Abstract
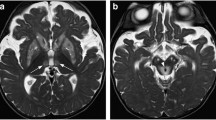
Cerebral amyloid angiopathy-related inflammation (CAARI) is a recently recognized syndrome of reversible encephalopathy seen in a subset of patients with cerebral amyloid angiopathy (CAA). CAA is a disorder of the elderly in which amyloid peptides are deposited in the walls of cerebral arteries, leading to microhemorrhages, macrohemorrhages, and eventually dementia. In a few cases, the amyloid deposition is accompanied by inflammation or edema. The clinical syndrome of CAARI is distinguished by subacute neurobehavioral symptoms, headaches, seizures, and stroke-like signs, contrasting the acute intracranial hemorrhage typically seen in CAA. Magnetic resonance imaging findings may be symmetric or asymmetric and involve patchy or confluent T2 hyperintense lesions in the cortex and subcortical white matter. Recent diagnostic criteria have been proposed which help distinguish CAARI from alternative diagnoses. Improvement has been reported in most cases with immunosuppression, although a few cases have had recurrent symptoms. Here, we review the clinical and radiologic features of CAARI and compare these with CAA.


Similar content being viewed by others
Abbreviations
- Amyloid beta peptides:
-
generally 40 or 42 amino acid peptides, which can bind together into beta-pleated sheets to form deposits of amyloid
- Cerebral amyloid angiopathy:
-
the condition of amyloid deposition in cerebral arteries
- CAARI:
-
cerebral amyloid angiopathy-related inflammation
- Gradient echo MR imaging:
-
a sequence in which blood products “bloom” and become easily visible on MRI
- Microhemorrhages or microbleeds:
-
punctuate hemorrhages ≤5 mm, usually seen in the posterior areas of the brain, near the cortical/subcortical junction in cerebral amyloid angiopathy
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Vinters HV. Cerebral amyloid angiopathy. A critical review. Stroke 1987;18(2):311–24.
Attems J, Jellinger K, Thal DR, Van Nostrand W. Review: sporadic cerebral amyloid angiopathy. Neuropathol Appl Neurobiol. 2011;37(1):75–93. This is an excellent review of amyloid angiopathy.
Glenner GG, Wong CW. Alzheimer’s disease: initial report of the purification and characterization of a novel cerebrovascular amyloid protein. Biochem Biophys Res Commun. 1984;120(3):885–90.
Jellinger KA. Alzheimer disease and cerebrovascular pathology: an update. J Neural Transm. 2002;109(5-6):813–36.
Greenberg SM. Cerebral amyloid angiopathy: prospects for clinical diagnosis and treatment. Neurology. 1998;51(3):690–4.
McCarron MO, Nicoll JA, Stewart J, et al. The apolipoprotein E epsilon2 allele and the pathological features in cerebral amyloid angiopathy-related hemorrhage. J Neuropathol Exp Neurol. 1999;58(7):711–8.
Greenberg SM, Vonsattel JP, Segal AZ, et al. Association of apolipoprotein E epsilon2 and vasculopathy in cerebral amyloid angiopathy. Neurology. 1998;50(4):961–5.
McCarron MO, Nicoll JA, Ironside JW, Love S, Alberts MJ, Bone I. Cerebral amyloid angiopathy-related hemorrhage. Interaction of APOE epsilon2 with putative clinical risk factors. Stroke J Cereb Circ. 1999;30(8):1643–6.
Nicoll JA, Burnett C, Love S, et al. High frequency of apolipoprotein E epsilon 2 allele in hemorrhage due to cerebral amyloid angiopathy. Ann Neurol. 1997;41(6):716–21.
O’Donnell HC, Rosand J, Knudsen KA, et al. Apolipoprotein E genotype and the risk of recurrent lobar intracerebral hemorrhage. N Engl J Med. 2000;342(4):240–5.
Hernandez-Guillamon M, Delgado P, Penalba A, et al. Plasma beta-amyloid levels in cerebral amyloid angiopathy-associated hemorrhagic stroke. Neurodegener Dis. 2012;10(1-4):320–3.
Knudsen KA, Rosand J, Karluk D, Greenberg SM. Clinical diagnosis of cerebral amyloid angiopathy: validation of the Boston criteria. Neurology. 2001;56(4):537–9.
Chao CP, Kotsenas AL, Broderick DF. Cerebral amyloid angiopathy: CT and MR imaging findings. Radiogr. 2006;26(5):1517–31.
Greenberg SM, Vernooij MW, Cordonnier C, et al. Cerebral microbleeds: a guide to detection and interpretation. Lancet Neurol. 2009;8(2):165–74. This paper teaches the reader how to evaluate a gradient echo MRI for microhemorrhages.
Greenberg SM, Eng JA, Ning M, Smith EE, Rosand J. Hemorrhage burden predicts recurrent intracerebral hemorrhage after lobar hemorrhage. Stroke J Cereb Circ. 2004;35(6):1415–20.
Rosand J, Hylek EM, O’Donnell HC, Greenberg SM. Warfarin-associated hemorrhage and cerebral amyloid angiopathy: a genetic and pathologic study. Neurology. 2000;55(7):947–51.
Chung KK, Anderson NE, Hutchinson D, Synek B, Barber PA. Cerebral amyloid angiopathy related inflammation: three case reports and a review. J Neurol Neurosurg Psychiatry. 2011;82(1):20–6. This article provides clinical criteria for the diagnosis of CAARI.
Eng JA, Frosch MP, Choi K, Rebeck GW, Greenberg SM. Clinical manifestations of cerebral amyloid angiopathy-related inflammation. Ann Neurol. 2004;55(2):250–6. This article was the first clear description of the clinical features of CAARI.
Kinnecom C, Lev MH, Wendell L, et al. Course of cerebral amyloid angiopathy-related inflammation. Neurology. 2007;68(17):1411–6.
Martucci M, Sarria S, Toledo M, et al. Cerebral amyloid angiopathy-related inflammation: imaging findings and clinical outcome. Neuroradiology. 2014;56(4):283–9.
Scolding NJ, Joseph F, Kirby PA, et al. Abeta-related angiitis: primary angiitis of the central nervous system associated with cerebral amyloid angiopathy. Brain J Neurol. 2005;128(Pt 3):500–15.
Salvarani C, Hunder GG, Morris JM, Brown Jr RD, Christianson T, Giannini C. Abeta-related angiitis: comparison with CAA without inflammation and primary CNS vasculitis. Neurology. 2013;81(18):1596–603.
Rigney L, Sebire D, Cordato D. Acute dysphasia and reversible cognitive decline in a patient with probable cerebral amyloid angiopathy-related inflammation. Case Rep Neurol Med. 2015;2015:189581.
Cenina AR, De Leon J, Tay KY, Wong CF, Kandiah N. Cerebral amyloid angiopathy-related inflammation presenting with rapidly progressive dementia, responsive to IV Ig. Alzheimer Dis Assoc Disord. 2015.
Yamada M, Itoh Y, Shintaku M, et al. Immune reactions associated with cerebral amyloid angiopathy. Stroke J Cereb Circ. 1996;27(7):1155–62.
Orgogozo JM, Gilman S, Dartigues JF, et al. Subacute meningoencephalitis in a subset of patients with AD after Abeta42 immunization. Neurology. 2003;61(1):46–54.
Nicoll JA, Wilkinson D, Holmes C, Steart P, Markham H, Weller RO. Neuropathology of human Alzheimer disease after immunization with amyloid-beta peptide: a case report. Nat Med. 2003;9(4):448–52.
Ferrer I, Boada Rovira M, Sanchez Guerra ML, Rey MJ, Costa-Jussa F. Neuropathology and pathogenesis of encephalitis following amyloid-beta immunization in Alzheimer’s disease. Brain Pathol. 2004;14(1):11–20.
Sperling R, Salloway S, Brooks DJ, et al. Amyloid-related imaging abnormalities in patients with Alzheimer’s disease treated with bapineuzumab: a retrospective analysis. Lancet Neurol. 2012;11(3):241–9.
DiFrancesco JC, Brioschi M, Brighina L, et al. Anti-Abeta autoantibodies in the CSF of a patient with CAA-related inflammation: a case report. Neurology. 2011;76(9):842–4.
Piazza F, Greenberg SM, Savoiardo M, et al. Anti-amyloid beta autoantibodies in cerebral amyloid angiopathy-related inflammation: implications for amyloid-modifying therapies. Ann Neurol. 2013;73(4):449–58. This paper has a good discussion of the immunological aspects of CAARI.
Boncoraglio GB, Piazza F, Savoiardo M, et al. Prodromal Alzheimer’s disease presenting as cerebral amyloid angiopathy-related inflammation with spontaneous amyloid-related imaging abnormalities and high cerebrospinal fluid anti-abeta autoantibodies. J Alzheimers Dis : JAD. 2015;45(2):363–7.
Brotman DJ, Eberhart CG, Burger PC, McArthur JC, Hellmann DB. Primary angiitis of the central nervous system and Alzheimer’s disease: clinically and pathologically evident in a single patient. J Rheumatol. 2000;27(12):2935–7.
Greenberg SM, Parisi JE, Keegan BM. A 63-year-old man with headaches and behavioral deterioration. Neurology. 2007;68(10):782–7.
Compliance with Ethics Guidelines
Conflict of Interest
The authors declare that they have no competing interests.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Behavior
Rights and permissions
About this article
Cite this article
Kirshner, H.S., Bradshaw, M. The Inflammatory Form of Cerebral Amyloid Angiopathy or “Cerebral Amyloid Angiopathy-Related Inflammation” (CAARI). Curr Neurol Neurosci Rep 15, 54 (2015). https://doi.org/10.1007/s11910-015-0572-y
Published:
DOI: https://doi.org/10.1007/s11910-015-0572-y