Abstract
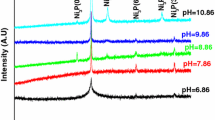
The high-temperature oxidation of Ni films electroplated on a steel substrate was studied at 700 and 800°C in air. During oxidation, outward diffusion of Ni and Fe ions and inward diffusion of oxygen ions occurred. NiO formed initially, and as the oxidation progressed, Fe2O3 and NiFe2O4 also formed. The oxidation resistance of Ni films was inferior to that of bulk Ni, which was attributed to the involvement of Fe during oxidation.
Similar content being viewed by others
References
R. Peraldi, D. Monceau, and B. Pieraggi,Oxid. Met. 58, 249 (2002).
R. Peraldi, D. Monceau, and B. Pieraggi,Oxid. Met. 58, 275 (2002).
M. Donten, H. Cesiulis, and Z. Stojek,Electrochim. Acta 45, 3389 (2000).
C. Louro, and A. Cavaleiro,J. Electrochem. Soc. 144, 259 (1997).
M. Pushpavanam, S. R. Natarajan, and K. Balakrishnan,Plat. Surf. Finish. 84, 88 (1997).
G. Zhongcheng, Z. Chengyi, Z. Dacheng, and Y. Xianwan,Trans. Nonferr. Met. Soc. China 10, 50 (2000).
ASM Handbook Vol. 5, p. 201, ASM Materials Park, Ohio, USA (1994).
N. Birks and G. H. Meier,Introduction to High Temperature Oxidation of Metals, p. 88, Edward Arnold, London (1983).
D. A. Jones,Principle and Prevention of Corrosion 2nd ed., p. 416, Prentice-Hall, USA (1996).
M. Venkatraman and J. P. Neumann,Binary Alloy Phase Diagrams 2nd ed., vol. 1, p. 842, ASM, Metals Park, Ohio, USA (1990).