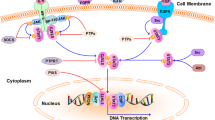
Abstract
As the most prevalent form of cancer found in women, breast cancer is an active area of research and clinical study. Treatments for cancer patients have traditionally relied upon chemotherapy, but an increasing emphasis is placed on targeted therapies as a safer and more effective way to treat cancer. Therapies that target specific aspects of cancerous cells may be especially significant for triple negative breast cancer (TNBC), which is often associated with poor patient prognosis and currently lacks reliable targeted treatment options. Many therapies for breast cancer have been extensively studied and examined in clinical settings, while others show future potential and require further investigation. This chapter will focus on six targeted therapies, describing mechanisms of action and current methods of inhibition. Inducible nitric oxide synthase (iNOS) overexpression is correlated with increased cell proliferation and mammosphere production, while inhibition has been shown to minimize these effects in cancerous cells. The PI3K pathway is an essential component of intracellular signaling that is frequently dysregulated in tumors. Poly(ADP-ribose) polymerases (PARP) are implicated in DNA damage repair, and are the focus of numerous ongoing clinical trials. Protein tyrosine kinase 6 (PTK6) has been associated with cell proliferation and tumor growth in breast cancer cells, while the serine/threonine cyclin-dependent kinase (CDK) protein family is a crucial component of cell cycle regulation and an important area of research for targeted therapies. Finally, aberrant activation of the Wnt/β-catenin signaling pathway has been implicated in multiple breast cancer subtypes, particularly with respect to the prevalence β-catenin mutations in TNBC. These pathways represent promising topics in the field of targeted therapies, and with increased research may contribute to the development of superior and precise treatments for breast cancer.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- ABC:
-
ATP-binding cassette
- APC:
-
Adenomatous polyposis coli
- CDK:
-
Cyclin-dependent kinase
- CK1α:
-
Casein kinase-1α
- CSC:
-
Cancer stem cell
- DSB:
-
Double-stranded break
- DVL:
-
Dishevelled
- EGF:
-
Epidermal growth factor
- EGFR:
-
Epidermal growth factor receptor
- EMT:
-
Epithelial-mesenchymal transition
- FAK:
-
Focal adhesion kinase
- FOXM1:
-
Forkhead box M1
- FZD:
-
Frizzled
- GSK3β:
-
Glycogen synthase kinase 3β
- HER2:
-
Human epidermal growth factor 2
- HR:
-
Homologous recombination
- HR+:
-
Hormone receptor-positive
- iNOS:
-
Inducible nitric oxide synthase
- NOS:
-
Nitric oxide synthase
- NSAID:
-
Nonsteroidal anti-inflammatory drug
- PARP:
-
Poly(ADP-Ribose) polymerase
- PARPi:
-
PARP inhibitor
- PI3K:
-
Phosphoinositide 3-kinase
- Prcn:
-
Porcupine
- PTEN:
-
Phosphatase and tensin homolog
- PTK6:
-
Protein tyrosine kinase 6
- Rb:
-
Retinoblastoma
- RTK:
-
Receptor tyrosine kinase
- SSD:
-
Single-stranded break
- STAT:
-
Signal transducer and activator of transcription
- TCF/LEF:
-
T-cell factor/lymphoid enhancer factor
- TNBC:
-
Triple-negative breast cancer
References
Granados-Principal S, Liu Y, Guevara ML, Blanco E, Choi DS, Qian W, Patel T, Rodriguez AA, Cusimano J, Weiss HL, Zhao H, Landis MD, et al. Inhibition of iNOS as a novel effective targeted therapy against triple-negative breast cancer. Breast Cancer Res: BCR. 2015;17:25.
Glynn SA, Boersma BJ, Dorsey TH, Yi M, Yfantis HG, Ridnour LA, Martin DN, Switzer CH, Hudson RS, Wink DA, Lee DH, Stephens RM, et al. Increased NOS2 predicts poor survival in estrogen receptor-negative breast cancer patients. J Clin Invest. 2010;120:3843–54.
Jadeski LC, Chakraborty C, Lala PK. Nitric oxide-mediated promotion of mammary tumour cell migration requires sequential activation of nitric oxide synthase, guanylate cyclase and mitogen-activated protein kinase. Int J Cancer. 2003;106:496–504.
Rudge SA, Wakelam MJ. Phosphatidylinositolphosphate phosphatase activities and cancer. J Lipid Res. 2016;57:176–92.
Yuan TL, Cantley LC. PI3K pathway alterations in cancer: variations on a theme. Oncogene. 2008;27:5497–510.
Vanhaesebroeck B, Guillermet-Guibert J, Graupera M, Bilanges B. The emerging mechanisms of isoform-specific PI3K signalling. Nat Rev Mol Cell Biol. 2010;11:329–41.
Wang X, Ding J, Meng LH. PI3K isoform-selective inhibitors: next-generation targeted cancer therapies. Acta Pharmacol Sin. 2015;36:1170–6.
Barnes KR, Blois J, Smith A, Yuan H, Reynolds F, Weissleder R, Cantley LC, Josephson L. Fate of a bioactive fluorescent wortmannin derivative in cells. Bioconjug Chem. 2008;19:130–7.
Chen JS, Zhou LJ, Entin-Meer M, Yang X, Donker M, Knight ZA, Weiss W, Shokat KM, Haas-Kogan D, Stokoe D. Characterization of structurally distinct, isoform-selective phosphoinositide 3′-kinase inhibitors in combination with radiation in the treatment of glioblastoma. Mol Cancer Ther. 2008;7:841–50.
Slamon D, Eiermann W, Robert N, Pienkowski T, Martin M, Press M, Mackey J, Glaspy J, Chan A, Pawlicki M, Pinter T, Valero V, et al. Adjuvant trastuzumab in HER2-positive breast cancer. N Engl J Med. 2011;365:1273–83.
Berns K, Horlings HM, Hennessy BT, Madiredjo M, Hijmans EM, Beelen K, Linn SC, Gonzalez-Angulo AM, Stemke-Hale K, Hauptmann M, Beijersbergen RL, Mills GB, et al. A functional genetic approach identifies the PI3K pathway as a major determinant of trastuzumab resistance in breast cancer. Cancer Cell. 2007;12:395–402.
Elster N, Cremona M, Morgan C, Toomey S, Carr A, O’Grady A, Hennessy BT, Eustace AJ. A preclinical evaluation of the PI3K alpha/delta dominant inhibitor BAY 80-6946 in HER2-positive breast cancer models with acquired resistance to the HER2-targeted therapies trastuzumab and lapatinib. Breast Cancer Res Treat. 2015;149:373–83.
Livraghi L, Garber JE. PARP inhibitors in the management of breast cancer: current data and future prospects. BMC Med. 2015;13:188.
Helleday T. The underlying mechanism for the PARP and BRCA synthetic lethality: clearing up the misunderstandings. Mol Oncol. 2011;5:387–93.
Bouwman P, Jonkers J. Molecular pathways: how can BRCA-mutated tumors become resistant to PARP inhibitors? Clin Cancer Res. 2014;20:540–7.
Gilardini Montani MS, Prodosmo A, Stagni V, Merli D, Monteonofrio L, Gatti V, Gentileschi MP, Barila D, Soddu S. ATM-depletion in breast cancer cells confers sensitivity to PARP inhibition. J Exp Clin Cancer Res. 2013;32:95.
Nagasawa S, Sedukhina AS, Nakagawa Y, Maeda I, Kubota M, Ohnuma S, Tsugawa K, Ohta T, Roche-Molina M, Bernal JA, Narvaez AJ, Jeyasekharan AD, et al. LSD1 overexpression is associated with poor prognosis in basal-like breast cancer, and sensitivity to PARP inhibition. PLoS One. 2015;10:e0118002.
van der Noll R, Marchetti S, Steeghs N, Beijnen JH, Mergui-Roelvink MW, Harms E, Rehorst H, Sonke GS, Schellens JH. Long-term safety and anti-tumour activity of olaparib monotherapy after combination with carboplatin and paclitaxel in patients with advanced breast, ovarian or fallopian tube cancer. Br J Cancer. 2015;113:396–402.
Isakoff SJ. PARP inhibition outside of BRCA mutation carriers. Clin Adv Hematol Oncol. 2010;8:757–8.
Brauer PM, Tyner AL. Building a better understanding of the intracellular tyrosine kinase PTK6 - BRK by BRK. Biochim Biophys Acta. 2010;1806:66–73.
Vasioukhin V, Serfas MS, Siyanova EY, Polonskaia M, Costigan VJ, Liu B, Thomason A, Tyner AL. A novel intracellular epithelial cell tyrosine kinase is expressed in the skin and gastrointestinal tract. Oncogene. 1995;10:349–57.
Kamalati T, Jolin HE, Mitchell PJ, Barker KT, Jackson LE, Dean CJ, Page MJ, Gusterson BA, Crompton MR. Brk, a breast tumor-derived non-receptor protein-tyrosine kinase, sensitizes mammary epithelial cells to epidermal growth factor. J Biol Chem. 1996;271:30956–63.
Chen HY, Shen CH, Tsai YT, Lin FC, Huang YP, Chen RH. Brk activates rac1 and promotes cell migration and invasion by phosphorylating paxillin. Mol Cell Biol. 2004;24:10558–72.
Groner B, Lucks P, Borghouts C. The function of Stat3 in tumor cells and their microenvironment. Semin Cell Dev Biol. 2008;19:341–50.
Walker SR, Nelson EA, Zou L, Chaudhury M, Signoretti S, Richardson A, Frank DA. Reciprocal effects of STAT5 and STAT3 in breast cancer. Mol Cancer Res. 2009;7:966–76.
Zheng Y, Gierut J, Wang Z, Miao J, Asara JM, Tyner AL. Protein tyrosine kinase 6 protects cells from anoikis by directly phosphorylating focal adhesion kinase and activating AKT. Oncogene. 2013;32:4304–12.
Irie HY, Shrestha Y, Selfors LM, Frye F, Iida N, Wang Z, Zou L, Yao J, Lu Y, Epstein CB, Natesan S, Richardson AL, et al. PTK6 regulates IGF-1-induced anchorage-independent survival. PLoS One. 2010;5:e11729.
Park SH, Ito K, Olcott W, Katsyv I, Halstead-Nussloch G, Irie HY. PTK6 inhibition promotes apoptosis of Lapatinib-resistant Her2(+) breast cancer cells by inducing Bim. Breast Cancer Res: BCR. 2015;17:86.
Ito K, Park SH, Nayak A, Byerly JH, Irie HY. PTK6 inhibition suppresses metastases of triple-negative breast cancer via SNAIL-dependent E-cadherin regulation. Cancer Res. 2016;76:4406–17.
Nahta R, Esteva FJ. Trastuzumab: triumphs and tribulations. Oncogene. 2007;26:3637–43.
Hamilton E, Infante JR. Targeting CDK4/6 in patients with cancer. Cancer Treat Rev. 2016;45:129–38.
Nichols M. New directions for drug-resistant breast cancer: the CDK4/6 inhibitors. Future Med Chem. 2015;7:1473–81.
Cancer Genome Atlas Network. Comprehensive molecular portraits of human breast tumours. Nature. 2012;490:61–70.
Baker SJ, Reddy EP. CDK4: a key player in the cell cycle, development, and cancer. Genes Cancer. 2012;3:658–69.
Shapiro GI. Cyclin-dependent kinase pathways as targets for cancer treatment. J Clin Oncol: Off J Am Soc Clin Oncol. 2006;24:1770–83.
Xu H, Yu S, Liu Q, Yuan X, Mani S, Pestell RG, Wu K. Recent advances of highly selective CDK4/6 inhibitors in breast cancer. J Hematol Oncol. 2017;10:97.
Rihani A, Vandesompele J, Speleman F, Van Maerken T. Inhibition of CDK4/6 as a novel therapeutic option for neuroblastoma. Cancer Cell Int. 2015;15:76.
McCain J. First-in-class CDK4/6 inhibitor Palbociclib could usher in a new wave of combination therapies for HR+, HER2− breast cancer. Pharm Ther. 2015;40:511–20.
Anders L, Ke N, Hydbring P, Choi YJ, Widlund HR, Chick JM, Zhai H, Vidal M, Gygi SP, Braun P, Sicinski P. A systematic screen for CDK4/6 substrates links FOXM1 phosphorylation to senescence suppression in cancer cells. Cancer Cell. 2011;20:620–34.
Hortobagyi GN, Stemmer SM, Burris HA, Yap YS, Sonke GS, Paluch-Shimon S, Campone M, Blackwell KL, Andre F, Winer EP, Janni W, Verma S, et al. Ribociclib as first-line therapy for HR-positive, advanced breast cancer. N Engl J Med. 2016;375:1738–48.
Vora SR, Juric D, Kim N, Mino-Kenudson M, Huynh T, Costa C, Lockerman EL, Pollack SF, Liu M, Li X, Lehar J, Wiesmann M, et al. CDK 4/6 inhibitors sensitize PIK3CA mutant breast cancer to PI3K inhibitors. Cancer Cell. 2014;26:136–49.
Fujiwara Y, Tamura K, Kondo S, Tanabe Y, Iwasa S, Shimomura A, Kitano S, Ogasawara K, Turner PK, Mori J, Asou H, Chan EM, et al. Phase 1 study of abemaciclib, an inhibitor of CDK 4 and 6, as a single agent for Japanese patients with advanced cancer. Cancer Chemother Pharmacol. 2016;78:281–8.
Patnaik A, Rosen LS, Tolaney SM, Tolcher AW, Goldman JW, Gandhi L, Papadopoulos KP, Beeram M, Rasco DW, Hilton JF, Nasir A, Beckmann RP, et al. Efficacy and safety of abemaciclib, an inhibitor of CDK4 and CDK6, for patients with breast cancer, non-small cell lung cancer, and other solid tumors. Cancer Discov. 2016;6:740–53.
Thangavel C, Dean JL, Ertel A, Knudsen KE, Aldaz CM, Witkiewicz AK, Clarke R, Knudsen ES. Therapeutically activating RB: reestablishing cell cycle control in endocrine therapy-resistant breast cancer. Endocr Relat Cancer. 2011;18:333–45.
McClendon AK, Dean JL, Rivadeneira DB, JE Y, Reed CA, Gao E, Farber JL, Force T, Koch WJ, Knudsen ES. CDK4/6 inhibition antagonizes the cytotoxic response to anthracycline therapy. Cell Cycle. 2012;11:2747–55.
Gogolin S, Ehemann V, Becker G, Brueckner LM, Dreidax D, Bannert S, Nolte I, Savelyeva L, Bell E, Westermann F. CDK4 inhibition restores G(1)-S arrest in MYCN-amplified neuroblastoma cells in the context of doxorubicin-induced DNA damage. Cell Cycle. 2013;12:1091–104.
Prosperi JR, Goss KH. A Wnt-ow of opportunity: targeting the Wnt/beta-catenin pathway in breast cancer. Curr Drug Targets. 2010;11:1074–88.
Ahmed K, Shaw HV, Koval A, Katanaev VL. A second WNT for old drugs: drug repositioning against WNT-dependent cancers. Cancers (Basel). 2016;8:66.
Khramtsov AI, Khramtsova GF, Tretiakova M, Huo D, Olopade OI, Goss KH. Wnt/beta-catenin pathway activation is enriched in basal-like breast cancers and predicts poor outcome. Am J Pathol. 2010;176:2911–20.
Karim R, Tse G, Putti T, Scolyer R, Lee S. The significance of the Wnt pathway in the pathology of human cancers. Pathology. 2004;36:120–8.
Kahn M. Can we safely target the WNT pathway? Nat Rev Drug Discov. 2014;13:513–32.
Liu J, Pan S, Hsieh MH, Ng N, Sun F, Wang T, Kasibhatla S, Schuller AG, Li AG, Cheng D, Li J, Tompkins C, et al. Targeting Wnt-driven cancer through the inhibition of porcupine by LGK974. Proc Natl Acad Sci U S A. 2013;110:20224–9.
Barker N, Clevers H. Mining the Wnt pathway for cancer therapeutics. Nat Rev Drug Discov. 2006;5:997–1014.
Solzak JP, Atale RV, Hancock BA, Sinn AL, Pollok KE, Jones DR, Radovich M. Dual PI3K and Wnt pathway inhibition is a synergistic combination against triple negative breast cancer. NPJ Breast Cancer. 2017;3:17.
Lu W, Lin C, Roberts MJ, Waud WR, Piazza GA, Li Y. Niclosamide suppresses cancer cell growth by inducing Wnt co-receptor LRP6 degradation and inhibiting the Wnt/beta-catenin pathway. PLoS One. 2011;6:e29290.
Naujokat C, Steinhart R. Salinomycin as a drug for targeting human cancer stem cells. J Biomed Biotechnol. 2012;2012:950658.
Stefanski CD, Prosperi JR. 2018. Role of Wnt/β -catenin pathway in cancer signaling. In: Predictive Biomarkers in Oncology: Applications in Precision Medicine (Eds: George Kumar and Sunil Badve). In press.
Acknowledgements
The authors thank Ms. Casey Stefanski for critical review of the document. Work in the Prosperi laboratory is supported by the Navari Family Foundation and the Indiana Clinical and Translational Sciences Institute, funded in part by grant #UL1 TR001108 (A. Shekhar, PI) from the National Institutes of Health, National Center for Advancing Translational Sciences, Clinical and Translational Sciences Award. The awards were made to JRP. ATL was supported by a COS-SURF fellowship through the University of Notre Dame.
No Conflict Statement
“No potential conflicts of interest were disclosed.”
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2017 Springer International Publishing AG
About this chapter
Cite this chapter
Lyons, A.T., Prosperi, J.R. (2017). Targeted Therapies in Breast Cancer. In: Prosperi, J. (eds) Resistance to Targeted Therapies in Breast Cancer. Resistance to Targeted Anti-Cancer Therapeutics, vol 16. Springer, Cham. https://doi.org/10.1007/978-3-319-70142-4_6
Download citation
DOI: https://doi.org/10.1007/978-3-319-70142-4_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-70141-7
Online ISBN: 978-3-319-70142-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)