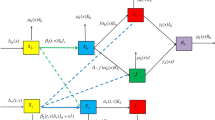
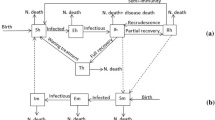
Abstract
Human habitat connectivity, movement rates, and spatial heterogeneity have tremendous impact on malaria transmission. In this paper, a deterministic system of differential equations for malaria transmission incorporating human movements and the development of drug resistance malaria in an \(n\) patch system is presented. The disease-free equilibrium of the model is globally asymptotically stable when the associated reproduction number is less than unity. For a two patch case, the boundary equilibria (drug sensitive-only and drug resistance-only boundary equilibria) when there is no movement between the patches are shown to be locally asymptotically stable when they exist; the co-existence equilibrium is locally asymptotically stable whenever the reproduction number for the drug sensitive malaria is greater than the reproduction number for the resistance malaria. Furthermore, numerical simulations of the connected two patch model (when there is movement between the patches) suggest that co-existence or competitive exclusion of the two strains can occur when the respective reproduction numbers of the two strains exceed unity. With slow movement (or low migration) between the patches, the drug sensitive strain dominates the drug resistance strain. However, with fast movement (or high migration) between the patches, the drug resistance strain dominates the drug sensitive strain.







Similar content being viewed by others
References
Adams B, Kapan DD (2009) Man bites mosquito: understanding the contribution of human movement to vector-borne disease dynamics. PLoS One 4(8):e6763
Anderson RM, May R (1991) Infectious diseases of humans. Oxford University Press, New York
Aneke SJ (2002) Mathematical modelling of drug resistant malaria parasites and vector populations. Math Methods Appl Sci 25:335–346
Ariey F, Robert V (2003) The puzzling links between malaria transmission and drug resistance. Trends Parasitol 19(4):158–160
Ariey F, Duchemin JB, Robert V (2003) Metapopulation concepts applied to falciparum malaria and their impact on the emergence and spread of chloroquine resistance. Infect Genet Evol 2:185–192
Arino J, van den Driessche P (2003) A multi-city epidemic model. Math Popul Stud 10:175–193
Arino J, van den Driessche P (2003) The basic reproducton number in a multi-city compartment model. LNCIS 294:135–142
Arino J, Ducrot A, Zongo P (2012) A metapopulation model for malaria with transmission-blocking partial immunity in hosts. J Math Biol 64(3):423–448
Auger P, Kouokam E, Sallet G, Tchuente M, Tsanou B (2008) The Ross–Macdonald model in a patchy environment. Math Biosci 216:123–131
Bacaer N, Sokna C (2005) A reaction–diffusion system modeling the spread of resistance to an antimalarial drug. Math Biosci Eng 2:227–238
Bowman C, Gumel AB, van den Driessche P, Wu J, Zhu H (2005) A mathematical model for assessing control strategies against West Nile virus. Bull Math Biol 67:1107–1133
Breman JG, Holloway CN (2007) Malaria surveillance counts. Am J Trop Med Hyg 77:36–47
Bush AO, Fernandez JC, Esch GW, Seedv JR (2001) Parasitism: the diversity and ecology of animal parasites, 1st edn. Cambridge University Press, Cambridge
Carrara VI, Sirilak S, Thonglairuam J, Rojanawatsirivet C (2006) Deployment of early diagnosis and mefloquine–artesunate treatment of falciparum malaria in Thailand: The Tak malaria initiative. PLoS Med 3(6):e183
Carrara VI, Zwang J, Ashley EA, Price RN et al (2009) Changes in the treatment responses to artesunate–mefloquine on the northwestern border of Thailand during 13 Years of continuous deployment. PLoS One 4:e4551
Cheeseman IH, Miller BA, Nair S, Nkhoma S et al (2012) A major genome region underlying artemisinin resistance in malaria. Science 336:79–82. doi:10.1126/science.1215966
Chitnis N, Cushing JM, Hyman JM (2006) Bifurcation analysis of a mathematical model for malaria transmission. SIAM J Appl Math 67:24–45
Chiyaka C, Tchuenche JM, Garira W, Dube S (2008) A mathematical analysis of the effects of control strategies on the transmission dynamics of malaria. Appl Math Comput 195:641–662
Cosner C, Beier JC, Cantrell RS, Impoinvil D, Kapitanski L, Potts MD, Troyo A, Ruan S (2009) The effects of human movement on the persistence of vector-borne diseases. J Theor Biol 258(4):550–560. doi:10.1016/j.jtbi.2009.02.016
Denis MB, Tsuyuoka R, Lim P, Lindegardh N et al (2006) Efficacy of artemetherlumefantrine for the treatment of uncomplicated falciparum malaria in northwest Cambodia. Trop Med Int Health 11(12):1800–1807. doi:10.1111/j.1365-3156.2006.01739.x
Diekmann O, Heesterbeek JAP, Metz JAP (1990) On the definition and computation of the basic reproduction ratio \(R_0\) in models for infectious diseases in heterogeneous populations. J Math Biol 28:503–522
Dietz K (1988) Mathematical models for transmission and control of malaria. In: Wensdorfer WH, McGregor I (eds) Malaria. Churchill Livingstone, Edinburgh, pp 1091–1133
Dondorp AM, Nosten F, Yi P, Das D et al (2009) Artemisinin resistance in Plasmodium falciparum malaria. The N Engl J Med 361(5):455–467
Esteva L, Vargas C (2000) Influence of vertical and mechanical transmission on the dynamics of dengue disease. Math Biosci 167:51–64
Esteva L, Gumel AB (2009) Qualitative study of transmission dynamics of drug-resistant malaria. Math Comput Model 50:611–630
Feng Z, Yinfei Y, Zhu H (2004) Fast and slow dynamics of malaria and the s-gene frequency. J Dyn Differ Equ 16:869–895
Flahault A, Le Menach A, McKenzie EF, Smith DL (2005) The unexpected importance of mosquito oviposition behaviour for malaria: non-productive larval habitats can be sources for malaria transmissionn. Malar J 4(1):23
Grimwade K, French N, Mbatha DD, Zungu DD, Dedicoat M, Gilks CF (2004) HIV infection as a cofactor for severe falciparum malaria in adults living in a region of unstable malaria transmission in South Africa. AIDS 18:547–554
Hastings IM (1997) A model for the origins and spread of drug resistant malaria. Parasitol 115:133–141
Hethcote HW (2000) The mathematics of infectious diseases. SIAM Rev 42(4):599–653
Hsieh Y, van den Driessche P, Wang L (2007) Impact of travel between patches for spatial spread of disease. Bull Math Biol 69:1355–1375
Koella JC, Antia R (2003) Epidemiological models for the spread of antimalarial resistance. Malar J 2:3
Lakshmikantham V, Leela S, Martynyuk AA (1989) Stability analysis of nonlinear systems. Marcel Dekker, New York and Basel
Le Menach A, Ellis Mckenzie F (2005) The unexpected importance of mosquito oviposition behaviour for malaria: non-producive larval habitats can be sources for malaria transmission. Malar J 4(1):23
Lindsay SW, Martens WJM (1998) Malaria in the African highlands: past, present and future. Bull WHO 76:33–45
Mackinnon MJ (2005) Drug resistance models for malaria. Acta Trop 94:207–217
Mbogob CM, Gu W, Killeena GF (2003) An individual-based model of Plasmodium falciparum malaria transmission on the coast of Kenya. Trans R Soc Trop Med Hyg 97:43–50
Molineaux L, Gramiccia G (1980) The Garki project. World Health Organization, Geneva
Niger AM, Gumel AB (2008) Mathematical analysis of the role of repeated exposure on malaria transmission dynamics. Differ Equ Dyn Syst 16(3):251–287
Phyo AP, Nkhoma S, Stepniewska K, Ashley EA et al (2012) Emergence of artemisinin-resistant malaria on the western border of Thailand: a longitudinal study. Lancet 379:1960–1966. doi:10.1016/S0140-6736(12)60484-X
Pongtavornpinyo W, Yeung S, Hastings IM, Dondorp AM, Day NPJ, White NJ (2008) Spread of anti-malarial drug resistance: mathematical model with implications for ACT drug policies. Malar J 7:229
Prosper OF, Ruktanoncha N, Martcheva M (2012) Assessing the role of spatial heterogeneity and human movement in malaria dynamics and control. J Theor Biol 303:1–14
Rodrguez DJ, Torres-Sorando L (2001) Models of infectious diseases in spatially heterogeneous environments. Bull Math Biol 63:547–571
Ross R (1911) The prevention of malaria. John Murray, London
Salmani M, van den Driessche P (2006) A model for disease transmission in a patchy environment. Discret Contin Dyn Syst Ser B 6:185–202
Smith HL, Waltman P (1995) The theory of the chemostat. Cambridge University Press, Cambridge
Smith DL, Mckenzie EF (2004) Statics and dynamics of malaria infection in anopheles mosquito. Malar J 3:13
Smith DL, Dushoff J, Ellis Mckenzie F (2005) The risk of a mosquito-borne infection in a heterogeneous environnement. PLoS Biol 2:1957–1964
Smith T, Killen GF, Maire N, Ross A, Molineaux L, Tediosi F, Hutton G, Utzinger J, Dietz K, Tanner M (2006) Mathematical modelling of the impact of malaria vaccines on the clinical epidemiology and natural history of Plasmodium falciparum malaria: Overview. Am J Trop Med Hyg 75:1–10
Snow RW, Omumbo J (2006) In: Jamison DT et al (eds) Malaria, in diseases and mortality in Sub-Saharan Africa. The World Bank, Washington
US Census Bureau International database (2010)
van den Driessche P, Watmough J (2002) Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math Biosci 180:29–48
World Health Organization (WHO) Malaria (2010) http://www.who.int/mediacentre/factsheets/fs094/en/
World Health Organization (WHO) World malaria report 2009
Zhou G, Minakawa N, Githeko AK, Yan G (2004) Association between climate variability and malaria epidemics in the east African highlands. Proc Natl Acad Sci USA 101:2375–2380
Acknowledgments
The author likes to thank the anonymous reviewers for the constructive comments.
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix 1: Proof of Theorem 4
Proof
To prove Theorem 4, we follow the method given in Prosper et al. (2012). Assume \(\mathcal {R}^2_{0_{_{DR_1}}} >\mathcal {R}^2_{0_{_{DR_2}}}\), by this assumption and from Eq. (16) it follows that \(p_1k_4> p_2k_2\). Evaluating the reproduction number \(\mathcal {R}^2_{0_{_{DR}}}\) at the boundary of the domain \((\psi _{_{12}},\psi _{_{21}}) \in [0,\infty ) \times [0,\infty )\). We have,
Since by assumption \(p_1k_4 > p_2k_2\), and because \(q_2 = k_4 + \psi _{_{12}} \ge k_4\), we know that \(p_1q_2 - p_2k_2 > 0\), and so \(|p_1q_2 - p_2k_2| = p_1q_2 - p_2k_2\). Thus, Eq. (26) simplifies to \(\mathcal {R}^2_{0_{_{DR}}}(\psi _{_{12}}, 0) = \mathcal {R}^2_{0_{_{DR_1}}}(0,0)\) for all \(\psi _{_{12}} \in [0,\infty )\). Similarly,
Hence, \(\mathcal {R}^2_{0_{_{DR_2}}}(0,0) \le \mathcal {R}^2_{0_{_{DR}}}(0, \psi _{_{21}}) \le \mathcal {R}^2_{0_{_{DR_1}}}(0,0)\), for all \(\psi _{_{21}} \in [0,\infty )\).
Now, evaluating the reproduction number \(\mathcal {R}^2_{0_{_{DR}}}\) in the interior of the domain \((\psi _{_{12}},\psi _{_{21}}) \in [0,\infty ) \times [0,\infty )\). Consider the function
\(f\) is the characteristic polynomial of the next-generation matrix used to derive \(\mathcal {R}^2_{0_{_{DR}}}(\psi _{_{12}}, \psi _{_{21}})\) in Eq. (15). \(\mathcal {R}^2_{0_{_{DR}}}\) is the larger of the two roots of \(f(x)\). Consequently, \(f(\mathcal {R}^2_{0_{_{DR}}}) = 0\) and \(f^\prime (\mathcal {R}^2_{0_{_{DR}}}) > 0\). Thus, for any real number \(y\) for which the inequality \(f(y) < 0\) holds, implies that \(y<\mathcal {R}^2_{0_{_{DR}}}\). However, if \(f(y) > 0\) and \(f^\prime (y) > 0\), then \(y>\mathcal {R}^2_{0_{_{DR}}}\). Now, suppose \(\psi _{_{12}}\) and \(\psi _{_{21}}\) are positive, then it follows that
We can similarly show that
Thus, since \( (p_1k_4 - p_2k_2)>0\) by assumption, we have that \(f(\mathcal {R}^2_{0_{_{DR_1}}}) > 0\), \(f(\mathcal {R}^2_{0_{_{DR_2}}}) <0\) and \(f\bigg (\frac{\mathcal {R}^2_{0_{_{DR_1}}}}{1 + \frac{\psi _{_{21}}}{k_2}}\bigg )<0\).
Differentiating \(f(y)\) we have \(f^\prime (y) = 2\sigma _2y - (p_1q_2 + p_2q_1)\). Thus,
Since \((p_1k_4 - p_2k_2)>0\), it follows that \(f^\prime (\mathcal {R}^2_{0_{_{DR_1}}})>0\). Hence, it follows that, for \(\psi _{_{12}}\) and \(\psi _{_{21}}\) positive, \(f(\mathcal {R}^2_{0_{_{DR_2}}})<0\) and \(f\bigg (\frac{\mathcal {R}^2_{0_{_{DR_1}}}}{1 + \frac{\psi _{_{21}}}{k_2}}\bigg )<0\) implies that \(\mathcal {R}^2_{0_{_{DR}}} > \max \bigg \{\frac{\mathcal {R}^2_{0_{_{DR_1}}}}{1 + \frac{\psi _{_{21}}}{k_2}},\mathcal {R}^2_{0_{_{DR_2}}}\bigg \}.\) Since \(f(\mathcal {R}^2_{0_{_{DS_1}}})>0\) and \(f^\prime (\mathcal {R}^2_{0_{_{DS_1}}})>0\) we have that \(\mathcal {R}^2_{0_{_{DR}}}<\mathcal {R}^2_{0_{_{DS_1}}}\).
Hence, for all \(\psi _{_{12}}\) and \(\psi _{_{21}}\) positive, \(\max \bigg \{\frac{\mathcal {R}^2_{0_{_{DR_1}}}(0,0)}{1 + \frac{\psi _{_{21}}}{k_2}},\mathcal {R}^2_{0_{_{DS_2}}}(0,0)\bigg \} < \mathcal {R}^2_{0_{_{DS}}}(\psi _{_{12}},\psi _{_{21}})<\mathcal {R}^2_{0_{_{DR_1}}}(0,0).\) \(\square \)
Appendix 2: Proof of Theorem 6
Proof
In the proof of Theorem 4 we have that \(\mathcal {R}_{0_{_{DR}}}(0,\psi ) = \max \bigg \{\frac{\mathcal {R}_{0_{_{DR_1}}}(0,0)}{1 + \frac{\psi _{_{21}}}{k_2}},\mathcal {R}_{0_{_{DR_2}}}(0,0)\bigg \}\) and \(\mathcal {R}_{0_{_{DR}}}(\psi _{_{12}},\psi ) >\max \bigg \{\frac{\mathcal {R}_{0_{_{DR_1}}}(0,0)}{1 + \frac{\psi _{_{21}}}{k_2}}, \mathcal {R}_{0_{_{DR_2}}}(0,0)\bigg \} \) for \(\psi _{_{12}} > 0\). Thus \(\mathcal {R}_{0_{_{DR}}}(\psi _{_{12}},\psi )\ge \mathcal {R}_{0_{_{DR}}}(0,\psi )\) for all \( \psi _{_{12}}\ge 0\). Thus, for \(\mathcal {R}_{0_{_{DR}}}(\psi _{_{12}},\psi )\) to be an increasing function in \(\psi _{_{12}}\) we need to show that \(\mathcal {R}_{0_{_{DR}}}(\psi _{_{12}},\psi )\) is monotone in \(\psi _{_{12}}\).
Also, from Theorem 4 we also know that \(\mathcal {R}_{0_{_{DR}}}(\psi ,0) = \mathcal {R}_{0_{_{DR_1}}}(0,0,) \ge \mathcal {R}_{0_{_{DR}}}(\psi ,\psi _{_{21}})\) for all non-negative \(\psi _{_{21}}\). Again, we only need to show that \(\mathcal {R}_{0_{_{DR}}}(\psi ,\psi _{_{21}})\) is monotone in \(\psi _{_{21}}\) in order to show that it is a decreasing function in \(\psi _{_{12}}\).
To show that \(\mathcal {R}_{0_{_{DR}}}(\psi _{_{12}},\psi _{_{21}})\) is monotone, we first show that \(\mathcal {R}_{0_{_{DR}}}(\psi _{_{12}},\psi )\) is monotone in \(\psi _{_{12}}\). Since \(\mathcal {R}_{0_{_{DR}}}(\psi _{_{12}},\psi )\) is continuous in \(\psi _{_{12}}\), it is monotone with respect to \(\psi _{_{12}}\) if for every \(B \in (0,\infty )\) such that \(\mathcal {R}_{0_{_{DR}}}(\psi _{{12}},\psi ) = B\) has a non-negative solution \(\psi _{{12}} \in [0,\infty )\), then this solution is unique. Suppose \(\mathcal {R}_{0_{_{DR}}}(\psi _{_{12}},\psi ) = B\). The reproduction number \(\mathcal {R}_{0_{_{DR}}}(\psi _{_{12}},\psi )\) can be written as
where \(s = p_1q_2 + p_2q_1 = p_1(k_4 + \psi _{_{12}}) + p_2(k_2 + \psi )\) and \(\sigma _2 = k_2k_2 + k_2\psi _{_{12}} + \tau _2\psi .\)
Hence,
Equation (28) implies that
Note that both \(\sigma _2\) and \(s\) are linear in \(\psi _{_{12}}\). Thus, Eq. (29) is linear in \(\psi _{_{12}}\), implying that if there exists a \(\psi _{_{12}} \in [0,\infty )\) that is, a solution to Eq. (29), then this solution is unique. Hence, \(\mathcal {R}_{0_{_{DR}}}(\psi _{_{12}},\psi )\) is monotone for each \(\psi \in [0,\infty )\). By the same argument, \(\mathcal {R}_{0_{_{DR}}}(\psi ,\psi _{_{21}})\) is monotone for each \(\psi \in [0,\infty ))\). Since \(\mathcal {R}_{0_{_{DR}}}(\psi _{_{12}},\psi )\) is monotone for non-negative \(\psi _{_{12}}\) and \(\mathcal {R}_{0_{_{DR}}}(0,\psi ) \le \mathcal {R}_{0_{_{DR}}}(\psi _{{12}},\psi )\), for each fixed \(\psi _{_{21}} = \psi \in [0,\infty )\), \(\mathcal {R}_{0_{_{DR}}}(\psi _{{12}},\psi _{{21}})\) is an increasing function of \(\psi _{{12}}\). Likewise, since \(\mathcal {R}_{0_{_{DR}}}(\psi ,0) \ge \mathcal {R}_{0_{_{DR}}}(\psi ,\psi _{{21}})\) for non-negative \(\psi _{_{21}}\), for each fixed \(\psi _{{12}} = \psi \in [0,\infty )\), \(\mathcal {R}_{0_{_{DS}}}(\psi _{{12}},\psi _{{21}})\) is a decreasing function of \(\psi _{{21}}\). \(\square \)
Appendix 3: Proof of Theorem 8
Proof
The stability of the drug sensitive-only boundary equilibrium is explored by evaluating the Jacobian of system (19) at the boundary equilibrium \({\mathcal E}_{1S}\), taking the following order of the coordinates \(I_1,I_{v1},J_1,J_{v1}\).
The Jacobian is given as
where
The eigenvalues of \(\displaystyle J_S({{\mathcal E}_{1S}})\) are given by the eigenvalues of \(J_{S_{1}} \) and \(J_{S_{4}} \). The eigenvalues of \(J_{S_{1}} \) are determined from the roots of the characteristics equations obtained by substituting in \(J_{S_{1}}\), \(I^{**}_1,~I^{**}_{v1}\)
It follows that for \(\mathcal {R}^2_{0_{_{DS_1}}} > 1\), the coefficients of the characteristics equation \(P_{S_{11}} \) is positive. And thus, satisfies the Routh–Hurwitz criteria, for stability.
For the matrix \(J_{S_{4}}\), the eigenvalues are given by the roots of the characteristics equations obtained by substituting in \(J_{S_{4},}\) \(I^{**}_1,~I^{**}_{v1}\)
The roots of the characteristics equation \(P_{S_{41}}\) have negative real parts if and only if \(\mathcal {R}^2_{0_{_{DS_1}}} > \mathcal {R}^2_{0_{_{DR_1}}}\). \(\square \)
Appendix 4: Proof of Theorem 9
Proof
The stability of the drug resistance-only boundary equilibrium is explored by evaluating the Jacobian of system (20) at the boundary equilibrium \({\mathcal E}_{1R}\), using the following order of coordinates, \(I_1,I_{v1},\) \(J_1,J_{v1}\).
The Jacobian is given as
where
and
substituting \(J^{**}_1\) and \(J^{**}_{v1}\) into \(J_{R_{1}}\) and \(J_{R_{4}}\) gives the following characteristics equations,
and
By Routh–Hurwitz criteria for stability, it follows that the roots of the characteristics equations \(P_{R_{1}}\) and \(P_{R_{4}}\) have negative real parts if and only if \(\mathcal {R}^2_{0_{_{DR_1}}} > \mathcal {R}^2_{0_{_{DS_1}}}\). \(\square \)
Appendix 5: Proof of Theorem 10
Proof
To prove Theorem 10, we follow the method given in Esteva and Gumel (2009) and Esteva and Vargas (2000). The method essentially entails proving that the linearization of the model system (14), around the co-existence equilibrium \({\mathcal E}_{1}\), has no solutions of the form
with \(\tilde{Z}= \{Z_1, Z_2, Z_3, Z_4\}, ~~Z_i \in C, \omega \in C\), and Re \(\omega \ge 0\) (the implication of this is that the eigenvalues of the characteristic polynomial associated with the linearized model will have negative real part; in which case, the equilibrium \({\mathcal E}_{1}\) is LAS).
Let \(I^{**}_1,J^{**}_1,I^{**}_{v1},J^{**}_{v1}\) denote the coordinates of the co-existence equilibrium, \({\mathcal E}_{1}\). Substituting a solution of the form (34) into the linearized system of (17) around \({\mathcal E}_{1}\) gives the following system of linear equations
where \( p_1=\gamma _1 + \xi _1+\mu _h + \delta _{I_1}, ~p_2=\tau _1 + \mu _h + \delta _{J_1}\). Simplifying (35), gives the equivalent system
Adding Eqs. (36) and (37), (38) and (39) gives the system
where
and
Note that \(S^{**}_1 = (N^{**}_{h1}-I^{**}_{1}-J^{**}_{1}),~~S^{**}_{v1} = (N^{**}_{v1}-I^{**}_{v1}-J^{**}_{v1})\) above. It should further be noted that the matrix \(H\) has non-negative entries, and the equilibrium \( {\mathcal E}_{1} = (I^{**}_1,J^{**}_1,I^{**}_{v1},J^{**}_{v1})\) satisfies \( {\mathcal E}_{1} = H {\mathcal E}_{1}\). Furthermore, since the coordinates of \( {\mathcal E}_{1}\) are all positive, it follows then that if \(\tilde{Z}\) is a solution of (34), then it is possible to find a minimal positive real number \(r\) such that
Observe that \(r\) is also the minimal positive \(r\) such that \(|Z_1|+|Z_2| \le r(I^{**}_1+J^{**}_1)\) and \(|Z_3|+|Z_4| \le r(I^{**}_{v1}+J^{**}_{v1})\). We want to show that Re \(\omega < 0\). Assume the contrary (i.e., Re \(\omega \ge 0\)), we consider two cases: \(\omega = 0\) and \(\omega \ne 0\). Assume the first case \(\omega = 0\). Then, (35) is a homogeneous linear system in the variables \(Z_i ~(i = 1,\ldots ,4)\). The determinant of this system corresponds to that of the Jacobian of system (17) evaluated at \(\mathcal {E}_{1}\), which is given by
Consequently, the system (35) can only have the trivial solution \(\tilde{Z} = \bar{0}\).
The case \(\omega \ne 0\), is considered next. In this case, Re \(G_i(\omega )\ge 0,~~i=1,\ldots ,4\), since, by assumption, Re \(\omega >0\). It is easy to see that this implies \(|1+G(\omega )| > 1\) for all \(i\). Now, define \(G(\omega ) = \min |1+G_i(\omega )|, ~~i = 1,\ldots , 4\). Then, \(G(\omega ) > 1\), and therefore \(\frac{r}{G(\omega )} < r\). The minimality of \(r\) implies that \(|\tilde{Z}| > \frac{r}{G(\omega )} \mathcal {E}_{1}\). But, on the other hand, taking norms on both sides of the second equation of (40), and using the fact that \(H\) is non-negative, we obtain
Then, it follows from the above inequality that \(|Z|\le \frac{r}{G(\omega )} \mathcal {E}_{1}\) which is a contradiction. Hence, Re \(\omega < 0\), which implies that \(\mathcal {E}_{1}\) is locally asymptotically stable. \(\square \)
Rights and permissions
About this article
Cite this article
Agusto, F.B. Malaria Drug Resistance: The Impact of Human Movement and Spatial Heterogeneity. Bull Math Biol 76, 1607–1641 (2014). https://doi.org/10.1007/s11538-014-9970-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-014-9970-6