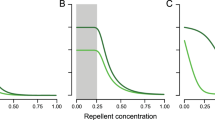
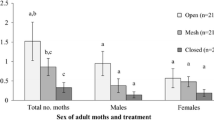
Abstract
Olfaction plays a major role in the host-finding behaviors of insects. However, the irregularity of insect responses to odor interactions has hindered our efforts to draw broad conclusions about how a host-finding insect uses the complex mixture of various odor plumes in natural environments. Particularly, it is still unclear so far why the use of non-host odors to control insect pests in practices have met with mixed results. To further understand the host-finding of a specific insect, we highlight the role of the real-time odor environment (ROE) that the host-finding insect is passing through. The ROE may contain various odors with different ranks and changes during the insect’s host finding. A host-finding insect may always prone to switch to the higher rank odor plumes in each ROE regardless of the distance is “short” or “long” from the odor source. For a specific herbivorous insect, only mixing degree of the given host and non-host plant odors reaches some certain level (threshold value), can the non-host odors significantly affect its ability to locate host plants. When the odor mixing degree is low, masking effects may not occur or the non-host plant odors’ “attractive” effects at long distances and “repellent” effects at short distances can even increase the pest loads. In forests, the mixing degree of different plant odors is determined by turbulence intensity which is mainly affected by plant structures. These may further advance our understanding of herbivorous insects’ host finding and have important implications for the development of pest management strategies.


Similar content being viewed by others
Availability of data and materials
Not applicable.
Code availability
Not applicable.
References
Alagarmalai J, Nestel D, Dragushich D, Nemny-Lavy E, Anshelevich L, Zada A, Soroker V (2009) Identification of host attractants for the ethiopian fruit fly, Dacus ciliatus loew. J Chem Ecol 35:542–551. https://doi.org/10.1007/s10886-009-9636-2
Andersson S (2003) Foraging responses in the butterflies Inachis io, Aglais urticae (Nymphalidae), and Gonepteryx rhamni (Pieridae) to floral scents. Chemoecology 13:1–11
Anderson P, Anton S (2014) Experience-based modulation of behavioural responses to plant volatiles and other sensory cues in insect herbivores. Plant, Cell Environ 37:1826–1835. https://doi.org/10.1111/pce.12342
Andersson S, Dobson HEM (2003) Behavioral foraging responses by the butterfly Heliconius melpomene to Lantana camara floral scent. J Chem Ecol 29:2303–2318
Baker TC, Fadamiro HY, Cosse AA (1998) Moth uses fine tuning for odour resolution. Nature 393:530–530. https://doi.org/10.1038/31131
Baldacchino F, Cadier J, Porciani A, Buatois B, Dormont L, Jay-Robert P (2013) Behavioural and electrophysiological responses of females of two species of tabanid to volatiles in urine of different mammals. Med Vet Entomol 27:77–85. https://doi.org/10.1111/j.1365-2915.2012.01022.x
Balkenius A, Rosen W, Kelber A (2006) The relative importance of olfaction and vision in a diurnal and a nocturnal hawkmoth. J Comp Physiol A Neuroethol Sens Neural Behav Physiol 192:431–437. https://doi.org/10.1007/s00359-005-0081-6
Balkenius A, Bisch-Knaden S, Hansson B (2009) Interaction of visual and odour cues in the mushroom body of the hawkmoth Manduca sexta. J Exp Biol 212:535–541. https://doi.org/10.1242/jeb.021220
Barbosa P, Hines J, Kaplan I, Martinson H, Szczepaniec A, Szendrei Z (2009) Associational resistance and associational susceptibility: Having right or wrong neighbors. Annu Rev Ecol Evol Syst 40:1–20. https://doi.org/10.1146/annurev.ecolsys.110308.120242
Beyaert I, Hilker M (2014) Plant odour plumes as mediators of plant-insect interactions. Biol Rev Camb Philos Soc 89:68–81. https://doi.org/10.1111/brv.12043
Birkett MA, Bruce TJA, Martin JL, Smart LE, Oakley J, Wadhams LJ (2004) Responses of female orange wheat blossom midge, Sitodiplosis mosellana, to wheat panicle volatiles. J Chem Ecol 30:1319–1328
Blight MM, Pickett JA, Wadhams LJ, Woodcock CM (1995) Antennal perception of oilseed rape, Brassica napus (Brassicaceae), volatiles by the cabbage seed weevil Ceutorhynchus assimilis (Coleoptera: Curculionidae). J Chem Ecol 21:1649–1664
Bodin A, Vinauger C, Lazzari CR (2009) Behavioural and physiological state dependency of host seeking in the blood-sucking insect Rhodnius prolixus. J Exp Biol 212:2386–2393. https://doi.org/10.1242/jeb.030668
Boyle SM, Mcinally S, Ray A (2013) Expanding the olfactory code by in silico decoding of odor-receptor chemical space. Elife 2:e01120. https://doi.org/10.7554/eLife.01120
Brady J, Gibson G, Packer MJ (1989) Odor movement, wind direction, and the problem of host-finding by tsetse flies. Physiol Entomol 14:369–380
Bruce TJ, Pickett JA (2011) Perception of plant volatile blends by herbivorous insects–finding the right mix. Phytochemistry 72:1605–1611. https://doi.org/10.1016/j.phytochem.2011.04.011
Bruce TJA, Wadhams LJ, Woodcock CM (2005) Insect host location: a volatile situation. Trends Plant Sci 10:269–274. https://doi.org/10.1016/j.tplants.2005.04.003
Budick SA (2006) Free-flight responses of Drosophila melanogaster to attractive odors. J Exp Biol 209:3001–3017. https://doi.org/10.1242/jeb.02305
Calatayud PA, Ahuya P, Le Ru B (2014) Importance of the experimental setup in research on attractiveness of odours in moths: an example with Busseola fusca. Entomol Exp Appl 152:72–76. https://doi.org/10.1111/eea.12201
Cardé RT, Willis MA (2008) Navigational strategies used by insects to find distant, wind-borne sources of odor. J Chem Ecol 34:854–866. https://doi.org/10.1007/s10886-008-9484-5
Carraher C, Dalziel J, Jordan MD, Christie DL, Newcomb RD, Kralicek AV (2015) Towards an understanding of the structural basis for insect olfaction by odorant receptors. Insect Biochem Mol Biol 66:31–41. https://doi.org/10.1016/j.ibmb.2015.09.010
Castagneyrol B, Jactel H, Vacher C, Brockerhoff EG, Koricheva J, Cadotte M (2014) Effects of plant phylogenetic diversity on herbivory depend on herbivore specialization. J Appl Ecol 51:134–141. https://doi.org/10.1111/1365-2664.12175
Castagneyrol B, Giffard B, Valdés-Correcher E, Hampe A (2019) Tree diversity effects on leaf insect damage on pedunculate oak: the role of landscape context and forest stratum. For Ecol Manage 433:287–294. https://doi.org/10.1016/j.foreco.2018.11.014
Conchou L, Lucas P, Meslin C, Proffit M, Staudt M, Renou M (2019) Insect odorscapes: from plant volatiles to natural olfactory scenes. Front Physiol. https://doi.org/10.3389/fphys.2019.00972
Cooperband MF, McElfresh JS, Millar JG, Cardé RT (2008) Attraction of female culex quinquefasciatus say (Diptera: Culicidae) to odors from chicken feces. J Insect Physiol 54:1184–1192. https://doi.org/10.1016/j.jinsphys.2008.05.003
Costantini C, Birkett MA, Gibson G, Ziesmann J, Sagnon NF, Mohammed HA, Coluzzi M, Pickett JA (2001) Electroantennogram and behavioural responses of the malaria vector Anopheles gambiae to human-specific sweat components. Med Vet Entomol 15:259–266
Couto A, Alenius M, Dickson BJ (2005) Molecular, anatomical, and functional organization of the Drosophila olfactory system. Curr Biol 15:1535–1547. https://doi.org/10.1016/j.cub.2005.07.034
Couty A, Van Emden H, Perry JN, Hardie JIM, Pickett JA, Wadhams LJ (2006) The roles of olfaction and vision in host-plant finding by the diamondback moth, Plutella xylostella. Physiol Entomol 31:134–145. https://doi.org/10.1111/j.1365-3032.2006.00499.x
Dethier VG, Browne BL, Smith CN (1960) The Designation of Chemicals in Terms of the Responses They Elicit from Insects. J Econ Entomol 53:134–136
Dicke M, de Boer JG, Höfte M, Rocha-Granados MC (2003) Mixed blends of herbivore-induced plant volatiles and foraging success of carnivorous arthropods. Oikos 101:38–48
Elkinton JS, Schal C, Ono T, Cardé RT (1987) Pheromone puff trajectory and upwind flight of male gypsy moths in a forest. Physiol Entomol 12:399–406
Fadamiro HY, Cossé AA, Baker TC (1999) Fine-scale resolution of closely spaced pheromone and antagonist filaments by flying male Helicoverpa zea. J Comp Physiol A 185:131–141. https://doi.org/10.1007/s003590050372
Faucher C, Forstreuter M, Hilker M, de Bruyne M (2006) Behavioral responses of Drosophila to biogenic levels of carbon dioxide depend on life-stage, sex and olfactory context. J Exp Biol 209:2739–2748. https://doi.org/10.1242/jeb.02297
Faucher CP, Hilker M, de Bruyne M (2013) Interactions of carbon dioxide and food odours in Drosophila: olfactory hedonics and sensory neuron properties. PLoS ONE 8:e56361. https://doi.org/10.1371/journal.pone.0056361
Ferreira RA, Guarneri AA, Lorenzo MG (2019) Activity and shelter-related behavior in Rhodnius prolixus: the role of host odours. Acta Trop 196:150–154. https://doi.org/10.1016/j.actatropica.2019.05.022
Finch S, Billiald H, Collier RH (2003) Companion planting – do aromatic plants disrupt host-plant finding by the cabbage root fly and the onion fly more effectively than non-aromatic plants? Entomol Exp Appl 109:185–193
Floater GJ, Zalucki MP (2000) Habitat structure and egg distributions in the processionary caterpillar Ochrogaster lunifer: lessons for conservation and pest management. J Appl Ecol 37:87–99
Galizia CG, Menzel R (2000) Odour perception in honeybees: coding information in glomerular patterns. Curr Opin Neurobiol 10:504–510
Galizia CG, Menzel R (2001) The role of glomeruli in the neural representation of odours: results from optical recording studies. J Insect Physiol 47:115–130
Gohole LS, Overholt WA, Khan ZR, Pickett JA, Vet LEM (2003) Effects of molasses grass, Melinis minutiflora volatiles on the foraging behavior of the cereal stemborer parasitoid, Cotesia sesamiae. J Chem Ecol 29:731–745
Hallem EA, Carlson JR (2006) Coding of odors by a receptor repertoire. Cell 125:143–160. https://doi.org/10.1016/j.cell.2006.01.050
Hallem EA, Ho MG, Carlson JR (2004) The molecular basis of odor coding in the Drosophila antenna. Cell 117:965–979. https://doi.org/10.1016/j.cell.2004.05.012
Haverkamp A, Hansson BS, Knaden M (2018) Combinatorial codes and labeled lines: How insects use olfactory cues to find and judge food, mates, and oviposition sites in complex environments. Front Physiol 9:49. https://doi.org/10.3389/fphys.2018.00049
Held DW, Gonsiska P, Potter DA (2003) Evaluating companion planting and non-host masking odors for protecting roses from the Japanese beetle (Coleoptera: Scarabaeidae). J Econ Entomol 96:81–87. https://doi.org/10.1093/jee/96.1.81
Hildebrand JG, Shepherd GM (1997) Mechanisms of olfactory discrimination: converging evidence for common principles across phyla. Annu Rev Neurosci 20:595–631
Hilker M, Kobs C, Varama M, Schrank K (2002) Insect egg deposition induces Pinus sylvestris to attract egg parasitoids. J Exp Biol 205:455–461
Isaacs R, Hardie J, Hick AJ, Pye BJ, Smart LE, Wadhams LJ, Woodcock CM (1993) Behavioural responses of Aphis fabae to isothiocyanates in the laboratory and field. Pest Manag Sci 39:349–355
Jactel H, Birgersson G, Andersson S, Schlyter F (2011) Non-host volatiles mediate associational resistance to the pine processionary moth. Oecologia 166:703–711. https://doi.org/10.1007/s00442-011-1918-z
Kadohisa M, Wilson DA (2006) Olfactory cortical adaptation facilitates detection of odors against background. J Neurophysiol 95:1888–1896. https://doi.org/10.1152/jn.00812.2005
Kaiser L, Willis MA, Cardé RT (1994) Flight manoeuvers used by a parasitic wasp to locate host-infested plant. Entomol Exp Appl 70:285–294. https://doi.org/10.1111/j.1570-7458.1994.tb00757.x
Kaissling KE (1986) Chemo-electrical transduction in insect olfactory receptors. Annu Rev Neurosci 9:121–145
Kaissling KE (1996) Peripheral mechanisms of pheromone reception in moths. Chem Senses 21:257–268. https://doi.org/10.1093/chemse/21.2.257
Kárpáti Z, Knaden M, Reinecke A, Hansson BS (2013) Intraspecific combinations of flower and leaf volatiles act together in attracting hawkmoth pollinators. PLoS ONE 8:e72805. https://doi.org/10.1371/journal.pone.0072805
Kaupp UB (2010) Olfactory signalling in vertebrates and insects: differences and commonalities. Nat Rev Neurosci 11:188–200. https://doi.org/10.1038/nrn2789
Kelling FJ, Ialenti F, Den Otter CJ (2002) Background odour induces adaptation and sensitization of olfactory receptors in the antennae of houseflies. Med Vet Entomol 16:161–169
Krieger J (1999) Olfactory reception in invertebrates. Science 286:720–723. https://doi.org/10.1126/science.286.5440.720
Lacey ES, Ray A, Cardé RT (2014) Close encounters: contributions of carbon dioxide and human skin odour to finding and landing on a host in Aedes aegypti. Physiol Entomol 39:60–68. https://doi.org/10.1111/phen.12048
Leal WS (2013) Odorant reception in insects: roles of receptors, binding proteins, and degrading enzymes. Annu Rev Entomol 58:373–391. https://doi.org/10.1146/annurev-ento-120811-153635
Legaspi JC, Simmons AM, Legaspi BC (2011) Evaluating mustard as a potential companion crop for collards to control the silverleaf whitefly, Bemisia argentifolii (Hemiptera: Aleyrodidae): olfactometer and outdoor experiments. Subtrop Plant Sci 63:36–44
Lelito JP, Myrick AJ, Baker TC (2008) Interspecific pheromone plume interference among sympatric heliothine moths: a wind tunnel test using live, calling females. J Chem Ecol 34:725–733. https://doi.org/10.1007/s10886-008-9475-6
Li FR, Kang LF, Zhang H, Zhao LY, Shirato Y, Taniyama I (2005) Changes in intensity of wind erosion at different stages of degradation development in grasslands of Inner Mongolia, China. J Arid Environ 62:567–585. https://doi.org/10.1016/j.jaridenv.2005.01.014
Linn CJ, Nojima S, Roelofs W (2005) Antagonist effects of non-host fruit volatiles on discrimination of host fruit by Rhagoletis flies infesting apple (Malus pumila), hawthorn (Crataegus spp.), and flowering dogwood (Cornus florida). Entomol Exp Appl 114:97–105
Linster C, Henry L, Kadohisa M, Wilson D (2007) Synaptic adaptation and odor-background segmentation. Neurobiol Learn Mem 87:352–360
Liu YB, Haynes KF (1992) Filamentous nature of pheromone plumes protects integrity of signal from background chemical noise in cabbage looper moth, Trichoplusia ni. J Chem Ecol 18:299–307. https://doi.org/10.1007/BF00994233
Mafra-Neto A, Cardé RT (1996) Dissection of the pheromone-modulated flight of moths using single-pulse response as a template. Experientia 52:373–379. https://doi.org/10.1007/bf01919543
Malnic B, Hirono J, Sato T, Buck LB (1999) Combinatorial receptor codes for odors. Cell 96:713–723
Mauchline AL, Osborne JL, Martin AP, Poppy GM, Powell W (2004) The effects of non-host plant essential oil volatiles on the behaviour of the pollen beetle Meligethes aeneus. Entomol Exp Appl 114:181–188
McMeniman Conor J, Corfas Román A, Matthews Benjamin J, Ritchie Scott A, Vosshall Leslie B (2014) Multimodal integration of carbon dioxide and other sensory cues drives mosquito attraction to humans. Cell 156:1060–1071. https://doi.org/10.1016/j.cell.2013.12.044
McNair C, Gries G, Gries R (2000) Cherry bark tortrix Enarmonia formosana: olfactory recognition of and behavioral deterrence by nonhost angio- and gymnosperm volatiles. J Chem Ecol 26:809–821
Miller JR, Siegert PY, Amimo FA, Walker ED (2009) Designation of chemicals in terms of the locomotor responses they elicit from insects: An update of dethier et al. (1960). Journal of Economic Entomology 102: 2056–2060 doi:https://doi.org/10.1603/029.102.0606
Moen AN (1974) Turbulence and the visualization of wind flow. Ecology 55:1420–1424
Moreau TL, Warman PR, Hoyle J (2006) An evaluation of companion planting and botanical extracts as alternative pest controls for the colorado potato beetle. Biol Agric Hortic 23:351–370. https://doi.org/10.1080/01448765.2006.9755336
Moreno CR, Racelis AE (2015) Attraction, repellence, and predation: role of companion plants in regulating Myzus persicae (Sulzer) (Hemiptera: Aphidae) in Organic Kale Systems of South Texas. Southwestern Entomologist 40:1–13
Morley K, Finch S, Collier RH (2005) Companion planting – behaviour of the cabbage root fly on host plants and non-host plants. Entomol Exp Appl 117:15–25
Mozuraitis R, Stranden M, Ramirez MI, Borg-Karlson A-K, Mustaparta H (2002) (-)-germacrene D increases attraction and oviposition by the tobacco budworm moth Heliothis virescens. Chemical Senses 27:505–509
Mumm R, Hilker M (2005) The significance of background odour for an egg parasitoid to detect plants with host eggs. Chem Senses 30:337–343. https://doi.org/10.1093/chemse/bji028
Mumm R, Hilker M (2006) Direct and indirect chemical defence of pine against folivorous insects. Trends Plant Sci 11:351–358. https://doi.org/10.1016/j.tplants.2006.05.007
Murlis J, Elkinton JS, Cardé RT (1992) Odor plumes and how insects use them. Annu Rev Entomol 37:505–532. https://doi.org/10.1146/annurev.en.37.010192.002445
Myrick AJ, Park KC, Hetling JR, Baker TC (2009) Detection and discrimination of mixed odor strands in overlapping plumes using an insect-antenna-based chemosensor system. J Chem Ecol 35:118–130. https://doi.org/10.1007/s10886-008-9582-4
Nikonov AA, Leal WS (2002) Peripheral coding of sex pheromone and a behavioral antagonist in the Japanese beetle, Popillia japonica. J Chem Ecol 28:1075–1089. https://doi.org/10.1023/a:1015274104626
Nordeus K, Webster B, Soderquist L, Bage R, Glinwood R (2014) Cycle-characteristic odour of cow urine can be detected by the female face fly (Musca autumnalis) Reproduction in domestic animals. Zuchthygiene 49:903–908. https://doi.org/10.1111/rda.12393
Nottingham SF, Hardie J, Dawson GW, Hick AJ, Pickett JA, Wadhams LJ, Woodcock CM (1991) Behavioral and electrophysiological responses of aphids to host and nonhost plant volatiles. J Chem Ecol 17:1231–1242
Omondi AB, Ghaninia M, Dawit M, Svensson T, Ignell R (2019) Age-dependent regulation of host seeking in Anopheles coluzzii. Scientific reports 9:9699. https://doi.org/10.1038/s41598-019-46220-w
Pelosi P, Iovinella I, Zhu J, Wang G, Dani FR (2018) Beyond chemoreception: diverse tasks of soluble olfactory proteins in insects. Biol Rev Camb Philos Soc 93:184–200. https://doi.org/10.1111/brv.12339
Perrin RM, Phillips ML (1978) Some effects of mixed cropping on the population dynamics of insect pests. Entomol Exp Appl 24:585–593
Perry JN, WalP C, Clark SJ (1998) Close-range behavioor of male pea moths, Cydia nigircana, responding to sex-pheromone rereleased via the substrate. Entomol Exp Appl 49:37–42
Pickett JA, Woodcock CM, Midega CAO, Khan ZR (2014) Push–pull farming systems. Curr Opin Biotechnol 26:125–132. https://doi.org/10.1016/j.copbio.2013.12.006
Plath M, Dorn S, Riedel J, Barrios H, Mody K (2011) Associational resistance and associational susceptibility: specialist herbivores show contrasting responses to tree stand diversification. Oecologia 169:477–487. https://doi.org/10.1007/s00442-011-2215-6
Raguso RA, Willis MA (2002) Synergy between visual and olfactory cues in nectar feeding by naı̈ve hawkmoths, Manduca sexta. Anim Behav 64:685–695. https://doi.org/10.1006/anbe.2002.4010
Randlkofer B, Obermaier E, Hilker M, Meiners T (2010) Vegetation complexity—The influence of plant species diversity and plant structures on plant chemical complexity and arthropods. Basic Appl Ecol 11:383–395. https://doi.org/10.1016/j.baae.2010.03.003
Riffell JA, Abrell L, Hildebrand JG (2008) Physical processes and real-time chemical measurement of the insect olfactory environment. J Chem Ecol 34:837–853. https://doi.org/10.1007/s10886-008-9490-7
Riffell JA, Lei H, Hildebrand JG (2009) Neural correlates of behavior in the moth Manduca sexta in response to complex odors. Proc Natl Acad Sci 106:19219–19226. https://doi.org/10.1073/pnas.0910592106
Rochat D, Meillour PN-L, Esteban-Duran JR, Malosse C, Perthuis B, Morin J-P, Descoins C (2000) Identification of pheromone synergists in american palm weevil, Rhynchophorus Palmarum, and attraction of related Dynamis Borassi. J Chem Ecol 26:155–187. https://doi.org/10.1023/a:1005497613214
Root RB, Kareiva PM (1984) The search for resources by cabbage butterflies (Pieris rapae): ecological consequences and adaptive significance of markovian movements in a patchy environment. Ecology 65:147–165
Røstelien T, Borg-Karlson A-K, Fäldt J, Jacobsson U, Mustaparta H (2000) The plant sesquiterpene germacrene D specifically activates a major type of antennal receptor neuron of the tobacco budworm moth Heliothis virescens. Chem Senses 25:141–148
Saïd I, Renou M, Morin J-P, Ferreira JMS, Rochat D (2005) Interactions between acetoin, a plant volatile, and pheromone In Rhynchophorus palmarum: behavioral and olfactory neuron responses. J Chem Ecol 31:1789–1805. https://doi.org/10.1007/s10886-005-5927-4
Saveer AM, Kromann SH, Birgersson G, Bengtsson M, Lindblom T, Balkenius A, Hansson BS, Witzgall P, Becher PG, Ignell R (2012) Floral to green: mating switches moth olfactory coding and preference. Proc Biol Sci 279:2314–2322. https://doi.org/10.1098/rspb.2011.2710
Schröder R, Hilker M (2008) The relevance of background odor in resource location by insects: a behavioral approach. Bioscience 58:308–316
Shrewsbury PM, Raupp MJ (2000) Evaluation of components of vegetational texture for predicting azalea lace bug, Stephanitis pyrioides (Heteroptera: Tingidae), abundance in managed landscapes. Environ Entomol 29:919–926. https://doi.org/10.1603/0046-225x-29.5.919
Sobek S, Scherber C, Steffan-Dewenter I, Tscharntke T (2009) Sapling herbivory, invertebrate herbivores and predators across a natural tree diversity gradient in Germany’s largest connected deciduous forest. Oecologia 160:279–288. https://doi.org/10.1007/s00442-009-1304-2
Späthe A, Reinecke A, Haverkamp A, Hansson BS, Knaden M (2013) Host plant odors represent immiscible information entities-blend composition and concentration matter in hawkmoths. PLoS ONE 8:e77135. https://doi.org/10.1371/journal.pone.0077135
Spitzen J, Smallegange RC, Takken W (2008) Effect of human odours and positioning of CO2 release point on trap catches of the malaria mosquito Anopheles gambiae sensu stricto in an olfactometer. Physiological Entomology 33:116–122. https://doi.org/10.1111/j.1365-3032.2008.00612.x
Spitzen J, Spoor CW, Grieco F, ter Braak C, Beeuwkes J, van Brugge SP, Kranenbarg S, Noldus LPJJ, van Leeuwen JL, Takken W (2013) A 3D analysis of flight behavior of anopheles gambiae sensu stricto malaria mosquitoes in response to human odor and heat. PLoS ONE 8:e62995. https://doi.org/10.1371/journal.pone.0062995
Stoeckli S, Mody K, Dorn S (2008) Influence of canopy aspect and height on codling moth (Lepidoptera: Tortricidae) larval infestation in apple, and relationship between infestation and fruit size. J Econ Entomol 101:81–89. https://doi.org/10.1093/jee/101.1.81
Su CY, Martelli C, Emonet T, Carlson JR (2011) Temporal coding of odor mixtures in an olfactory receptor neuron. Proc Natl Acad Sci 108:5075–5080. https://doi.org/10.1073/pnas.1100369108
Tasin M, Backman AC, Bengtsson M, Ioriatti C, Witzgall P (2006) Essential host plant cues in the grapevine moth. Naturwissenschaften 93:141–144. https://doi.org/10.1007/s00114-005-0077-7
Thiery D, Visser JH (1986) Masking of host plant odour in the olfactory orientation of the Colorado potato beetle. Entomol Exp Appl 41:165–172
Thiery D, Visser JH (1987) Misleading the Colorado potato beetle with an odor blend. J Chem Ecol 13:1139–1146. https://doi.org/10.1007/bf01020544
Turner SL, Ray A (2009) Modification of CO2 avoidance behaviour in Drosophila by inhibitory odorants. Nature 461:277–281
Vargas RI, Pinero JC, Miller NW (2018) Effect of physiological state on female melon fly (Diptera: Tephritidae) attraction to host and food odor in the field. J Econ Entomol 111:1318–1322. https://doi.org/10.1093/jee/toy092
Vehvilainen H, Koricheva J, Ruohomaki K (2007) Tree species diversity influences herbivore abundance and damage: meta-analysis of long-term forest experiments. Oecologia 152:287–298. https://doi.org/10.1007/s00442-007-0673-7
Visser J (1986) Host odor perception in phytophagous insects. Annu Rev Entomol 31:121–144. https://doi.org/10.1146/annurev.en.31.010186.001005
Visser JH, Ave DA (1978) General green leaf volatiles in the olfactory orientation of the Colorado beetle, Leptinotarsa decemlineata. Entomol Exp Appl 24:738–749
Webster B, Cardé RT (2017) Use of habitat odour by host-seeking insects. Biol Rev Camb Philos Soc 92:1241–1249. https://doi.org/10.1111/brv.12281
Webster B, Bruce T, Dufour S, Birkemeyer C, Birkett M, Hardie J, Pickett J (2008) Identification of volatile compounds used in host location by the black bean aphid, Aphis fabae. J Chem Ecol 34:1153–1161. https://doi.org/10.1007/s10886-008-9510-7
Webster B, Bruce T, Pickett J, Hardie J (2010a) Volatiles functioning as host cues in a blend become nonhost cues when presented alone to the black bean aphid. Anim Behav 79:451–457. https://doi.org/10.1016/j.anbehav.2009.11.028
Webster B, Gezan S, Bruce T, Hardie J, Pickett J (2010b) Between plant and diurnal variation in quantities and ratios of volatile compounds emitted by Vicia faba plants. Phytochemistry 71:81–89. https://doi.org/10.1016/j.phytochem.2009.09.029
Webster B, Lacey ES, Cardé RT (2015) Waiting with bated breath: opportunistic orientation to human odor in the malaria mosquito, Anopheles gambiae, is modulated by minute changes in carbon dioxide concentration. J Chem Ecol 41:59–66. https://doi.org/10.1007/s10886-014-0542-x
Whicker JJ, Breshears DD, Wasiolek PT, Kirchner TB, Tavani RA, Schoep DA, Rodgers JC (2002) Temporal and spatial variation of episodic wind erosion in unburned and burned semiarid shrubland. J Environ Qual 31:599–612. https://doi.org/10.2134/jeq2002.0599
Wicher D (2018) Tuning insect odorant receptors. Front Cell Neurosci 12:94. https://doi.org/10.3389/fncel.2018.00094
Wilson RI, Mainen ZF (2006) Early events in olfactory processing. Annu Rev Neurosci 29:163–201. https://doi.org/10.1146/annurev.neuro.29.051605.112950
Witzgall P, Priesner E (1991) Wind tunnel study on an attraction inhibitor in male Coleophora laricella Hbn. (Lepidoptera: Coleophoridae). J Chem Ecol 17:1355–1362
Wolf H, Wehner R (2000) Pinpointing food sources: olfactory and anemotactic orientation in desert ants, Cataglyphis fortis. J Exp Biol 203:857–868
Wolfin MS, Chilson RR, Thrall J, Liu Y, Volo S, Cha DH, Loeb GM, Linn CE (2019) Proximate mechanisms of host plant location by a specialist phytophagous insect, the grape berry moth, Paralobesia viteana. J Chem Ecol 45:946–958. https://doi.org/10.1007/s10886-019-01112-1
Yamasaki T, Sato M, Sakoguchi H (1997) -)-Germacrene D: masking substance of attractants for the cerambycid beetle, Monochamus alternatus (Hope). Appl Entomol Zool 32:423–429. https://doi.org/10.1303/aez.32.423
Acknowledgements
We thank Professor Mingxian Yang and Professor Lijun Wang for their valuable comments on this manuscript. We thank several anonymous reviewers for giving helpful suggestions that improved the manuscript. This study was supported by the National Key Research and Development Project of China (2018YFD060024-04).
Funding
This study was supported by the National Key Research and Development Project of China (2018YFD060024-04).
Author information
Authors and Affiliations
Contributions
Xinliang Shao, Ke Cheng, Zhengwei Wang and Qin Zhang conceived the ideas and wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Consent to participate
The authors obtained consent from the university to participate in this work.
Consent for publication
The authors and the university where they work agree to submit the manuscript.
Ethics approval
Not applicable.
Additional information
Communicated by Günther Raspotnig.
Rights and permissions
About this article
Cite this article
Shao, X., Cheng, K., Wang, Z. et al. Use of odor by host-finding insects: the role of real-time odor environment and odor mixing degree. Chemoecology 31, 149–158 (2021). https://doi.org/10.1007/s00049-021-00342-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00049-021-00342-8