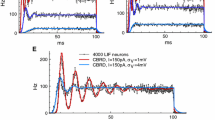
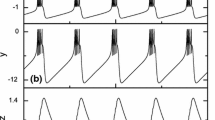
Abstract
The conductance-based refractory density (CBRD) approach is a parsimonious mathematical–computational framework for modelling interacting populations of regular spiking neurons, which, however, has not been yet extended for a population of bursting neurons. The canonical CBRD method allows to describe the firing activity of a statistical ensemble of uncoupled Hodgkin–Huxley-like neurons (differentiated by noise) and has demonstrated its validity against experimental data. The present manuscript generalises the CBRD for a population of bursting neurons; however, in this pilot computational study, we consider the simplest setting in which each individual neuron is governed by a piecewise linear bursting dynamics. The resulting population model makes use of slow–fast analysis, which leads to a novel methodology that combines CBRD with the theory of multiple timescale dynamics. The main prospect is that it opens novel avenues for mathematical explorations, as well as, the derivation of more sophisticated population activity from Hodgkin–Huxley-like bursting neurons, which will allow to capture the activity of synchronised bursting activity in hyper-excitable brain states (e.g. onset of epilepsy).





Similar content being viewed by others
References
Amari SI (1977) Dynamics of pattern formation in lateral-inhibition type neural fields. Biol Cybern 27(2):77–87
Apfaltrer F, Ly C, Tranchina D (2006) Population density methods for stochastic neurons with realistic synaptic kinetics: firing rate dynamics and fast computational methods. Network 17(4):373–418
Avitabile D, Desroches M, Knobloch E (2016) Spatiotemporal canards in neural field equations. Phys Rev E 95(4):042205
beim Graben P, Rodrigues S (2013) A biophysical observation model for field potentials of networks of leaky integrate-and-fire neurons. Front Comput Neurosci 6:100
Berglund N, Gentz B (2006) Noise-induced phenomena in slow-fast dynamical systems: a sample-paths approach. Springer, Berlin
Breakspear M, Roberts JA, Terry JR, Rodrigues S, Mahant N, Robinson PA (2015) A unifying explanation of primary generalized seizures through nonlinear brain modeling and bifurcation analysis. Cerebral Cortex 16(9):1296–1313
Brunel N, Hakim H (1999) Fast global oscillations in networks of integrate-and-fire neurons with low firing rates. Neural Comput 11(7):1621–1671
Brunel N, Chance FS, Fourcaud N, Abbott LF (2001) Effects of synaptic noise and filtering on the frequency response of spiking neurons. Phys Rev Lett 86(10):2186–2189
Cañizo JA, Yoldaş H (2018) Asymptotic behaviour of neuron population models structured by elapsed-time. arXiv:1803.07062
Casti ARR, Omurtag A, Sornborger A, Kaplan E, Knight B, Victor J, Sirovich L (2002) A population study of integrate-and-fire-or-burst neurons. Neural Comput 14:957–986
Chevallier J, Cáceres MJ, Doumic M, Reynaud-Bouret P (2015) Microscopic approach of a time elapsed neural model. Math Models Methods Appl Sci 25(14):2669–2719
Chizhov AV (2014) Conductance-based refractory density model of primary visual cortex. J Comput Neurosci 36(2):297–319
Chizhov AV (2017) Conductance-based refractory density approach: comparison with experimental data and generalization to lognormal distribution of input current. Biol Cybern 111(5–6):353–364
Chizhov AV, Graham LJ (2007) Population model of hippocampal pyramidal neurons, linking a refractory density approach to conductance-based neurons. Phys Rev E 75:011924
Chizhov AV, Graham LJ (2008) Efficient evaluation of neuron populations receiving colored-noise current based on a refractory density method. Phys Rev E 77:011910
Chizhov AV, Graham LJ, Turbin AA (2006) Simulation of neural population dynamics with a refractory density approach and a conductance-based threshold neuron model. Neurocomputing 70:252–262
Chizhov AV, Rodrigues S, Terry JR (2007) A comparative analysis of a detailed neural population model and a mean-field EEG model. Phys Lett A 369:31–36
Chizhov AV, Amakhin DV, Zaitsev AV (2017) Computational model of interictal discharges triggered by interneurons. PLoS ONE 12(10):e0185752
Chizhov AV, Amakhin DV, Zaitsev AV (2019a) Spatial propagation of interictal discharges along the cortex. Biochem Biophys Res Commun 508:1245–1251
Chizhov AV, Amakhin DV, Zaitsev AV (2019b) Mathematical model of Na–K–Cl homeostasis in ictal and interictal discharges. PLoS ONE 14(3):e0213904
Coombes S, Thül R, Wedgwood KCA (2012) Nonsmooth dynamics in spiking neuron models. Physics D 241:2042–2057
Desroches M, Kaper TJ, Krupa M (2013) Mixed-mode bursting oscillations: dynamics created by a slow passage through spike-adding canard explosion in a square-wave burster. Chaos 23(4):046106
Desroches M, Guillamon A, Ponce E, Prohens E, Rodrigues S, Teruel AE (2016) Canards, folded nodes, and mixed-mode oscillations in piecewise-linear slow-fast systems. SIAM Rev 58(4):653–691
Dumont G, Payeur A, Longtin A (2017) A stochastic-field description of finite-size spiking neural networks. PLoS Comput Biol 13(8):e1005691
Eggert J, van Hemmen JL (2001) Modeling neuronal assemblies: theory and implementation. Neural Comput 13:1923–1974
Fenichel N (1979) Geometric singular perturbation theory for ordinary differential equations. J Differ Equ 31(1):53–98
Fernandez FR, White JA (2010) Gain control in CA1 pyramidal cells using changes in somatic conductance. J Neurosci 30(1):230–241
Freeman W (1972) Waves, pulses, and the theory of neural masses. Prog Theor Biol 2(1):1–10
Freeman W (1975) Mass action in the nervous system. Academic Press, Cambridge
Fröhlich F, Bazhenov M (2006) Coexistence of tonic firing and bursting in cortical neurons. Phys Rev E 74:031922
Gerstner W, Kistler WM (2002) Spiking neuron models: single neurons, populations, plasticity. Cambridge University Press
Gerstner W, Naud R (2009) How good are neuron models? Science 326:379–326
Izhikevich EM (2000) Neuronal excitability, spiking and bursting. Int J Bifurc Chaos 10(6):1171–1266
Izhikevich EM (2003) Simple model of spiking neurons. IEEE Trans Neural Netw 14(6):1569–1572
Jensen MS, Yaari Y (1997) Role of intrinsic burst firing, potassium accumulation, and electrical coupling in the elevated potassium model of hippocampal epilepsy. J Neurophysiol 77(3):1224–1233
Jones CKRT (1995) Geometric singular perturbation theory. In: Arnold L, Jones CKRT, Mischaikow K, Raugel G (eds) Dynamical systems, lecture notes in mathematics, vol 1609. Springer, Berlin, pp 44–118
Knight B (1972) Dynamics of encoding in a population of neurons. J Gener Physiol 59:734–766
Knight BW, Manin D, Sirovich L (1996) Dynamical models of interacting neuron populations. In: Gerf EC (ed) Symposium on robotics and cybernetics: computational engineering in systems applications. Cite Scientifique, Lille
Knight BW, Omurtag A, Sirovich L (2000) The approach of a neuron population firing rate to a new equilibrium: an exact theoretical result. Neural Comput 12:1045–1055
Ly C, Doiron B (2009) Divisive gain modulation with dynamic stimuli in integrate-and-fire neurons. PLoS Comput Biol 5(4):e1000365
Ly C, Tranchina D (2007) Critical analysis of dimension reduction by a moment closure method in a population density approach to neural network modeling. Neural comput 19(8):2032–2092
Ly C, Tranchina D (2009) Spike train statistics and dynamics with synaptic input from any renewal process: a population density approach. Neural Comput 21:360–396
Marten F, Rodrigues S, Benjamin O, Richardson MP, Terry JR (2009) Onset of polyspike complexes in a mean-field model of human electroencephalography and its application to absence epilepsy. Philos Trans R Soc A 367(1891):1145–1161
Montbrió E, Pazó D, Roxin A (2015) Macroscopic description for networks of spiking neurons. Phys Rev X 5:021028
Nykamp DQ, Tranchina D (2000a) A population density approach that facilitates large-scale modeling of neural networks: analysis and an application to orientation tuning. J Comput Neurosci 8:19–50
Nykamp D, Tranchina D (2000b) A population density approach that facilitates large-scale modeling of neural networks: analysis and application to orientation tuning. J Comput Neurosci 8:19–50
Ott E, Antonsen TM (2008) Low dimensional behavior of large systems of globally coupled oscillators. Chaos 18(3):037113
Perthame B, Tumuluri SK (2008) Selected topics in cancer modeling. In: Bellomo N, de Angelis E (eds) Nonlinear renewal equations. Springer, Berlin
Rinzel J (1987) A formal classification of bursting mechanisms in excitable systems. In: International congress of mathematicians, Berkeley, California, USA, August 3-11, 1986, volume II, pp 1578–1593. American Mathematical Society
Rodrigues S, Barton D, Szalai R, Benjamin O, Richardson MP, Terry JR (2009) Transitions to spike-wave oscillations and epileptic dynamics in a human cortico-thalamic mean-field model. J Comput Neurosci 27(3):507–526
Schwalger T, Deger M, Gerstner W (2017) Towards a theory of cortical columns: from spiking neurons to interacting neural populations of finite size. PLoS Comput Biol 13(4):e1005507
Smirnova EY, Zaitsev AV, Kim KKh, Chizhov AV (2015) The domain of neuronal firing on a plane of input current and conductance. J Comput Neurosci 39(2):217–33
Tchumatchenko T, Malyshev A, Wolf F, Volgushev M (2011) Ultrafast population encoding by cortical neurons. J Neurosci 31(34):12171–12179
Terman D (1991) Chaotic spikes arising from a model of bursting in excitable membranes. SIAM J Appl Math 51(5):1418–1450
Ventriglia F (1974) Kinetic approach to neural systems: I. Bull Math Biol 36(5–6):535–544
Wilson HR, Cowan JD (1972) Excitatory and inhibitory interactions in localized populations of model neurons. Biophys J 12(1):1–24
Yu Y, Shu Y, McCormick DA (2008) Cortical action potential back propagation explains spike threshold variability and rapid-onset kinetics. J Neurosci 28(29):7260–7272
Acknowledgements
This research is supported by the Basque Government through the BERC 2018-2021 programme and by the Spanish State Research Agency through BCAM Severo Ochoa excellence accreditation SEV-2017-0718 and through Project RTI2018-093860-B-C21 funded by (AEI/FEDER, UE) and acronym “MathNEURO”. SR would further like to acknowledge the support of Ikerbasque (The Basque Foundation for Science). Moreover, the research of AC was supported by the Russian Science Foundation Grant (Project 16-15-10201). The research of AG was supported by the Spanish Grant MINECO-FEDER-UE MTM-2015-71509-C2-2-R and the Catalan Grant Number 2017SGR1049. Finally, SR, FC and MD would like to acknowledge the support of Inria through the associated team project NeuroTransSF.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chizhov, A., Campillo, F., Desroches, M. et al. Conductance-Based Refractory Density Approach for a Population of Bursting Neurons. Bull Math Biol 81, 4124–4143 (2019). https://doi.org/10.1007/s11538-019-00643-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-019-00643-8