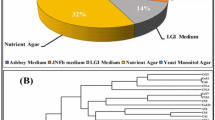
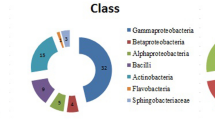
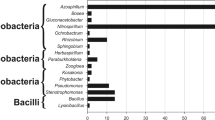
Abstract
This study aimed to evaluate the genetic diversity of bacterial community associated to different sugarcane genotypes, association habitat and phenological phase of the culture, as well as to isolate, to identify and to characterize your potential for plant growth-promoting. Root and rhizospheric soil samples from RB 92579 and RB 867515 varieties were collected at 120 and 300 days after regrowth (DAR). The diversity of bacterial was evaluated through of the 16S rRNA and nifH genes. We found greater genetic diversity in the root endophytic habitat at 120 DAR. We identify the genera Burkholderia sp., Pantoea sp., Erwinia sp., Stenotrophomonas sp., Enterobacter sp. and Pseudomonas sp. The genera Bacillus sp. and Dyella sp. were only identified in the variety RB 92579. We found indices above 50% for biological nitrogen fixation, production of indole acetic acid and phosphate solubilization, showing that the use of these bacteria in biotechnological products is very promising.






Similar content being viewed by others
References
Alvares CA, Stape JL, Sentelhas PC, Gonçalves JLM, Sparovek V (2013) Köppen’s climate classification map for Brazil. Meteorol Z 22:711–728. https://doi.org/10.1127/0941-2948/2013/0507
Araújo FF, Guerreiro RT (2010) Bioprospecção de isolados de Bacillus promotores de crescimento de milho cultivado em solo natural e autoclavados. Ciênc agrotec 34:837–844. https://doi.org/10.1590/S1413-70542010000400007
Backer R, Rokem JS, Ilangumaran G, Lamont J, Praslickova D, Ricci E, Subramanian S, Smith DL (2018) Plant growth-promoting rhizobacteria: context, mechanisms of action, and roadmap to commercialization of biostimulants for sustainable agriculture. Front Plant Sci 9:1473. https://doi.org/10.3389/fpls.2018.01473
Beneduzi A, Moreira F, Costa PB, Vargas LK, Lisboa BB, Favreto R, Baldani JI, Passaglia LMP (2013) Diversity and plant growth promoting evaluation abilities of bacteria isolated from sugarcane cultivated in the South of Brazil. Applied Soil Ecology 63:94–104
Beltrão BA, Mascarenhas JC, Miranda JLF, Souza Junior LC, Galvão MJTG, Pereira SN (2005) Projeto cadastro de fontes de abastecimento por água subterrânea do estado de Pernambuco: diagnóstico do município de Carpina. IOP Publishig PhysicsWeb. https://rigeo.cprm.gov.br/jspui/handle/doc/15830. Accessed 29 June 2020
Berraqueiro FR, Baya AM, Cormenzana AR (1976) Establecimiento de índices para el estudio de la solubilizacion de fosfatos por bacterias del suelo. ARS Pharm 17:399–406
Bonnett GD (2013) Developmental stages (phenology). In: Moore PH, Botha FC (eds) Sugarcane: physiology, biochemistry, and functional Biology, 3rd edn. Wiley, New York, pp 35–54
Chelius MK, Triplett EW (2001) The diversity of archaea and bacteria in association with the roots of Zea mays L. Microbial Ecol 41:252–263. https://doi.org/10.1007/s002480000087
Clarke KR, Gorley RN (2001) Primer v5: user manual/tutorial. PRIMER-E, Plymouth
Compant S, Samad A, Faist H, Sessitsch A (2019) A review on the plant microbiome: ecology, functions, and emerging trends in microbial application. J Adv Res 19:29–37. https://doi.org/10.1016/j.jare.2019.03.004
Cordero I, Ruiz-Díez B, Balaguer L, RichteR A, Pueyo JJ, Rincón A (2016) Rhizospheric microbial community of Caesalpinia spinosa in preserved and deforested soils of the fog forest of Atiquipa, Peru. Soil Biol Biochem 114:132–141. https://doi.org/10.1016/j.apsoil.2017.02.015
Crozier A, Arruda P, Jasmim JM, Monteiro AM, Sandberg G (1988) Analysis of indole-3-acetic acid and related indóis in culture medium from Azospirillum lipoferum and Azospirillum brasilense. Appl Environ Microb 54:2833–2837
Dobereiner J, Ruschel AP (1958) Uma nova espécie de Beijerinckia. Rev Biol 1:261–272
Dobereiner J, Baldani VLD, Baldani JI (1995) Como isolar e identificar bactérias diazotróficas de plantas não-leguminosas. IOP Publishing PhysicsWeb. https://ainfo.cnptia.embrapa.br/digital/bitstream/item/98370/1/Como-isolar-e-identificar-bacterias.pdf. Accessed 29 June 2020
Heuer H, Krsek M, Baker PK, Smalla K, Wellington EW (1997) Analysis of actinomycete communities by specific amplification of genes encoding 16S rRNA and gel-electrophoretic separation in denaturing gradients. Appl Environ Microb 63:3233–3241
Ho A, Di Lonardo DP, Bodelier PLE (2017) Revisiting life strategy concepts in environmental microbial ecology. FEMS Microbiol Ecol 93:01–14. https://doi.org/10.1093/femsec/fix006
Hussain S, Anwar-Ul-Haq M, Hussain S, Akram Z, Afzal M, Shabbir I (2017) Best suited timing schedule of inorganic NPK fertilizers and its effect on qualitative and quantitative attributes of spring sown sugarcane (Saccharum officinarum L.). J Saudi Soc Agric Sci 16:66–71. https://doi.org/10.1016/j.jssas.2015.02.004
du Jardin P (2015) Plant biostimulants: Definition, concept, main categories and regulation. Scientia Horticulturae 196:3–14
Jorquera MA, Shaharoona B, Nadeem SM, Mora ML, Crowley DE (2012) Plant growth-promoting rhizobacteria associated with ancient clones of creosote bush (Larrea tridentata). Microb Ecol 64:1008–1017. https://doi.org/10.1007/s00248-012-0071-5
Kyllmar K, Stjernman FL, Andersson S, Martensson K (2014) Small agricultural monitoring catchments in Sweden representing environmental impact. Agric Ecosyst Environ 198:25–35. https://doi.org/10.1016/j.agee.2014.05.016
Lamizadeha E, Enayatizamira N, Motamedib H (2019) Difference in some biological properties of saline and non-saline soil under sugarcane cultivation. Eurasian Soil Sci 52:690–695. https://doi.org/10.1134/S1064229319060085
Lane DJ (1991) 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M (eds) Nucleic acid techniques in bacterial systematic, 4th edn. Wiley, New York, pp 115–175
Leite MCBS, Farias ARB, Freire FJ, Andreote FD, Kuklinsky-Sobral J, Freire MBGS (2014) Isolation, bioprospecting and diversity of salt-tolerant bacteria associated with sugarcane in soils of Pernambuco, Brazil. R Bras Eng Agrí Ambiental 18:S73–S79. https://doi.org/10.1590/1807-1929/agriambi.v18nsupps73-s79
Li D, Voigt TB, Kent AD (2016) Plant and soil effects on bacterial communities associated with Miscanthus giganteus rhizosphere and rhizomes. GCB Bioenergy 8:183–193. https://doi.org/10.1111/gcbb.12252
Lima DRM, Santos IB, Oliveira JTC, Barbosa JG, Diniz WPS, Farias ARB, Freire FJ, Sobral JK (2018) Tolerance of potentially diazotrophic bacteria to adverse environmental conditions and plant growth-promotion in sugarcane. Arch Agron Soil Sci 64:1534–1548. https://doi.org/10.1080/03650340.2018.1443212
Magnani GS, Didonet CM, Cruz LM, Picheth CF, Pedrosa FO, Souza EM (2010) Diversity of endophytic bacteria in Brazilian sugarcane. Genet Mol Res 9:250–258. https://doi.org/10.4238/vol9-1gmr703
Nübel U, Engelen B, Felske A, Snaidr J, Wieshuber A, Amann RI, Ludwig W, Backhaus H (1996) Sequence heterogeneities of genes encoding 16\thinspaceS rRNAs in Paenibacillus polymyxa detected by temperature gradient gel electrophoresis. J Bacteriol 178:5636–5643. https://doi.org/10.1128/jb.178.19.5636-5643.1996
Oliveira JTC, Figueredo EF, Diniz WPS, Oliveira LFP, Andrade PAM, Andreote FD, Kuklinsky-Sobral J, Lima DR, Freire FJ (2017) Diazotrophic bacterial community of degraded pastures. Appl Environ Soil Sci 1:1–10. https://doi.org/10.1155/2017/2561428
Ovreas L, Forney L, Daae FL, Torsvik V (1997) Distribution of bacterioplankton in meromictic Lake Saelenvannet, as determined by denaturing gradient gel electrophoresis of PCR-amplified gene fragments coding for 16S rRNA. Appl Environ Microbiol 63:3369–3373
Poly F, Ranjard L, Nazaret S, Gourbière F, Monrozier LJ (2001) Comparison of nifH gene pools in soils and soil microenvironments with contrasting properties. Appl Environ Microbiol 67:2255–2262. https://doi.org/10.1128/AEM.67.5.2255-2262.2001
Rodríguez-Blanco A, Sicardi M, Frioni L (2015) Plant genotype and nitrogen fertilization effects on abundance and diversity of diazotrophic bacteria associated with maize (Zea mays L.). Biol Fertil Soils 51:391–402. https://doi.org/10.1007/s00374-014-0986-8
Saharan BS, Nehra V (2011) Plant growth promoting rhizobacteria: a critical review. Life Sci Med Res 21:1–30
Santos HG, Jacomine PKT, Anjos LHC, Oliveira VA, Lumbreras JF, Coelho MR, Almeida JA, Cunha TJF, Oliveira JB (2013) Sistema brasileiro de classificação de solos. Embrapa, Brasília
Santos RL, Freire FJ, Oliveira ECA, Trivelin PCO, Freire MBGS, Bezerra PC, Oliveira RI, Santos MBC (2019) Changes in biological nitrogen fixation and natural-abundance N isotopes of sugarcane under molybdenum fertilization. Sugar Tech 21:925–935. https://doi.org/10.1007/s12355-019-00717-w
Sattolo TMS, Mariano E, Boschiero BN, Otto R (2017) Soil carbon and nitrogen dynamics as affected by land use change and successive nitrogen fertilization of sugarcane. Agric Ecosyst Environ 247:63–74. https://doi.org/10.1016/j.agee.2017.06.005
Shimoura N, Miyaji M, Tajima S, Nomura M (2020) Diversity of the bacteria in the roots of sugarcane used to produce Wasanbon in Kagawa, Japan. Plant Biotechnol 37:99–103. https://doi.org/10.5511/plantbiotechnology.20.0107a
Silva MO, Freire FJ, Kuklinsky-Sobral J, Oliveira ECA, Freire MBGS, Apolinário VXO (2016) Bacteria associated with sugarcane in Northeastern Brazil. Afr J Microbiol Res 10:1586–1594. https://doi.org/10.5897/AJMR2016.8241
Simões Neto DE, Melo LJOT, Chaves A, Lima ROR (2005) Lançamento de novas variedades RB de cana-de-açúcar. UFRPE, Carpina
Simonet P, Grosjean MC, Misra AK, Nazaret S, Cournoyer B (1991) Frankia genus-specific characterization by polymerase chain reaction. Appl Environ Microbiol 57:3278–3286
Soil Survey Staff (2014) Keys to soil taxonomy. USDA, Washington
Steindler L, Venturi V (2007) Detection of quorum-sensing N-acyl homoserine lactone signal molecules by bacterial bisensores. FEMS Microb Lett 266:1–9. https://doi.org/10.1111/j.1574-6968.2006.00501.x
Tikoo A, Tripathi AK, Verma SC, Agrawal N, Nath G (2001) Application of PCR fingerprinting techniques for identification and discrimination of Salmonella isolates. Curr Sci 80:1049–1052
Urquiaga S, Xavier RP, Morais RF, Batista RB, Schultz JML, Sá JM, Barbosa KP, Resende AS, Alves BJR, Boddey RM (2012) Evidence from field nitrogen balance and 15N natural abundance data for the contribution of biological N2 fixation to Brazilian sugarcane varieties. Plant Soil 356:5–21. https://doi.org/10.1007/s11104-011-1016-3
Vejan P, Abdullah R, Khadiran T, Ismail S, Boyce AN (2016) Role of plant growth promoting rhizobacteria in agricultural sustainability—a review. Molecules 21:573–590. https://doi.org/10.3390/molecules21050573
Versalovic J, Schneider M, Bruijn FJ, Lupski JR (1994) Genomic fingerprint of bacteria using repetitive sequence-based polymerase chain reaction. Methods Mol Cell Biol 5:25–40
Wissuwa M, Mazzola M, Picard C (2009) Novel approaches in plant breeding for rhizosphere-related traits. Plant Soil 321:409–430. https://doi.org/10.1007/s11104-008-9693-2
Yeoh YK, Paungfoo-Lonhienne C, Dennis PG, Robinson N, Ragan MA, Schmidt S, Hugenholtz P (2016) The core root microbiome of sugarcanes cultivated under varying nitrogen fertilizer application. Environ Microbiol 18:1338–1351. https://doi.org/10.1111/1462-2920.12925
Zehr JP, Turner PJ (2001) Nitrogen fixation: nitrogenase genes and gene expression. Methods Microbiol 30:271–286. https://doi.org/10.1016/S0580-9517
Zhan J, Sun Q (2012) Diversity of free-living nitrogen-fixing microorganisms in the rhizosphere and non-rhizosphere of pioneer plants growing on wastelands of copper mine tailings. Microbiol Res 167:157–165. https://doi.org/10.1016/j.micres.2011.05.006
Acknowledgements
Thanks to the Laboratory of Genetics and Microbial Biotechnology of the Federal University of the Agreste of Pernambuco (UFAPE), Federal Rural University of Pernambuco (UFRPE), University of São Paulo (USP), coordination and improvement of higher level personnel (CAPES), Foundation of protection of science and technology of the state of Pernambuco (FACEPE) and the National Councilor Scientific and Technological Development (CNPQ).
Funding
Funding not applicable.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. All authors contributed too for material preparation, data collection and analysis. In addition, the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
de Lima, D.R.M., dos Santos, I.B., Oliveira, J.T.C. et al. Genetic diversity of N-fixing and plant growth-promoting bacterial community in different sugarcane genotypes, association habitat and phenological phase of the crop. Arch Microbiol 203, 1089–1105 (2021). https://doi.org/10.1007/s00203-020-02103-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-020-02103-7