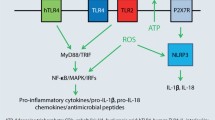
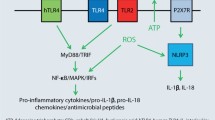
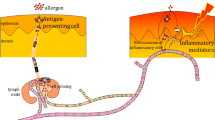
Abstract
Allergies are highly prevalent hypersensitivity responses to usually harmless substances. They are mediated by the immune system which causes pathologic responses such as type I (rhinoconjunctivitis, allergic asthma, atopy) or type IV hypersensitivity (allergic contact dermatitis). The different types of allergy are mediated by effector and memory T cells and, in the case of type I hypersensitivity, B cells. A prerequisite for the activation of these cells of the adaptive immune system is the activation of the innate immune system. The resulting inflammation is essential not only for the initiation but also for the elicitation and maintenance of allergies. Great progress has been made in the elucidation of the cellular and molecular pathomechanisms underlying allergen-induced inflammation. It is now recognized that the innate immune system in concert with tissue stress and damage responses orchestrates inflammation. This should enable the development of novel mechanism-based anti-inflammatory treatment strategies as well as of animal-free in vitro assays for the identification and potency classification of contact allergens.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Abbreviations
- ACD:
-
Allergic contact dermatitis
- ASC:
-
Apoptotic speck protein
- CHS:
-
Contact hypersensitivity
- DAMPs:
-
Damage-associated molecular patterns
- DNFB:
-
2,4-Dinitrofluorobenzene
- FITC:
-
Fluorescein isothiocyanate
- ICD:
-
Irritant contact dermatitis
- ILC:
-
Innate lymphoid cell
- Keap1:
-
Kelch-like ECH-associated protein 1
- ko:
-
Knockout
- MAMPs:
-
Microbe-associated molecular patterns
- NLRP:
-
NOD-like receptor protein
- Nrf2:
-
Nuclear factor erythroid 2-related factor 2
- PAMPs:
-
Pathogen-associated molecular patterns
- RAG:
-
Recombination activating gene
- ROS:
-
Reactive oxygen species
- TLR:
-
Toll-like receptor
- TNCB:
-
2,4,6-Trinitrochlorobenzene
- Trm:
-
Tissue-resident memory T cell
References
Ahlström MG, Thyssen JP, Wennervaldt M et al (2019) Nickel allergy and allergic contact dermatitis: a clinical review of immunology, epidemiology, exposure, and treatment. Contact Dermatitis 81:227–241. https://doi.org/10.1111/cod.13327
Akamatsu M, Mikami N, Ohkura N et al (2019) Conversion of antigen-specific effector/memory T cells into Foxp3-expressing Treg cells by inhibition of CDK8/19. Sci Immunol 4:eaaw2707. https://doi.org/10.1126/sciimmunol.aaw2707
Alinaghi F, Bennike NH, Egeberg A et al (2019) Prevalence of contact allergy in the general population: a systematic review and meta-analysis. Contact Dermatitis 80:77–85. https://doi.org/10.1111/cod.13119
Annunziato F, Romagnani C, Romagnani S (2015) The 3 major types of innate and adaptive cell-mediated effector immunity. J Allergy Clin Immunol 135:626–635. https://doi.org/10.1016/j.jaci.2014.11.001
Bains SN, Nash P, Fonacier L (2019) Irritant contact dermatitis. Clinic Rev Allerg Immunol 56:99–109. https://doi.org/10.1007/s12016-018-8713-0
Bergmann K-C, Heinrich J, Niemann H (2016) Current status of allergy prevalence in Germany: position paper of the Environmental Medicine Commission of the Robert Koch Institute. Allergo J Int 25:6–10. https://doi.org/10.1007/s40629-016-0092-6
Bonefeld CM, Nielsen MM, Rubin IMC et al (2011) Enhanced sensitization and elicitation responses caused by mixtures of common fragrance allergens. Contact Dermatitis 65:336–342. https://doi.org/10.1111/j.1600-0536.2011.01945.x
Bonefeld CM, Geisler C, Gimenéz-Arnau E et al (2017) Immunological, chemical and clinical aspects of exposure to mixtures of contact allergens. Contact Derm 77:133–142. https://doi.org/10.1111/cod.12847
Brys AK, Rodriguez-Homs LG, Suwanpradid J et al (2019) Shifting paradigms in allergic contact dermatitis: the role of innate immunity. J Investig Dermatol. https://doi.org/10.1016/j.jid.2019.03.1133
De Filippo K, Dudeck A, Hasenberg M et al (2013) Mast cell and macrophage chemokines CXCL1/CXCL2 control the early stage of neutrophil recruitment during tissue inflammation. Blood 121:4930–4937. https://doi.org/10.1182/blood-2013-02-486217
Dhingra N, Shemer A, Correa da Rosa J et al (2014) Molecular profiling of contact dermatitis skin identifies allergen-dependent differences in immune response. J Allergy Clin Immunol 134:362–372. https://doi.org/10.1016/j.jaci.2014.03.009
Diepgen T, Ofenloch RF, Bruze M et al (2016) Prevalence of contact allergy in the general population in different European regions. Br J Dermatol 174:319–329. https://doi.org/10.1111/bjd.14167
Dudeck A, Dudeck J, Scholten J et al (2011) Mast cells are key promoters of contact allergy that mediate the adjuvant effects of Haptens. Immunity 34:973–984. https://doi.org/10.1016/j.immuni.2011.03.028
Dudeck J, Ghouse SM, Lehmann CHK et al (2015) Mast-cell-derived TNF amplifies CD8(+) dendritic cell functionality and CD8(+) T cell priming. Cell Rep 13:399–411. https://doi.org/10.1016/j.celrep.2015.08.078
Dudeck J, Froebel J, Kotrba J et al (2019) Engulfment of mast cell secretory granules on skin inflammation boosts dendritic cell migration and priming efficiency. J Allergy Clin Immunol 143:1849–1864.e4. https://doi.org/10.1016/j.jaci.2018.08.052
El Ali Z, Gerbeix C, Hemon P et al (2013) Allergic skin inflammation induced by chemical sensitizers is controlled by the transcription factor Nrf2. Toxicol Sci 134:39–48. https://doi.org/10.1093/toxsci/kft084
Engeman T, Gorbachev AV, Kish DD, Fairchild RL (2004) The intensity of neutrophil infiltration controls the number of antigen-primed CD8 T cells recruited into cutaneous antigen challenge sites. J Leukoc Biol 76:941–949. https://doi.org/10.1189/jlb.0304193
Esser PR, Wolfle U, Durr C et al (2012) Contact sensitizers induce skin inflammation via ROS production and hyaluronic acid degradation. PLoS One 7:e41340. https://doi.org/10.1371/journal.pone.0041340
Esser PR, Zech A, Idzko M, Martin SF (2018) Lack of biglycan reduces contact hypersensitivity in mice. Contact Derm 79:326–328. https://doi.org/10.1111/cod.13082
Eyerich S, Metz M, Bossios A, Eyerich K (2019) New biological treatments for asthma and skin allergies. Allergy. https://doi.org/10.1111/all.14027
Ezendam J, Braakhuis HM, Vandebriel RJ (2016) State of the art in non-animal approaches for skin sensitization testing: from individual test methods towards testing strategies. Arch Toxicol 90:2861–2883. https://doi.org/10.1007/s00204-016-1842-4
Fullerton JN, Gilroy DW (2016) Resolution of inflammation: a new therapeutic frontier. Nat Rev Drug Discov 15:551–567. https://doi.org/10.1038/nrd.2016.39
Galbiati V, Papale A, Galli CL et al (2014) Role of ROS and HMGB1 in contact allergen-induced IL-18 production in human keratinocytes. J Invest Dermatol 134:2719–2727. https://doi.org/10.1038/jid.2014.203
Gaspari AA, Katz SI, Martin SF (2016) Contact hypersensitivity. Curr Protoc Immunol 113:4.2.1–4.2.7. https://doi.org/10.1002/0471142735.im0402s113
Gimenez-Rivera V-A, Siebenhaar F, Zimmermann C et al (2016) Mast cells limit the exacerbation of chronic allergic contact dermatitis in response to repeated allergen exposure. J Immunol 197:4240–4246. https://doi.org/10.4049/jimmunol.1600236
Gonzalez-Teuber V, Albert-Gasco H, Auyeung VC et al (2019) Small molecules to improve ER proteostasis in disease. Trends Pharmacol Sci 40:684–695. https://doi.org/10.1016/j.tips.2019.07.003
Goubier A, Vocanson M, Macari C et al (2013) Invariant NKT cells suppress CD8+ T-cell-mediated allergic contact dermatitis independently of regulatory CD4+ T cells. J Invest Dermatol 133:980–987
Guo Z, Wang G, Lv Y et al (2019) Inhibition of Cdk8/Cdk19 activity promotes Treg cell differentiation and suppresses autoimmune diseases. Front Immunol 10. https://doi.org/10.3389/fimmu.2019.01988
Helou DG, Martin SF, Pallardy M et al (2019a) Nrf2 involvement in chemical-induced skin innate immunity. Front Immunol 10. https://doi.org/10.3389/fimmu.2019.01004
Helou DG, Noël B, Gaudin F et al (2019b) Cutting edge: Nrf2 regulates neutrophil recruitment and accumulation in skin during contact hypersensitivity. J Immunol 202:2189–2194. https://doi.org/10.4049/jimmunol.1801065
Hetz C, Axten JM, Patterson JB (2019) Pharmacological targeting of the unfolded protein response for disease intervention. Nat Chem Biol 15:764–775. https://doi.org/10.1038/s41589-019-0326-2
Ho AW, Kupper TS (2019) T cells and the skin: from protective immunity to inflammatory skin disorders. Nat Rev Immunol 19:490–502. https://doi.org/10.1038/s41577-019-0162-3
Honda T, Egawa G, Grabbe S, Kabashima K (2013) Update of immune events in the murine contact hypersensitivity model: toward the understanding of allergic contact dermatitis. J Invest Dermatol 133:303–315. https://doi.org/10.1038/jid.2012.284
Jakasa I, Thyssen JP, Kezic S (2018) The role of skin barrier in occupational contact dermatitis. Exp Dermatol 27:909–914. https://doi.org/10.1111/exd.13704
Janssens S, Pulendran B, Lambrecht BN (2014) Emerging functions of the unfolded protein response in immunity. Nat Immunol 15:910–919. https://doi.org/10.1038/ni.2991
Johansen JD, Werfel T (2019) Highlights in allergic contact dermatitis 2018/2019. Curr Opin Allergy Clin Immunol 19:334–340. https://doi.org/10.1097/ACI.0000000000000552
Joosten LAB, Abdollahi-Roodsaz S, Dinarello CA et al (2016) Toll-like receptors and chronic inflammation in rheumatic diseases: new developments. Nat Rev Rheumatol 12:344–357. https://doi.org/10.1038/nrrheum.2016.61
Kaplan DH, Igyártó BZ, Gaspari AA (2012) Early immune events in the induction of allergic contact dermatitis. Nat Rev Immunol 12:114–124. https://doi.org/10.1038/nri3150
Kaufman BP, Alexis AF (2018) Biologics and small molecule agents in allergic and immunologic skin diseases. Curr Allergy Asthma Rep 18:55. https://doi.org/10.1007/s11882-018-0804-8
Kim BS (2015) Innate lymphoid cells in the skin. J Invest Dermatol 135:673–678. https://doi.org/10.1038/jid.2014.401
Kim HS, Jang J-H, Lee MB et al (2016) A novel IL-10-producing innate lymphoid cells (ILC10) in a contact hypersensitivity mouse model. BMB Rep 49:293–296. https://doi.org/10.5483/bmbrep.2016.49.5.023
Kish DD, Min S, Dvorina N et al (2019) Neutrophil cathepsin G regulates dendritic cell production of IL-12 during development of CD4 T cell responses to antigens in the skin. J Immunol 202:1045–1056. https://doi.org/10.4049/jimmunol.1800841
Kortekaas Krohn I, Shikhagaie MM, Golebski K et al (2018) Emerging roles of innate lymphoid cells in inflammatory diseases: clinical implications. Allergy 73:837–850. https://doi.org/10.1111/all.13340
Leonard A, Guttman-Yassky E (2019) The unique molecular signatures of contact dermatitis and implications for treatment. Clin Rev Allergy Immunol 56:1–8. https://doi.org/10.1007/s12016-018-8685-0
Luís A, Martins JD, Silva A et al (2014) Oxidative stress-dependent activation of the eIF2α–ATF4 unfolded protein response branch by skin sensitizer 1-fluoro-2,4-dinitrobenzene modulates dendritic-like cell maturation and inflammatory status in a biphasic manner [corrected]. Free Radic Biol Med 77:217–229. https://doi.org/10.1016/j.freeradbiomed.2014.09.008
Maeda K, Caldez MJ, Akira S (2019) Innate immunity in allergy. Allergy 74:1660–1674. https://doi.org/10.1111/all.13788
Majewska-Szczepanik M, Paust S, von Andrian UH et al (2013) Natural killer cell-mediated contact sensitivity develops rapidly and depends on interferon-α, interferon-γ and interleukin-12. Immunology 140:98–110. https://doi.org/10.1111/imm.12120
Martin SF (2013) Induction of contact hypersensitivity in the mouse model. Methods Mol Biol 961:325–335. https://doi.org/10.1007/978-1-62703-227-8_21
Martin SF (2014) Adaptation in the innate immune system and heterologous innate immunity. Cell Mol Life Sci 71:4115–4130. https://doi.org/10.1007/s00018-014-1676-2
Martin SF (2015) New concepts in cutaneous allergy. Contact Derm 72:2–10. https://doi.org/10.1111/cod.12311
Martin SF, Esser PR, Schmucker S et al (2010) T-cell recognition of chemicals, protein allergens and drugs: towards the development of in vitro assays. Cell Mol Life Sci 67:4171–4184. https://doi.org/10.1007/s00018-010-0495-3
Morita H, Moro K, Koyasu S (2016) Innate lymphoid cells in allergic and nonallergic inflammation. J Allergy Clin Immunol 138:1253–1264. https://doi.org/10.1016/j.jaci.2016.09.011
Mussotter F, Potratz S, Budczies J et al (2018) A multi-omics analysis reveals metabolic reprogramming in THP-1 cells upon treatment with the contact allergen DNCB. Toxicol Appl Pharmacol 340:21–29. https://doi.org/10.1016/j.taap.2017.12.016
Nassau S, Fonacier L (2020) Allergic contact dermatitis. Med Clin N Am 104:61–76. https://doi.org/10.1016/j.mcna.2019.08.012
O’Leary JG, Goodarzi M, Drayton DL, von Andrian UH (2006) T cell- and B cell-independent adaptive immunity mediated by natural killer cells. Nat Immunol 7:507–516. https://doi.org/10.1038/ni1332
Paust S, Gill HS, Wang B-Z et al (2010) Critical role for the chemokine receptor CXCR6 in NK cell-mediated antigen-specific memory of haptens and viruses. Nat Immunol 11:1127–1135. https://doi.org/10.1038/ni.1953
Pedersen LK, Johansen JD, Held E, Agner T (2004) Augmentation of skin response by exposure to a combination of allergens and irritants – a review. Contact Derm 50:265–273. https://doi.org/10.1111/j.0105-1873.2004.00342.x
Peng H, Jiang X, Chen Y et al (2013) Liver-resident NK cells confer adaptive immunity in skin-contact inflammation. J Clin Invest 123:1444–1456. https://doi.org/10.1172/JCI66381
Pivniouk V, Gimenes Junior JA, Honeker L, Vercelli D (2019) The role of innate immunity in asthma development and protection: lessons from the environment. Clin Exp Allergy. https://doi.org/10.1111/cea.13508
Rafei-Shamsabadi DA, Klose CSN, Halim TYF et al (2019) Context dependent role of type 2 innate lymphoid cells in allergic skin inflammation. Front Immunol 10:2591. https://doi.org/10.3389/fimmu.2019.02591
Rankin LC, Artis D (2018) Beyond host defense: emerging functions of the immune system in regulating complex tissue physiology. Cell 173:554–567. https://doi.org/10.1016/j.cell.2018.03.013
Rouzaire P, Luci C, Blasco E et al (2012) Natural killer cells and T cells induce different types of skin reactions during recall responses to haptens. Eur J Immunol 42:80–88. https://doi.org/10.1002/eji.201141820
Simon D (2018) Recent advances in clinical allergy and immunology. Int Arch Allergy Immunol 177:324–333. https://doi.org/10.1159/000494931
Suto H, Nakae S, Kakurai M et al (2006) Mast cell-associated TNF promotes dendritic cell migration. J Immunol 176:4102–4112. https://doi.org/10.4049/jimmunol.176.7.4102
Sutterwala FS, Ogura Y, Szczepanik M et al (2006) Critical role for NALP3/CIAS1/cryopyrin in innate and adaptive immunity through its regulation of caspase-1. Immunity 24:317–327. https://doi.org/10.1016/j.immuni.2006.02.004
Swanson KV, Deng M, Ting JP-Y (2019) The NLRP3 inflammasome: molecular activation and regulation to therapeutics. Nat Rev Immunol 19:477–489. https://doi.org/10.1038/s41577-019-0165-0
Tan H-TT, Sugita K, Akdis CA (2016) Novel biologicals for the treatment of allergic diseases and asthma. Curr Allergy Asthma Rep 16:70. https://doi.org/10.1007/s11882-016-0650-5
Toksoy A, Sennefelder H, Adam C et al (2017) Potent NLRP3 inflammasome activation by the HIV reverse transcriptase inhibitor Abacavir. J Biol Chem 292:2805–2814. https://doi.org/10.1074/jbc.M116.749473
van den Boorn JG, Jakobs C, Hagen C et al (2016) Inflammasome-dependent induction of adaptive NK cell memory. Immunity 44:1406–1421. https://doi.org/10.1016/j.immuni.2016.05.008
Vivier E, Artis D, Colonna M et al (2018) Innate lymphoid cells: 10 years on. Cell 174:1054–1066. https://doi.org/10.1016/j.cell.2018.07.017
Vocanson M, Hennino A, Rozières A et al (2009) Effector and regulatory mechanisms in allergic contact dermatitis. Allergy 64:1699–1714. https://doi.org/10.1111/j.1398-9995.2009.02082.x
Weber FC, Esser PR, Muller T et al (2010) Lack of the purinergic receptor P2X(7) results in resistance to contact hypersensitivity. J Exp Med 207:2609–2619. https://doi.org/10.1084/jem.20092489
Weber FC, Németh T, Csepregi JZ et al (2015) Neutrophils are required for both the sensitization and elicitation phase of contact hypersensitivity. J Exp Med 212:15–22. https://doi.org/10.1084/jem.20130062
Woolbright BL, Jaeschke H (2017) Role of the inflammasome in acetaminophen-induced liver injury and acute liver failure. J Hepatol 66:836–848. https://doi.org/10.1016/j.jhep.2016.11.017
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Martin, S.F., Esser, P.R. (2021). Innate Immune Mechanisms in Contact Dermatitis. In: Traidl-Hoffmann, C., Zuberbier, T., Werfel, T. (eds) Allergic Diseases – From Basic Mechanisms to Comprehensive Management and Prevention . Handbook of Experimental Pharmacology, vol 268. Springer, Cham. https://doi.org/10.1007/164_2021_482
Download citation
DOI: https://doi.org/10.1007/164_2021_482
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-84047-1
Online ISBN: 978-3-030-84048-8
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)