Abstract
Allee effects increasingly are recognized as influential determinants of population dynamics, especially in disturbed landscapes. We developed a predator–prey metapopulation model to study the impact of an Allee effect on predator–prey. The model incorporates habitat destruction and predators with imperfect information about prey distribution. Criteria are established for the existence and stability of equilibria, and the possible existence of a limit cycle is discussed. Numerical bifurcation analysis of the model is carried out to examine the impact of Allee effects as well as other key processes on trophic dynamics. Inclusion of Allee effects produces a richer array of dynamics than earlier models in which it was absent. When prey interacts with generalist predators, Allee effects operate synergistically to depress prey populations. Allee effects are more likely to depress occupancy levels when destruction of habitat patches is moderate; at severe levels of destruction, Allee effects are swamped by demographic effects of habitat loss. Stronger Allee effects correspond to lower thresholds of predator colonization rates at which prey become extinct. We discuss implications of our model for conservation of rare species as well as pest management via biocontrol.











Similar content being viewed by others

References
Allee WC (1931) Animal aggregations: a study in general sociology. University of Chicago Press, Chicago
Amarasekare P (1998) Allee effects in metapopulation dynamics. Am Nat 152(2):298–302
Bascompte J, Solé RV (1998) Effects of habitat destruction in a prey-predator metapopulation model. J Theor Biol 195(3):383–393
Brassil CE (2001) Mean time to extinction of a metapopulation with an Allee effect. Ecol Model 143:9–16
Clavel J, Julliard R, DeVictor V (2011) Worldwide decline of specialist species: toward a global functional homogenization? Front Ecol Environ 9:222–228
Essl F, Dullinger S, Rabitsch W, Hulme PE, Pyšek P, Wilson JRU, Richardson DM (2015) Historical legacies accumulate to shape future biodiversity in an era of rapid global change. Divers Distrib 21:534–547
Estavillo C, Pardini R, Bernardo da Rocha PL (2013) Forest loss and the biodiversity threshold: an evaluation considering species habitat requirements and the use of matrix habitats. PLoS One 8(12):e82369
Farias AA, Jaksic FM (2011) Low functional richness and redundancy of a predator assemblage in native forest fragments of Chiloe Island, Chile. J Anim Ecol 80:809–817
Fauvergue X, Vercken E, Malausa T, Hufbauer RA (2012) The biology of small, introduced populations, with special reference to biological control. Evol Appl 5(5):424–443
Fernandez AA, Hance T, Deneubourg JL (2012) Interplay between Allee effects and collective movement in metapopulations. Oikos 121:813–822
Gascoigne J, Lipcius RN (2004) Allee effects driven by predation. J Appl Ecol 41:801–810
Hanski I (1999) Metapopulation ecology. Oxford University Press, Oxford
Hart EM, Aviles L (2014) Reconstructing local population dynamics in noisy metapopulations—the role of random catastrophes and Allee effects. PLoS One 9(10):e110049. doi:10.1371/journal.pone.0110049
Kuussaari M, Bommarco R, Heikkinen RK, Helm A, Krauss J, Lindborg R, Öckinger E, Pärtel M, Pino J, Rodà F, Steffan-Dewener I (2009) Extinction debt: a challenge for biodiversity conservation. Trends Ecol Evol 24:564–571
Levins R (1969) Some demographic and genetic consequences of environmental heterogeneity for biological control. Bull Entomol Soc Am 15:237–240
Monastersky R (2014) Life—a status report. Nature 516(7530):159
Smyser TJ, Johnson SA, Page LK, Hudson CM, Rhodes OE Jr (2013) Use of experimental translocations of Allegheny woodrat to decipher causal agents of decline. Conserv Biol 27:752–762
Stephens PA, Sutherland WJ, Freckleton RP (1999) What is the Allee effect? Oikos 87:185–190
Stephens PA, Sutherland WJ (1999) Consequences of the Allee effect for behavior, ecology and conservation. Trends Ecol Evol 14:401–405
Swihart RK, Feng ZL, Slade NA, Mason DM, Gehring TM (2001) Effects of habitat destruction and resource supplementation in a predator-prey metapopulation model. J Theor Biol 210:287–303
Swihart RK, Gehring TM, Kolozsvary MB, Nupp TE (2003) Responses of ”resistant” vertebrates to habitat loss and fragmentation: the importance of niche breadth and range boundaries. Divers Distrib 9:1–18
Swihart RK, Lusk JJ, Duchamp JE, Rizkalla CE, Moore JE (2006) The roles of landscape context, niche breadth, and range boundaries in predicting species responses to habitat alteration. Divers Distrib 12:277–287
Taylor CM, Hastings A (2005) Allee effects in biological invasions. Ecol Lett 8:895–908
Weterings R, Umponstira C, Buckley HL (2014) Container-breeding mosquitoes and predator community dynamics along an urban-forest gradient: the effects of habitat type and isolation. Basic Appl Ecol 15:486–495
Zhong SR, Wang G (2004) Allee-like effects of metapopulation. Math Biosci 189:103–113
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by NSFC-11271065, RFPD-20130043110001, and RFCP-CMSP (MF).
Appendices
Appendix 1: The Proof of Lemma 2.1
Since \(E^{*}(x^{*},y^{*})\) is an interior equilibrium of (6), one has
Then
Hence, if
then \(E^{*}\) is l.a.s.
Note that, if \(x^{*}\ge (a+p)/2\), then (12) is satisfied and hence \(E^{*}\) is l.a.s.
Now we deal with the case of \(x^{*}<(a+p)/2\). If
then \(\det (J(E^{*}(x^{*},y^{*})))>0\). Moreover, if
then \(\mathrm{Tr}(J(E^{*}(x^{*},y^{*})))<0\). Therefore, \(E^{*}\) is l.a.s. In addition, if
then \(\mathrm{Tr}(J(E^{*}(x^{*},y^{*})))>0\). Hence, \(E^*\) is an unstable node or focus. If
then \(\det (J(E^{*}(x^{*},y^{*})))<0\). Therefore, \(E^{*}\) is a saddle. This completes the proof. \(\square \)
Appendix 2: Existence of Limit Cycle
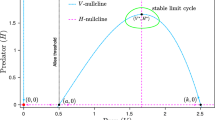
If (6) admits a unique non-saddle interior equilibrium \(E^*\) being unstable (e.g., (10) is satisfied), then (6) has one limit cycle in \(\Omega \) with \(E^*\) lying in its interior.
Proof
Define
where \(y_0>\max \left\{ q+{\psi p}/{c_{y}}, {c_{x}(a-p)^{2}}/{(4\mu p)}\right\} \). We prove the existence of a limit cycle by the Poincaré–Bendixson Theorem and show that \(\Omega \) is the desired trapping region. Let us now consider the behavior of the vector field on the boundary of \(\Omega \). On the straight line \(x=p\), one has
so that the vector field points right-to-left into the interior of \(\Omega \). On \(y=y_0\) with \(x<p\), we have
so that the vector field points top-down into the interior of \(\Omega \). In addition, \(x=0\) and \(y=0\) are trajectories of (6). Therefore, \(x=p, y=y_0\), positive x-axis and positive y-axis constitute the outer boundary curve. Since the unique interior equilibrium is unstable, by the Poincaré–Bendixson theorem for annular regions, (6) has at least one stable limit cycle in \(\Omega \) containing \(E^*\) in its interior. \(\square \)
Rights and permissions
About this article
Cite this article
Fan, M., Wu, P., Feng, Z. et al. Dynamics of Predator–Prey Metapopulations with Allee Effects. Bull Math Biol 78, 1727–1748 (2016). https://doi.org/10.1007/s11538-016-0197-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-016-0197-6