Abstract
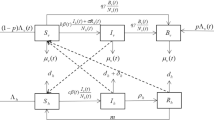
Malaria is an infectious disease caused by Plasmodium parasites and is transmitted among humans by female Anopheles mosquitoes. Climate factors have significant impact on both mosquito life cycle and parasite development. To consider the temperature sensitivity of the extrinsic incubation period (EIP) of malaria parasites, we formulate a delay differential equations model with a periodic time delay. We derive the basic reproduction ratio \(R_0\) and establish a threshold type result on the global dynamics in terms of \(R_0\), that is, the unique disease-free periodic solution is globally asymptotically stable if \(R_0<1\); and the model system admits a unique positive periodic solution which is globally asymptotically stable if \(R_0>1\). Numerically, we parameterize the model with data from Maputo Province, Mozambique, and simulate the long-term behavior of solutions. The simulation result is consistent with the obtained analytic result. In addition, we find that using the time-averaged EIP may underestimate the basic reproduction ratio.




Similar content being viewed by others
References
Ai S, Li J, Lu J (2012) Mosquito-stage-structured malaria models and their global dynamics. SIAM J Appl Math 72(4):1213–1237
Bacaër N, Ait Dads EH (2012) On the biological interpretation of a definition for the parameter \(R_0\) in periodic population models. J Math Biol 65:601–621
Bacaër N, Guernaoui S (2006) The epidemic threshold of vector-borne diseases with seasonality. J Math Biol 53:421–436
Beck-Johnson LM, Nelson WA, Paaijmans KP, Read AF, Thomas MB, Bjornstad ON (2013) The effects of temperature on Anopheles mosquito population dynamics and the potential for malaria transmission. PLoS ONE 8(11):e79276. doi:10.1371/journal.pone.0079276
Beier JC (1998) Malaria parasite development in mosquitoes. Annu Rev Entomol 43:519–543
Chamchod F, Britton NF (2011) Analysis of a vector-bias model on malaria transmission. Bull Math Biol 73:639–657
Chitnis N, Hyman JM, Cushing JM (2008) Determining important parameters in the spread of malaria through the sensitivity analysis of a mathematical model. Bull Math Biol 70:1272–1296
Craig MH, Snow RW, le Sueur D (1999) A climate-based distribution model of malaria transmission in sub-Saharan Africa. Parasitol Today 15(3):105–111
Diekmann O, Heesterbeek JAP, Metz JAJ (1990) On the definition and the computation of the basic reproduction ratio \(R_0\) in the models for infectious disease in heterogeneous populations. J Math Biol 28:365–382
Hale JK, Verduyn Lunel SM (1993) Introduction to functional differential equations. Springer, New York
Hirsch MW, Smith HL, Zhao X-Q (2001) Chain transitivity, attractivity and strong repellors for semidynamical systems. J Dyn Differ Equ 13:107–131
Inaba H (2012) On a new perspective of the basic reproduction number in heterogeneous environments. J Math Biol 22:113–128
Kesavan SK, Reddy NP (1985) On the feeding strategy and the mechanics of blood sucking in insects. J Theor Biol 113:781–783
Kingsolver JG (1987) Mosquito host choice and the epidemiology of malaria. Am Nat 130:811–827
Kot M (2001) Elements of mathematical ecology. Cambridge University Press, Cambridge
Lacroix R, Mukabana WR, Gouagna LC, Koella JC (2005) Malaria infection increases attractiveness of humans to mosquitoes. PLoS Biol 3:e298
Lou Y, Zhao X-Q (2010) A climate-based malaria transmission model with structured vector population. SIAM J Appl Math 70(6):2023–2044
Lou Y, Zhao X-Q (2017) A theoretical approach to understanding population dynamics with seasonal developmental durations. J Nonlinear Sci 27:573–603
Macdonald G (1957) The epidemiology and control of malaria. Oxford University Press, London
McCauley E, Nisbet RM, De Roos AM, Murdoch WW, Gurney WSC (1996) Structured population models of herbivorous zooplankton. Ecol Monogr 66:479–501
Molnár PK, Kutz SJ, Hoar BM, Dobson AP (2013) Metabolic approaches to understanding climate change impacts on seasonal host-macroparasite dynamics. Ecol Lett 16:9–21
Ngarakana-Gwasira ET, Bhunu CP, Mashonjowa E (2014) Assessing the impact of temperature on malaria transmission dynamics. Afr Mat 25:1095–1112
Nisbet RM, Gurney WS (1982) Modelling fluctuating populations. The Blackburn Press, Newark
Nisbet RM, Gurney WS (1983) The systematic formulation of population models for insects with dynamically varying instar duration. Theor Popul Biol 23:114–135
Omori R, Adams B (2011) Disrupting seasonality to control disease outbreaks: the case of koi herpes virus. J Theor Biol 271:159–165
Posny D, Wang J (2014) Computing the basic reproductive numbers for epidemiological models in nonhomogeneous environments. Appl Math Comput 242:473–490
Rittenhouse MA, Revie CW, Hurford A (2016) A model for sea lice (Lepeophtheirus salmonis) dynamics in a seasonally changing environment. Epidemics 16:8–16
Ross R (1911) The prevention of malaria, 2nd edn. Murray, London
Smith HL (1995) Monotone dynamical systems: an introduction to the theory of competitive and cooperative systems. American Mathematical Society, Providence
Thieme HR (2009) Spectral bound and reproduction number for infinite-dimensional population structure and time heterogeneity. SIAM J Appl Math 70:188–211
van den Driessche P, Watmough J (2002) Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math Biosci 180:29–48
Walter W (1997) On strongly monotone flows. Ann Polon Math LXVI:269–274
Wang W, Zhao X-Q (2008) Threshold dynamics for compartmental epidemic models in periodic environments. J Dyn Differ Equ 20:699–717
Wang X, Zhao X-Q (2017) A periodic vector-bias malaria model with incubation period. SIAM J Appl Math 77:181–201
Wang X, Zhao X-Q. A climate-based malaria model with the use of bed nets (submitted)
World Health Organisation (2015) Global Malaria Programme, World Malaria Report
Wu X, Magpantay FMG, Wu J, Zou X (2015) Stage-structured population systems with temporally periodic delay. Math Meth Appl Sci 38:3464–3481
Zhao X-Q (2003) Dynamical systems in population biology. Springer, New York
Zhao X-Q (2017) Basic reproduction ratios for periodic compartmental models with time delay. J Dyn Differ Equ 29:67–82
Acknowledgements
We are very grateful to two anonymous referees for their careful reading and helpful suggestions which led to an important improvement of our original manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work is supported in part by the NSERC of Canada.
Rights and permissions
About this article
Cite this article
Wang, X., Zhao, XQ. A Malaria Transmission Model with Temperature-Dependent Incubation Period. Bull Math Biol 79, 1155–1182 (2017). https://doi.org/10.1007/s11538-017-0276-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-017-0276-3