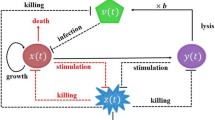
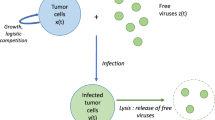
Abstract
Several viruses preferentially infect and replicate in cancer cells by usurping pathways that are defective in the tumor cell population. Such viruses have a potential as oncolytic agents. The aim of tumor virotherapy is that after injection of the replicating virus, it propagates in the tumor cell population with amplification. As a result, the oncolytic virus spreads to eradicate the tumor. The outcome of tumor virotherapy is determined by population dynamics and different from standard cancer therapy. Several models have been developed that provided considerable insights on the potential therapeutic scenarios. However, virotherapy is potentially risky since large amounts of a replicating virus are injected in the host with a risk of adverse effects. Therefore, the optimal dose, number of doses, and timing are expected to play an important role on the outcome both for the tumor and the host. In the current work, we combine a model of the dynamics of tumor virotherapy that was validated with experimental data with optimization theory to illustrate how we can improve the outcome of tumor therapy. In this first report, we demonstrate that (i) in most circumstances, anything more than two administrations of a vector is not helpful, (ii) correctly timed delivery of the virus provides superior results compared to regularly scheduled therapy or continuous infusion, (iii) a second dose of virus that is not properly timed leads to a worse outcome compared to a single dose of virus, and (iv) it is less costly to treat larger tumors.
Similar content being viewed by others
References
Anderson, B.D., Nakamura, T., Russel, S.J., Peng, K.W., 2004. High cd46 receptor density determines preferential killing of tumor cells by oncolytic measles virus. Cancer Res. 64(14), 4919–4926.
Bajzer, Ž., Marušić, M., Vuk-Pavlović, S., 1996. Conceptual frameworks for mathematical modeling of tumor growth dynamics. Math. Comput. Model. 23, 31–46.
Bajzer, Ž., Vuk-Pavlović, S., Huzak, M., 1997. Mathematical modeling of tumor growth kinetics. In: Adam, J., Bellomo, N. (Eds.), A Survey of Models for Tumor-immune System Dynamics, pp. 89–133. Birkhäuser, New York. Chapter 3.
Bajzer, Ž., Carr, T., Josic, K., Russel, S.J., Dingli, D., 2008. Modelling of cancer virotherapy with recombinant measles virus. J. Theor. Biol. 252, 109–122.
Byrne, H.M., 2003. Modelling avascular tumor growth. In: Preziosi, L. (Ed.), Cancer Modelling and Simulation, pp. 75–120. CRC Press, Boca Raton. Chapter 4.
Cappuccio, A., Castiglione, F., Piccoli, B., 2007. Determination of the optimal therapeutic protocol in cancer immunotherapy. Math. Biosci. 209, 1–13.
Castiglione, F., Piccoli, B., 2007. Cancer immunotherapy, mathematical modelling, and optimal control. J. Theor. Biol. 247, 713–732.
de Pillis, L.G., Radunskaya, A.E., 2003. The dynamics of an optimally controlled tumor model: A case study. Math. Comput. Model. 37(11), 1221–1244.
Dingli, D., Peng, K.W., Harvey, M.E., Greipp, P.R., O’Connor, M.K., Cattaneo, R., Morris, J.C., Russell, S.J., 2004. Image-guided radiovirotherapy for multiple myeloma using a recombinant measles virus expressing the thyroidal sodium iodide symporter. Blood 103, 1641–1646.
Dingli, D., Cascino, M.D., Josić, K., Russell, S.J., Bajzer, Ž., 2006. Mathematical modeling of cancer radiovirotherapy. Math. Biosci. 199(1), 55–78.
Dingli, D., Pacheco, J.M., Dispenzieri, A., Hayman, S.R., Kumar, S.K., Lacy, M.Q., Gastineau, D.A., Gertz, M.A., 2007. Serum m-spike and transplant outcome in patients with multiple myeloma. Cancer Sci. 98(7), 1035–1040.
Fister, K.R., Panetta, J.C., 2000. Optimally control applied to cell-cycle specific cancer chemotherapy. SIAM J. Appl. Math. 60, 1059–1072.
Friedman, A., Tian, J., Fulci, G., Chiocca, E., Wang, J., 2006. Glioma virotherapy: Effects of innate immune supression and increased viral replication capacity. Cancer Res. 66, 2314–2319.
Galanis, E., Carlson, S.K., Foster, N.R., Lowe, V., Quevedo, F., McWilliams, R.R., Grothey, A., Jatoi, A., Alberts, S.R., Rubin, J., 2008. Phase 1 trial of a pathotropic retroviral vector expressing a cytocidal cyclin g1 construct (rexin-g) in patients with advanced pancreatic cancer. Mol. Ther. 16(5), 979–984.
Grote, D., Russell, S.J., Cornu, T.I., Cattaneo, R., Vile, R., Poland, G.A., Fielding, A.K., 2001. Live attenuated measles virus induces regression of human lymphoma xenografts in immunodeficient mice. Blood 97(12), 3746–3754.
Hirasawa, K., Nishikawa, S.G., Normal, K.L., Coffey, M.C., Thompson, B.G., Yoon, C.S., Waisman, D.M., Lee, P.W., 2003. Systemic reovirus therapy of metastaic cancer in immune-competent mice. Cancer Res. 63(2), 348–353.
Kirn, D., Martuza, R.L., Zwiebel, J., 2001. Replication-selective virotherapy for cancer: Biological principles, risk management and future directions. Nat. Med. 7(7), 781–877.
Liu, Y., Teo, K.L., Jennings, L.S., Wang, S., 1998. On a class of optimal control problems with state jumps. J. Optim. Theory Appl. 98(1), 65–82.
Martin, R., Teo, K.L., 1994. Optimal Drug Administration in Cancer Chemotherapy. Singapore.
Mukhopadhyay, B., Bhattacharyya, R., 2008. Analysis of a virus-immune system model with two distinct delays. Nonlinear Stud. 15(1), 37–50.
Myers, R., Greiner, S., Harvey, M., Soeffker, D., Frenzke, M., Abraham, K., Shaw, A., Rozenblatt, S., Federspiel, M.J., Russell, S.J., Peng, K.W., 2005. Oncolytic activities of approved mumps and measles vaccines for therapy of ovarian cancer. Cancer Gene Ther. 12(7), 593–599.
Nemunaitis, J., Khuri, F., Ganly, I., Arseneau, J., Posner, M., Vokes, E., Kuhn, J., McCarty, T., Landers, S., Blackburn, A., Romel, L., Randlev, B., Kaye, S., Kirn, D., 2001. Phase ii trial of intratumoral administration of onyx-015, a replication-selective adenovirus, in patients with refractory head and neck cancer. J. Clin. Oncol. 19(2), 289–298.
Ong, H.T., Timm, M.M., Greipp, P.R., Witzig, T.E., Dispenzieri, A., Russell, S.J., Peng, K.W., 2006. Oncolytic measles virus targets high cd46 expression on multiple myeloma cells. Exp. Hematol. 34(6), 713–720.
Paiva, L.T., Binny, C., Ferreira, S.C., Martins, M.L., 2009. A multiscale mathematical model for oncolytic virotherapy. Cancer Res. 69(3), 1205–1211.
Pecora, A.L., Rizvi, N., Cohen, G.I., Meropol, N.J., Sterman, D., Marshall, J.L., Goldberg, S., Gross, P., O’Neil, J.D., Groene, W.S., Roberts, M.S., Rabin, H., Bamat, M.K., Lorence, R.M., 2002. Phase i trial of intravenous administration of pv701, an oncolytic virus, in patients with advanced solid cancers. J. Clin. Oncol. 20(9), 2251–2266.
Peng, K.W., Ahmann, G.J., Pham, L., Greipp, P.R., Cattaneo, R., Russell, S.J., 2001. Systemic therapy of myeloma xenografts by an attenuated measles virus. Blood 98(7), 2002–2007.
Peng, K.W., Facteau, S., Wegman, T., O’Kane, D., Russell, S.J., 2002. Non-invasive in vivo monitoring of trackable viruses expressing soluble marker peptides. Nat. Med. 8(5), 527–531.
Peng, K.W., Hadac, E.M., Anderson, B.D., Myers, R., Harvey, M., Greiner, S.M., Soeffker, D., Federspiel, M.J., Russell, S.J., 2006. Pharmacokinetics of oncolytic measles virotherapy: Eventual equilibrium between virus and tumor in an ovarian cancer xenograft model 13(8), 732–738
Peng, K.W., TenEyck, C.J., Galanis, E., Kalli, K.R., Hartmann, L.C., Russell, S.J., 2002. Intraperitoneal therapy of ovarian cancer using an engineered measles virus. Cancer Res. 62(16), 4656–4662.
Phuong, L.K., Allen, C., Peng, K.W., Giannini, C., Greiner, S., TenEyck, C.J., Mishra, P.K., Macura, S.I., Russell, S.J., Galanis, E.C., 2003. Use of a vaccine strain of measles virus genetically engineered to produce carcinoembryonic antigen as a novel therapeutic agent against glioblastoma multiforme. Cancer Res. 63(10), 2462–2469.
Pratt, G., Goodyear, O., Moss, P., 2007. Immunodeficiency and immunotherapy in multiple myeloma. Br. J. Haematol. 138(5), 563–579.
Reid, T., Galanis, E., Abbruzzese, J., Sze, D., Andrews, J., Hatfield, M., Romel, L., Rubin, J., Kirn, D., 2001. Intra-arterial administration of a replication-selective adenovirus (dl1520) in patients with colorectal carcinoma metastatic to the liver: A phase i trial. Gene Ther. 8(21), 1618–1626.
Reid, T., Galanis, E., Abbruzzese, J., Sze, D., Wein, L.M., Andrews, J., Randlev, B., Heise, C., Uprichard, M., Hatfield, M., Rome, L., Rubin, J., Kirn, D., 2002. Hepatic arterial infusion of a replication-selective oncolytic adenovirus (dl1520): phase ii viral, immunologic, and clinical endpoints. Cancer Res. 62(21), 6070–6079.
Russell, S.J., 2002. RNA viruses as virotherapy agents. Cancer Gene Ther. 9(12), 961–966.
Schneider, U., von Messling, V., Devaux, P., Cattaneo, R., 2002. Efficiency of measles virus entry and dissemination through different receptors. J. Virol. 76(15), 7460–7467.
Spratt, J.A., von Fournier, D., Spratt, J.S., Weber, E.E., 1993. Decelerating growth and human breast cancer. Cancer 71(6), 2013–2019.
Tao, Y., Guo, Q., 2005. The competitive dynamics between tumor cells, a replication-competent virus and an immune response. J. Math. Biol. 51(1), 37–74.
Todo, T., Rabkin, S.D., Sundaresan, P., Wu, A., Meehan, K.R., Herscowitz, H.B., Martuza, R.L., 1999. Systemic antitumor immunity in experimental brain tumor therapy using a multimutated, replication-competent herpes simplex virus. Hum. Gene Ther. 10(17), 2741–2755.
Venkataraman, P., 2001. Applied Optimization with Matlab Programming. Wiley Interscience, New York.
Walter, W., 1998. Ordinary Differential Equations. Springer, New York.
Wodarz, D., 2001. Viruses as antitumor weapons: Defining conditions for tumor remission. Cancer Res. 61, 3501–3507.
Wodarz, D., 2003. Gene therapy for killing p53-negative cancer cells: use of replicating versus nonreplicating agents. Hum. Gene Ther. 14(2), 153–159.
Wu, J.T., Byrne, H.M., Kirn, D.H., Wein, L.M., 2001. Modeling and analysis of a virus that replicates selectively in tumor cells. Bull. Math. Biol. 63(4), 731–768.
Wu, J.T., Kirn, D.H., Wein, L.M., 2004. Analysis of a three-way race between tumor growth, a replication-competent virus and an immune response. Bull. Math. Biol. 66(4), 605–625.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Biesecker, M., Kimn, JH., Lu, H. et al. Optimization of Virotherapy for Cancer. Bull. Math. Biol. 72, 469–489 (2010). https://doi.org/10.1007/s11538-009-9456-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-009-9456-0