Abstract
Background
In the healthy brain, small oscillations in intracranial pressure (ICP) occur synchronously with those in cerebral blood volume (CBV), cerebrovascular resistance, and consequently cerebral blood flow velocity (CBFV). Previous work has shown that the usual synchrony between ICP and CBFV is lost during intracranial hypertension. Moreover, a continuously computed measure of the ICP/CBFV association (Fix index) was a more sensitive predictor of outcome after traumatic brain injury (TBI) than a measure of autoregulation (Mx index). In the current study we computed Fix during ICP plateau waves, to observe its behavior during a defined period of cerebrovascular vasodilatation.
Methods
Twenty-nine recordings of arterial blood pressure (ABP), ICP, and CBFV taken during ICP plateau waves were obtained from the Addenbrooke’s hospital TBI database. Raw data was filtered prior to computing Mx and Fix according to previously published methods. Analyzed data was segmented into three phases (pre, peak, and post), and a median value of each parameter was stored for analysis.
Results
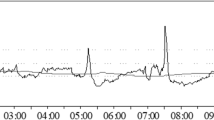
ICP increased from a median of 22–44 mmHg before falling to 19 mmHg. Both Mx and Fix responded to the increase in ICP, with Mx trending toward +1, while Fix trended toward −1. Mx and Fix correlated significantly (Spearman’s R = −0.89, p < 0.000001), however, Fix spanned a greater range than Mx. A plot of Mx and Fix against CPP showed a plateau (Mx) or trough (Fix) consistent with a zone of “optimal CPP”.
Conclusions
The Fix index can identify complete loss of cerebral autoregulation as the point at which the normally positive CBF/CBV correlation is reversed. Both CBF and CBV can be monitored noninvasively using near-infrared spectroscopy (NIRS), suggesting that a noninvasive method of monitoring autoregulation using only NIRS may be possible.





Similar content being viewed by others
References
Diehl RR, Diehl B, Sitzer M, Hennerici M. Spontaneous oscillations in cerebral blood flow velocity in normal humans and in patients with carotid artery disease. Neurosci Lett. 1991;127:5–8.
Lang EW, Diehl RR, Timmermann L, et al. Spontaneous oscillations of arterial blood pressure, cerebral and peripheral blood flow in healthy and comatose subjects. Neurol Res. 1999;21:665–9.
Lewis PM, Smielewski P, Pickard JD, Czosnyka M. Slow oscillations in middle cerebral artery cerebral blood flow velocity and aging. Neurol Res. 2007;29:260–3.
Lundar T, Lindegaard KF, Nornes H. Continuous recording of middle cerebral artery blood velocity in clinical neurosurgery. Acta Neurochir (Wien). 1990;102:85–90.
Lundberg N. Continuous recording and control of ventricular fluid pressure in neurosurgical practice. Acta Psychiatr Scand Suppl. 1960;36:1–193.
Castellani G, Zweifel C, Kim DJ, et al. Plateau waves in head injured patients requiring neurocritical care. Neurocrit Care. 2009;11:143–50.
Aalkjaer C, Boedtkjer D, Matchkov V. Vasomotion—what is currently thought? Acta Physiol (Oxf). 2011;202:253–69.
Auer LM, Sayama I. Intracranial pressure oscillations (B-waves) caused by oscillations in cerebrovascular volume. Acta Neurochir (Wien). 1983;68:93–100.
Droste DW, Krauss JK, Berger W, Schuler E, Brown MM. Rhythmic oscillations with a wavelength of 0.5–2 min in transcranial Doppler recordings. Acta Neurol Scand. 1994;90:99–104.
Newell DW, Aaslid R, Stooss R, Reulen HJ. The relationship of blood flow velocity fluctuations to intracranial pressure B waves. J Neurosurg. 1992;76:415–21.
Zhang R, Zuckerman JH, Giller CA, Levine BD. Transfer function analysis of dynamic cerebral autoregulation in humans. Am J Physiol. 1998;274:H233–41.
Reinhard M, Roth M, Guschlbauer B, et al. Dynamic cerebral autoregulation in acute ischemic stroke assessed from spontaneous blood pressure fluctuations. Stroke. 2005;36:1684–9.
Rosner MJ. The vasodilatory cascade and intracranial pressure. In: Miller JD, Teasdale GM, Rowan JO, Galbraith SL, Mendelow AD, editors. Intracranial pressure IV. Berlin: Springer; 1986.
Czosnyka M, Smielewski P, Piechnik S, et al. Hemodynamic characterization of intracranial pressure plateau waves in head-injury patients. J Neurosurg. 1999;91:11–9.
Goh D, Minns RA, Pye SD, Steers AJ. Cerebral blood-flow velocity and intermittent intracranial pressure elevation during sleep in hydrocephalic children. Dev Med Child Neurol. 1992;34:676–89.
Czosnyka M, Smielewski P, Piechnik S, Steiner LA, Pickard JD. Cerebral autoregulation following head injury. J Neurosurg. 2001;95:756–63.
Lewis PM, Smielewski P, Rosenfeld JV, Pickard JD, Czosnyka M. Monitoring of the association between cerebral blood flow velocity and intracranial pressure. Acta Neurochir Suppl. 2012;114:147–51.
Budohoski KP, Czosnyka M, de Riva N, et al. The relationship between cerebral blood flow autoregulation and cerebrovascular pressure reactivity after traumatic brain injury. Neurosurgery. 2012;71:652–60 discussion 60–1.
Kim DJ, Kasprowicz M, Carrera E, et al. The monitoring of relative changes in compartmental compliances of brain. Physiol Meas. 2009;30:647–59.
Lassen NA. Cerebral blood flow and oxygen consumption in man. Physiol Rev. 1959;39:183–238.
Weerakkody RA, Czosnyka M, Zweifel C, et al. Slow vasogenic fluctuations of intracranial pressure and cerebral near infrared spectroscopy–an observational study. Acta Neurochir (Wien). 2010;152:1763–9.
Risberg J, Ancri D, Ingvar DH. Correlation between cerebral blood volume and cerebral blood flow in the cat. Exp Brain Res. 1969;8:321–6.
Barzo P, Doczi T, Csete K, Buza Z, Bodosi M. Measurements of regional cerebral blood flow and blood flow velocity in experimental intracranial hypertension: infusion via the cisterna magna in rabbits. Neurosurgery. 1991;28:821–5.
Grubb RL Jr, Raichle ME, Phelps ME, Ratcheson RA. Effects of increased intracranial pressure on cerebral blood volume, blood flow, and oxygen utilization in monkeys. J Neurosurg. 1975;43:385–98.
Miller JD, Stanek A, Langfitt TW. Concepts of cerebral perfusion pressure and vascular compression during intracranial hypertension. Prog Brain Res. 1972;35:411–32.
Wolff HG, Forbes HS. The cerebral circulation v: observations of the pial circulation during changes in intracranial pressure. Arch Neurol Psych. 1928;20:1035–47.
Vignes JR, Dagain A, Guerin J, Liguoro D. A hypothesis of cerebral venous system regulation based on a study of the junction between the cortical bridging veins and the superior sagittal sinus. Laboratory investigation. J Neurosurg. 2007;107:1205–10.
Cushing HMD. Some experimental and clinical observations concerning states of increased intracranial tension. Am J Med Sci. 1902;124:375–400.
Nakagawa Y, Tsuru M, Yada K. Site and mechanism for compression of the venous system during experimental intracranial hypertension. J Neurosurg. 1974;41:427–34.
Luce JM, Huseby JS, Kirk W, Butler J. A starling resistor regulates cerebral venous outflow in dogs. J Appl Physiol Respir Environ Exerc Physiol. 1982;53:1496–503.
Johnston IH, Rowan JO. Raised intracranial pressure and cerebral blood flow. 3. Venous outflow tract pressures and vascular resistances in experimental intracranial hypertension. J Neurol Neurosurg Psychiatry. 1974;37:392–402.
Marmarou A, Maset AL, Ward JD, et al. Contribution of CSF and vascular factors to elevation of ICP in severely head-injured patients. J Neurosurg. 1987;66:883–90.
Risberg J, Lundberg N, Ingvar DH. Regional cerebral blood volume during acute transient rises of the intracranial pressure (plateau waves). J Neurosurg. 1969;31:303–10.
Rosner MJ, Becker DP. Origin and evolution of plateau waves. Experimental observations and a theoretical model. J Neurosurg. 1984;60:312–24.
Marmarou A, Signoretti S, Fatouros PP, Portella G, Aygok GA, Bullock MR. Predominance of cellular edema in traumatic brain swelling in patients with severe head injuries. J Neurosurg. 2006;104:720–30.
Brady KM, Lee JK, Kibler KK, et al. The lower limit of cerebral blood flow autoregulation is increased with elevated intracranial pressure. Anesth Analg. 2009;108:1278–83.
Anile C, De Bonis P, Di Chirico A, Ficola A, Mangiola A, Petrella G. Cerebral blood flow autoregulation during intracranial hypertension: a simple, purely hydraulic mechanism? Childs Nerv Syst. 2009;25:325–35 discussion 37–40.
Avezaat CJJ. Cerebral blood flow autoregulation during intracranial hypertension: a simple, purely hydraulic mechanism? Childs Nerv Syst. 2009;25:339–40.
Wagner EM, Traystman RJ. Cerebrovascular transmural pressure and autoregulation. Ann Biomed Eng. 1985;13:311–20.
Strandgaard S, Paulson OB. Cerebral autoregulation. Stroke. 1984;15:413–6.
Brierley JB, Brown AW, Excell BJ, Meldrum BS. Brain damage in the rhesus monkey resulting from profound arterial hypotension. I. Its nature, distribution and general physiological correlates. Brain Res. 1969;13:68–100.
Wolf M, Ferrari M, Quaresima V. Progress of near-infrared spectroscopy and topography for brain and muscle clinical applications. J Biomed Opt. 2007;12:062104.
Lewis PM, Rosenfeld JV, Diehl RR, Mehdorn HM, Lang EW. Phase shift and correlation coefficient measurement of cerebral autoregulation during deep breathing in traumatic brain injury (TBI). Acta Neurochir (Wien). 2008;150:139–46 discussion 46–7.
Diehl RR, Linden D, Lucke D, Berlit P. Phase relationship between cerebral blood flow velocity and blood pressure. A clinical test of autoregulation. Stroke. 1995;26:1801–4.
Steiner LA, Czosnyka M, Piechnik SK, et al. Continuous monitoring of cerebrovascular pressure reactivity allows determination of optimal cerebral perfusion pressure in patients with traumatic brain injury. Crit Care Med. 2002;30:733–8.
Jaeger M, Schuhmann MU, Soehle M, Meixensberger J. Continuous assessment of cerebrovascular autoregulation after traumatic brain injury using brain tissue oxygen pressure reactivity. Crit Care Med. 2006;34:1783–8.
Bhuiyan MR, Deb S, Mitchell RA, Teddy PJ, Drummond KJ. The effect of formal training on the clinical utility of transcranial Doppler ultrasound monitoring in patients with aneurysmal subarachnoid haemorrhage. J Clin Neurosci. 2012;19:1255–60.
Czosnyka M, Balestreri M, Steiner L, et al. Age, intracranial pressure, autoregulation, and outcome after brain trauma. J Neurosurg. 2005;102:450–4.
Conflict of interest
Marek Czosnyka and Peter Smielewski have a financial interest in a part of the licensing fee for ICM+. Philip M. Lewis, Jeffrey V. Rosenfeld, and John D. Pickard declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lewis, P.M., Smielewski, P., Rosenfeld, J.V. et al. A Continuous Correlation Between Intracranial Pressure and Cerebral Blood Flow Velocity Reflects Cerebral Autoregulation Impairment During Intracranial Pressure Plateau Waves. Neurocrit Care 21, 514–525 (2014). https://doi.org/10.1007/s12028-014-9994-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-014-9994-7