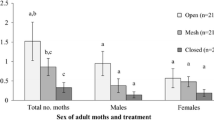
Abstract
There are contrasting hypotheses regarding the role of plant volatiles in host plant location. We used the grape berry moth (GBM; Paralobesia viteana)-grape plant (Vitis spp.) complex as a model for studying the proximate mechanisms of long distance olfactory-mediated, host-plant location and selection by a specialist phytophagous insect. We used flight tunnel assays to observe GBM female in-flight responses to host (V. riparia) and non-host (apple, Malus domestica; and gray dogwood, Cornus racimosa,) odor sources in the form of plant shoots, extracts of shoots, and synthetic blends. Gas chromatography-electroantennographic detection and gas chromatography/mass spectrometry analyses were used to identify antennal-active volatile compounds. All antennal-active compounds found in grape shoots were also present in dogwood and apple shoots. Female GBM flew upwind to host and non-host extracts and synthetic blends at similar levels, suggesting discrimination is not occurring at long distance from the plant. Further, females did not land on sources releasing plant extracts and synthetic blends, suggesting not all landing cues were present. Additionally, mated and unmated moths displayed similar levels of upwind flight responses to all odor sources, supporting the idea that plant volatiles are not functioning solely as ovipositional cues. The results of this study support a hypothesis that GBM females are using volatile blends to locate a favorable habitat rather than a specific host plant, and that discrimination is occurring within the habitat, or even post-landing.





Similar content being viewed by others
References
Ahuja I, Rohloff J, Bones AM (2009) Defence mechanisms of brassicaceae: implications for plant-insect interactions and potential for integrated pest management. Sustain Agric 30:623–670. https://doi.org/10.1007/978-94-007-0394-0_28
Bell WJ (1990) Searching behavior patterns in insects. Annu Rev Entomol 35:447–467. https://doi.org/10.1146/annurev.en.35.010190.002311
Blaney WM, Simmonds MSJ (1990) A behavioural and electrophysiological study of the role of tarsal chemoreceptors in feeding by adults of Spodoptera, Heliothis virescens and Helicoverpa armigera. J Insect Physiol 36. https://doi.org/10.1016/0022-1910(90)90048-K
Brantjes NBM (1973) Sphingophilous flowers, function of their scent. In: Brantjes NBM, Linskens HF (eds) Pollination and dispersal. University of Nijmegan, Nijmegan, Netherlands, pp 27–46
Bruce TJ, Pickett JA (2011) Perception of plant volatile blends by herbivorous insects-finding the right mix. Phytochemistry 72:1605–1611. https://doi.org/10.1016/j.phytochem.2011.04.011
Bruce T, Wadhams L, Woodcock C (2005) Insect host location: a volatile situation. Trends Plant Sci 10:269–274. https://doi.org/10.1016/j.tplants.2005.04.003
Cha DH, Hesler SP, Moser CL, Nojima S, Linn, Charles E. Jr, Roelofs WL, Loeb GM (2008a) Flight tunnel responses of female grape berry moth (Paralobesia viteana) to host plants. J Chem Ecol 34:622–627. https://doi.org/10.1007/s10886-008-9474-7
Cha DH, Nojima S, Hesler SP, Zhang A, Linn CE Jr, Roelofs WL, Loeb GM (2008b) Identification and field evaluation of grape shoot volatiles attractive to female grape berry moth (Paralobesia viteana). J Chem Ecol 34:1180–1189. https://doi.org/10.1007/s10886-008-9517-0
Cha DH, Linn CE, Teal PE et al (2011) Eavesdropping on plant volatiles by a specialist moth: significance of ratio and concentration. PLoS One 6:e17033. https://doi.org/10.1371/journal.pone.0017033
Clark LG, Dennehy TJ (1988) Oviposition behavior of grape berry moth. Entomol Exp Appl 47:223–230. https://doi.org/10.1111/j.1570-7458.1988.tb01140.x
Clifford MR, Riffell JA (2013) Mixture and odorant processing in the olfactory systems of insects: a comparative perspective. J Comp Physiol A Neuroethol Sens Neural Behav Physiol 199:911–928. https://doi.org/10.1007/s00359-013-0818-6
Cole PG, Weltzin JF (2005) Light limitation creates patchy distribution of an invasive grass in eastern deciduous forests. Biol Invasions 7:477–488. https://doi.org/10.1007/s10530-004-5171-9
Contreras HL, Goyret J, von Arx M, Pierce CT, Bronstein JL, Raguso RA, Davidowitz G (2013) The effect of ambient humidity on the foraging behavior of the hawkmoth Manduca sexta. J Comp Physiol A Neuroethol Sens Neural Behav Physiol 199:1053–1063. https://doi.org/10.1007/s00359-013-0829-3
Dethier V (1941) The function of the antennal receptors in lepidopterous larvae. Biol Bull 80:403–414
Döring TF (2014) How aphids find their host plants, and how they don’t. Ann Appl Biol 165:3–26. https://doi.org/10.1111/aab.12142
Droney DC, Musto CJ, Mancuso K, Roelofs WL, Linn CE Jr (2012) The response to selection for broad male response to female sex pheromone and its implications for divergence in close-range mating behavior in the European corn borer moth, Ostrinia nubilalis. J Chem Ecol 38:1504–1512. https://doi.org/10.1007/s10886-012-0208-5
Ephrussi B, Beadle GW (1936) A technique of transplantation for Drosophila. Am Soc Nat 13:229–246
Faucher CP, Hilker M, de Bruyne M (2013) Interactions of carbon dioxide and food Odours in Drosophila: olfactory hedonics and sensory neuron properties. PLoS One 8. https://doi.org/10.1371/journal.pone.0056361
Finch S, Collier RH (2000) Host-plant selection by insects - a theory based on “appropriate/inappropriate landings” by pest insects of cruciferous plants. Entomol Exp Appl 96:91–102. https://doi.org/10.1046/j.1570-7458.2000.00684.x
Fraenkel GS (1959) The raison d ’ Etre substances of secondary plant substances. Science (80- ) 129:1466–1470
Galiano EF (1985) The small-scale pattern of Cynodon dactylon in Mediterranean pastures. Vegetatio 63:121–127. https://doi.org/10.1007/BF00044062
Hendrikse A, Vos-Bünnemeyer E (1987) Role of host-plant stimuli in sexual behaviour of small ermine moths (Yponomeuta). Ecol Entomol 12:363–371. https://doi.org/10.1111/j.1365-2311.1987.tb01017.x
Janzen DH (1987) How moths pass the dry season in a Costa Rican dry Forest. Insect Sci Its Appl 8:489–500. https://doi.org/10.1017/S1742758400022530
Karpati Z, Tasin M, Carde RT, Dekker T (2013) Early quality assessment lessens pheromone specificity in a moth. Proc Natl Acad Sci 110:7377–7382. https://doi.org/10.1073/pnas.1216145110
Keller M (2015) Botany and anatomy. In: The science of grapevines, 2nd edn, pp 1–57
Kostal VI, Finch S (1994) Influence of background on host-plant selection and subsequent oviposition by the cabbage root fly (Delia radicum). Entomol Exp Appl 70:153–163
Landolt PJ, Phillips TW (1997) Host plant influences on sex pheromone behavior of phytophagous insects. Annu Rev Entomol 42:371–391. https://doi.org/10.1146/annurev.ento.42.1.371
Landolt PJ, Heath RR, Millar JG, Davis-Hernandez KM, Dueben BD, Ward KE (1994) Effects of host plant, Gossypium hirsutum, on sexual attraction of cabbage looper moths, Trichoplusia (Hubner) (Lepidoptera: Noctuidae). J Chem Ecol 20:2959–2974
Linn C, O’Connor M, Roelofs W (2003a) Silent genes and rare males: a fresh look at pheromone blend response specificity in the European corn borer moth, Ostrinia nubilalis. J Insect Sci 3:15. https://doi.org/10.1093/jis/3.1.15
Linn CE, Feder JL, Nojima S et al (2003b) Fruit odor discrimination and sympatric host race formation in Rhagoletis. Proc Natl Acad Sci U S A 100:11490–11493. https://doi.org/10.1073/pnas.1635049100
Linn CE, Dambroski H, Nojima S et al (2005) Variability in response specificity of apple, hawthorn, and flowering dogwood-infesting Rhagoletis flies to host fruit volatile blends: implications for sympatric host shifts. Entomol Exp Appl 116:55–64. https://doi.org/10.1111/j.1570-7458.2005.00310.x
Linn CE, Yee WL, Sim SB et al (2012) Behavioral evidence for fruit odor discrimination and sympatric host races of Rhagoletis Pomonella flies in the Western United States. Evolution (N Y) 66:3632–3641. https://doi.org/10.1111/j.1558-5646.2012.01719.x
Martin N, Moore K, Musto CJ, Linn CE (2016) Flight tunnel response of male European corn borer moths to cross-specific mixtures of European and Asian corn borer sex pheromones: evidence supporting a critical stage in evolution of a new communication system. J Chem Ecol 42:51–54. https://doi.org/10.1007/s10886-015-0656-9
Meiners T (2015) Chemical ecology and evolution of plant-insect interactions: a multitrophic perspective. Curr Opin Insect Sci 8:22–28. https://doi.org/10.1016/j.cois.2015.02.003
Miller JR, Strickler KL (1984) Finding and Accepting Host Plants. In: Bell WJ, Cardé RT (eds) Chemical Ecology of Insects. Springer, Boston, MA
Mozuraitis R, Stranden M, Ramirez MI, Borg-Karlson AK, Mustaparta H (2002) (-)-Germacrene D increases attraction and oviposition by the tobacco budworm moth Heliothis virescens. Chem Senses 27:505–509. https://doi.org/10.1093/chemse/27.6.505
Nagarkatti S, Muza A, Saunders M (2000) Meridic diet for Endopiza viteana (Lepidoptera: Tortricidae). Can Entomol 132:259–261. https://doi.org/10.4039/Ent132259-2
Nojima S, Linn CE, Morris B (2003a) Identification of host fruit volatiles from hawthorn (Crataegus spp.) attractive to hawthorn-origin Rhagoletis pomonella flies. J Chem Ecol 29:321–336
Nojima S, Linn CE, Morris B et al (2003b) Identification of host fruit volatiles from flowering dogwood (Cornus florida) attractive to dogwood-origin Rhagoletis pomonella flies. J Chem Ecol 29:321–336
Pickett J (1992) The chemical ecology of aphids. Annu Rev Entomol 37:67–90. https://doi.org/10.1146/annurev.ento.37.1.67
Powell THQ, Cha DH, Linn CE, Feder JL (2012) On the scent of standing variation for speciation: Beehavioral evidence for native sympatric host races of Rhagoletis pomonella (Diptera: tephritidae) in the southern United States. Evolution (N Y) 66:1215–1221. https://doi.org/10.1111/j.1558-5646.2012.01625.x
Prokopy R, Collier R, Finch S (1983) Visual detection of host plants by cabbage root flies. Entomol Exp Appl 34:85–89
Raina AK, Kingan TG, Mattoo AK (1992) Chemical signals from host plant and sexual behavior in a moth. Science (80- ) 255:592–594
Roessingh P, Städler E, Fenwick GR et al (1992) Oviposition and tarsal chemoreceptors of the cabbage root fly are stimulated by glucosinlolates and host plant-extracts. Entomol Exp Appl 65:267–282. https://doi.org/10.1111/j.1570-7458.1992.tb00680.x
Rojas JC, Wyatt TD (1999) Role of visual cues and interaction with host odour during the host-finding behaviour of the cabbage moth. Entomol Exp Appl 91:59–65. https://doi.org/10.1023/A:1003605125191
Schoonhoven LM, van Loon JJA, Dicke M (2005) Insect-plant biology, 2nd edn. Oxford University Press, Oxford
Shorey HH (1974) Environmental and physiological control of insect sex pheromone behaviour. In: Birch MC (ed) Pheromones. North-Holland Publishing Company, New York, pp 68–20
Stadler E, Renwick JAA, Radke CD, Sachdev-Gupta K (1995) Tarsal contact chemoreceptor response to glucosinolates and cardenolides mediating oviposition in Pieris rapae. Physiol Entomol 20:175–187. https://doi.org/10.1111/j.1365-3032.1995.tb00814.x
Taschenberg E, Cardé R, Hill A et al (1974) Sex pheromone trapping of the grape berry moth. Environ Entomol 3:1973–1975
Tasin M, Bäckman A, Anfora G (2009) Attraction of female grapevine moth to common and specific olfactory cues from 2 host plants. Chem Senses 35:57–64. https://doi.org/10.1093/chemse/bjp082
Thiéry D, Moreau J (2005) Relative performance of European grapevine moth (Lobesia botrana) on grapes and other hosts. Oecologia 143:548–557. https://doi.org/10.1007/s00442-005-0022-7
Visser J (1986) Host odor perception in Phytophagous insects. Annu Rev Entomol 31:121–144. https://doi.org/10.1146/annurev.ento.31.1.121
Visser J (1988) Host-plant finding by insects: orientation, sensory input and search patterns. J Insect Physiol 34:259–268
von Arx M, Goyret J, Davidowitz G, Raguso R (2012) Floral humidity as a reliable sensory cue for profitability assessment by nectar-foraging hawkmoths. Proc Natl Acad Sci U S A 109:9471–9476. https://doi.org/10.1073/pnas.1121624109
Webster B, Cardé RT (2016) Use of habitat odour by host-seeking insects. Biol Rev 92:1241–1249. https://doi.org/10.1111/brv.12281
Webster B, Bruce T, Dufour S, Birkemeyer C, Birkett M, Hardie J, Pickett J (2008) Identification of volatile compounds used in host location by the black bean aphid, Aphis fabae. J Chem Ecol 34:1153–1161. https://doi.org/10.1007/s10886-008-9510-7
Williamson J, Johnson D (2005) Effects of grape berry moth management practices and landscape on arthropod diversity in grape vineyards in the southern United States. Horttechnology 15:232–238
Wolfin MS, Raguso RA, Davidowitz G, Goyret J (2018) Context-dependency of in-flight responses by Manduca sexta moths to ambient differences in relative humidity. J Exp Biol. https://doi.org/10.1242/jeb.177774
Acknowledgements
We thank Shinyoung Park, Callie Musto, and Stephen Hesler for help maintaining the greenhouse, GBM colonies, and for setting up cohorts for flight tunnel tests. We thank Stephen Parry at the Cornell University Statistical Consulting Unit for his statistical guidance. Sara Volo, Yuxi Liu, and Jonathan Thrall were undergraduates at Hobart and William Smith Colleges, Geneva, NY participating in a Summer Scholars Program supported by funding from the David and Brenda Rickey Foundation. We also thank Paul Robbins for his support, advice, and optimism regarding GC-EAD problem solving. We thank the Chong Lab for providing the dimethyl-1,3(E),7-nonatriene. The research was supported by a USDA-AFRI proposal # 2012-67013-19364, and a USDA Federal Formula Fund Initiative #2014-15-154.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wolfin, M.S., Chilson, R.R., Thrall, J. et al. Proximate Mechanisms of Host Plant Location by a Specialist Phytophagous Insect, the Grape Berry Moth, Paralobesia Viteana. J Chem Ecol 45, 946–958 (2019). https://doi.org/10.1007/s10886-019-01112-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-019-01112-1