Abstract
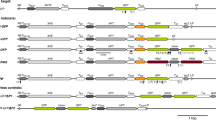
Post-transcriptional gene silencing (PTGS) greatly inhibits the use of transgenic plants in the agricultural and biotechnology industries. One cause of PTGS is transcription activated by nearby promoters that produces RNA transcripts that are complementary to the transgene. Therefore, strong termination elements flanking the transgene could potentially eliminate this type of silencing. Three potential sequences were identified in Arabidopsis thaliana between actively expressed plant genes with reading frames in a tail-to-tail orientation. We predicted that these elements have strong termination activity because these genes are expressed in all plant organs and structures and during all growth stages. In this study, we investigated the effects of these Arabidopsis termination elements (ATs) on the expression of transgenes encoding gusA and gfp and controlled by promoters of plant or viral origin. The presence of ATI and ATI-IV in the upstream region increased GFP expression from the constitutive promoters CaMV 35S and RuBisCo almost two-fold, and the construct flanked by ATI, ATI-IV and ATII enhanced RuBisCo-GFP expression approximately 2.3-fold. These ATs, however, had no effect on GUS expression when combined with a weak minimal 35S promoter and had very little effect on expression when combined with a shortened ELIP promoter. Finally, we also found that for strong promoters, especially in the case of the plant RuBisCo promoter, most of the variance component arises from differences between separate lines, but for weak promoters, approximately 60 % could be attributed to differences among the clones.



Similar content being viewed by others
Abbreviations
- TGS:
-
Transcriptional gene silencing
- PTGS:
-
Post-transcriptional gene silencing
- AT:
-
Arabidopsis Termination element
- CaMV:
-
Cauliflower mosaic virus
- RuBisCo:
-
Ribulose-1,5 bisphosphate carboxylase small subunit
- MAR:
-
Matrix attachment region
- GUS:
-
Beta glucuronidase
- GFP:
-
Green fluorescent protein II
- gusA :
-
Gene encoding beta glucuronidase
- gfp :
-
Gene encoding green fluorescent protein
- CS:
-
Cleavage site
- NUE:
-
Near upstream element
- FUE:
-
Far upstream element
References
Baulcombe D (2004) RNA silencing in plants. Nature 431:356–363
Bennet J (1993) Genes for crop improvement. In: Setlow JK (ed) Genetic engineering, vol 15. Plenum, New York, pp 165–189
Bhat SR, Srinivasan S (2002) Molecular and genetic analyses of transgenic plants: considerations and approaches. Plant Sci 164:673–681
Bhattacharyya MK, Stermer BA, Dixon RA (1994) Reduced variation in transgene expression from a binary vector with selectable markers at the right and the left T-DNA borders. Plant J 6:957–968
Birch RG (1997) Plant transformation: problems and strategies for practical application. Annu Rev Plant Physiol Plant Mol Biol 48:297–326
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Bruno AK, Wetzel CM (2004) The early light-inducible protein (ELIP) gene is expressed during the chloroplast-to-chromoplast transition in ripening tomato fruit. J Exp Bot 55(408):2541–2548
Butaye K, Goderis I, Wouters P, Pues J, Delauré S, Broekaert W, Depicker A, Cammue B, De Bolle M (2004) Stable high-level transgene expression in Arabidopsis thaliana using gene silencing mutants and matrix attachment regions. Plant J 39:440–449
Clark MF, Adams AN (1977) Characteristics of micro plate method of enzyme linked immunosorbent assay for detection of plant viruses. J Gen Virol 34:475–483
Dolgova A, Zaripova D, Dolgov S, Georgiev P, Maksimenko O (2009) Artificial regulatory elements which enables stable expression of a foreign gene in a tobacco plants. In: Gustafson P (ed) Proceedings of the 9th international plant molecular biology congress: Saint Louis, Missouri USA, October 25–30. DEStech Publications, Lancaster, p 190
Engvall E, Pediman P (1971) Enzyme linked immunosorbent assay (ELISA) quantitative assay of immunoglobin G. Immunochemistry 8:871–874
Gleave AP (1992) A versatile binary vector system with a T-DNA organisational structure conducive to efficient integration of cloned DNA into the plant genome. Plant Mol Biol 20(6):1203–1207
Haseloff J, Siemering KR, Prasher DC, Hodge S (1997) Removal of a cryptic intron and subcellular localization of green fluorescent protein are required to mark transgenic Arabidopsis plants brightly. Proc Natl Acad Sci USA 94:2122–2127
Hobbs SAL, Warkentin TM, DeLong CMO (1993) Transgene copy number can be positively or negatively associated with transgene expression. Plant Mol Biol 21:17–26
Horsch RB, Fry JE, Hoffmann NL, Eichholtz D, Rogers SG, Fraley RT (1985) A simple and general method for transferring genes into plants. Science 227:1229–1231
Hu J, Lutz CS, Wilusz J, Tian B (2005) Bioinformatic identification of candidate cis-regulatory elements involved in human mRNA polyadenylation. RNA 11(10):1485–1493
Jefferson RA (1987) Assaying chimeric genes in plants. The GUS gene fusion system. Plant Mol Biol Rep 5:387–405
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusion b-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 6:3901–3907
Kooter JM, Matzke MA, Meyer P (1999) Listening to the silent genes: transgene silencing, gene regulation and pathogen control. Trends Plant Sci 4:340–347
Lanfranco L (2003) Engineering crops, a deserving venture. Riv Biol 96:31–54
Li QQ, Hunt AG (1997) The polyadenylation of RNA in plants. Plant Physiol 115:321–325
Liechtenstein C, Draper J (1985) Genetic engineering of plants. In: Glover DM (ed) DNA cloning, vol 2. IRL Press, Oxford, pp 67–120
Linn F, Heidmann I, Saedler H, Meyer P (1990) Epigenetic changes in the expression of the maize A1 gene in petunia hybrida: role of numbers of integrated gene copies and state of methylation. Mol Gen Genet 222:329–336
Loke JC, Stahlberg EA, Strenski DG, Haas BJ, Wood PC, Li QQ (2005) Compilation of mRNA polyadenylation signals in Arabidopsis revealed a new signal element and potential secondary structures. Plant Physiol 138:1457–1468
Matzke MA, Matzke AJM (1990) Gene interactions and epigenetic variation in transgenic plants. Genet Devel 11:214–223
Matzke AJ, Matzke MA (1998) Position effects and epigenetic silencing of plant transgenes. Curr Opin Plant Biol 1:142–148
Matzke MA, Aufsatz W, Kanno T, Mette MF, Matzke AJ (2002) Homology dependent gene silencing and host defense in plants. Adv Genet 46:235–275
Ovchinnikova EV, Pushin AS, Dolgov SV (2008) Modifications of ribulose bisphosphate carboxylase small subunit promoter (rbcs 3a) for transgenic plants expression system. International conference—the biology of plant cells in vitro and biotechnology: pp 286–287
Peach C, Velten J (1991) Transgene expression variability (position effect) of CAT and GUS reporter genes driven by linked divergent T-DNA promoters. Plant Mol Biol 17:49–60
Stam M, Mol JNM, Kooter JM (1997) The Silence of Genes in Transgenic Plants. Ann Bot 79:3–12
Torney F, Partier A, Barret P, Beckert M (2005). Enhancement of maize transformation efficiency by the use of maize matrix attachment regions. Plant Cell Tissue Organ Cult. 80: 295-303 CrossRef, ISI
Vaucheret H, Fagard M (2001) Transcriptional gene silencing in plants: targets, inducers and regulators. Trends Genet 17(1):29–35
Wassenegger M (2002) Gene silencing. Int Rev Cytol 219:61–113
Zhang J, Lu L, Ji L, Yang G, Zheng C (2009) Functional characterization of a tobacco matrix attachment region-mediated enhancement of transgene expression. Transgenic Res 18(3):377–385. doi:10.1007/s11248-008-9230-3.Epub
Zhao J, Hyman L, Moore C (1999) Formation of mRNA 3′ ends in eukaryotes: mechanism, regulation, and interrelationships with other steps in mRNA synthesis. Microbiol Mol Biol Rev 63(2):405–445
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dolgova, A.S., Dolgov, S.V., Nazipova, N.N. et al. Arabidopsis termination elements increase transgene expression in tobacco plants. Plant Cell Tiss Organ Cult 120, 1107–1116 (2015). https://doi.org/10.1007/s11240-014-0667-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-014-0667-1