G (2009) Thermostable lipases from the extreme thermophilic anaerobic bacteria Thermoanaerobacter thermohydrosulfuricus SOL1 and Caldanaerobacter subterraneus subsp. tengcongensis. Extremophiles 1:769–783. https://doi.org/10.1007/s00792-009-0265-z
Sarmah N, Revathi D, Sheelu G, Yamuna Rani K, Sridhar S, Mehtab V, Sumana C (2018) Recent advances on sources and industrial applications of lipases. Biotechnol Prog 34:5–28. https://doi.org/10.1002/btpr.2581
Schmidt-Dannert C, Rua ML, Atomi H, Schmid RD (1996) Thermoalkalophilic lipase of Bacillus thermocatenulatus. I. Molecular cloning nucleotide sequence purification and some properties. BBA 1301:105–114. https://doi.org/10.1016/0005-2760(96)00027-6
Segel IH (1976) Enzymes. Biochemical calculations 2nd 447 edn. John Wiley and Sons, pp 208–323.
Sharma R, Soni SK, Vohra RM, Gupta LK, Gupta JK (2002) Purification and characterisation of a thermostable alkaline lipase from a new thermophilic Bacillus sp. RSJ-1. Process Biochem 37:1075–1084. https://doi.org/10.1016/S0032-9592(01)00316-8
Sharon C, Nakazato M, Ogawa HI, Kato Y (1998) Lipase-induced hydrolysis of castor oil: effect of various metals. J Ind Microbiol Biotechnol 21:292–295. https://doi.org/10.1038/sj.jim.2900586
Sinchaikul S, Sookkheo B, Phutrakul S, Pan FM, Chen ST (2001) Optimization of a thermostable lipase from Bacillus stearothermophilus P1: overexpression, purification, and characterization. Protein Expr Purif 22:388–398. https://doi.org/10.1006/prep.2001.1456
Sugihara A, Tani T, Tominaga Y (1991) Purification and characterization of a novel thermostable lipase from Bacillus sp. J Biochem 109:211–216. https://doi.org/10.1093/oxfordjournals.jbchem.a123363
Treves C, Vincenzini MT, Favilli F, Vanni P, Baccari V (1984) On the interaction between synthetic detergents and enzymatic proteins. Canadian J Biochem Cell Biol 62:55–59. https://doi.org/10.1139/o84-009
Tyndall JD, Sinchaikul S, Fothergill-Gilmore LA, Taylor P, Walkinshaw MD (2002) Crystal structure of a thermostable lipase from Bacillus stearothermophilus P1. J Mol Biol 323:859–869. https://doi.org/10.1016/S0022-2836(02)01004-5
Wang Y, Srivastava KC, Shen GJ, Wang HY (1995) Thermostable alkaline lipase from a newly isolated thermophilic Bacillus strain A30–1 (ATCC 53841). J Ferment Bioeng 79:433–438. https://doi.org/10.1016/0922-338X(95)91257-6
Whitaker JR (1993) Principles of enzymology for the food sciences. CRC press
Acknowledgements
The authors are thankful to University Grant Commission (UGC), New Delhi, Government of India for providing financial support for the research work. Author is obliged to The Head, Department of Environmental Science, Babasaheb Bhimrao Ambedkar University, Lucknow (India) for providing the laboratory facilities during the investigation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that this manuscript does not have any conflicts of interest regarding the financial, scientific contents and otherwise.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sonkar, K., Singh, D.P. Kinetic and thermodynamic characterization of novel alkaline lipase from halotolerant Bacillus gibsonii. Arch Microbiol 203, 2199–2209 (2021). https://doi.org/10.1007/s00203-021-02197-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-021-02197-7