Abstract
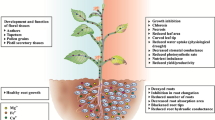
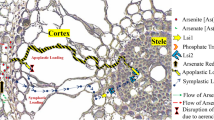
Present study is the first to explore physiological, biochemical and molecular changes in the medicinal plant Artemisia annua under arsenic (As) stress. A. annua grown hydroponically in a nutrient solution was spiked with increasing doses of As (0, 1,500, 3,000 and 4,500 μg l−1) for 7 days. Plants accumulated As in a dose dependent manner with bioconcentration factor 13.4 and translocation factor 0.97. While a similar trend of As accumulation was observed under soil culture experiments, the transfer factor went up to 2.1, depicting high efficiency of As translocation from roots to shoots by A. annua. Plants raised in 0–3,000 μg l−1 As containing nutrient solution registered increase in root length, biomass, and carotenoid contents without any visual toxicity symptoms. A dose dependent increase in the activities of enzymes such as superoxide dismutase, ascorbate peroxidase, glutathione reductase and guaiacol peroxidase followed by a gradual decline at higher concentrations suggested their role in alleviating oxidative stress. Significant increase in the levels of thiols, GSH, and pcs gene transcript up to 3,000 μg l−1 As attested their roles in As detoxification. Enhanced artemisinin production (an antimalarial compound) under As stress and upregulation of the transcripts (measured by RT-PCR) of the genes HMGR, FDS, ADS, and CYP71AV1 involved in artemisinin biosynthesis reaffirmed induction of artemisinin biosynthesis in A. annua under As stress. The results of the present study vividly suggested that A. annua has considerable As tolerance, and thus can be successfully cultivated in As contaminated fields.







Similar content being viewed by others
References
Abdin MZ, Israr M, Rehman RU, Jain SK (2003) Artemisinin, a novel antimalarial drug: biochemical and molecular approaches for enhanced production. Planta Med 69:1–11. doi:10.1055/s-2003-38871
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126. doi:10.1016/S0076-6879(84)05016-3
Alscher RG, Erturk N, Heath LS (2002) Role of superoxide dismutases (SODs) in controlling oxidative stress in plants. J Exp Bot 53:1331–1341. doi:10.1093/jexbot/53.372.1331
Anderson ME (1985) Determination of glutathione and glutathione disulphide in biological samples. Methods Enzymol 113:548–555. doi:10.1016/S0076-6879(85)13073-9
Arnon DI (1949) Copper enzymes in isolated chloroplast: polyphenol oxidase in beet (Beta vulgaris). Plant Physiol 24:1–15. doi:10.1104/pp.24.1.1
Arsenault PR, Wobbe KK, Weathers PJ (2008) Recent advances in artemisinin production through heterologous expression. Curr Med Chem 15(27):2886–2896. doi:10.2174/092986708786242813
Bhargava P, Srivastava AK, Urmil S, Rai LC (2005) Phytochelatin plays a role in UV-B tolerance in N2-fixing cyanobacterium Anabaena doliolum. J Plant Physiol 162:1220–1225. doi:10.1016/j.jplph.2004.12.006
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Ann Biochem 44:276–287. doi:10.1016/0003-2697(71)90370-8
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. doi:10.1006/abio.1976.9999
Cakmak T (2000) Possible roles of zinc in protecting plant cells from damage by reactive oxygen species. New Phytol 146:185–205. doi:10.1046/j.1469-8137.2000.00630.x
Cao X, Ma LQ, Tu C (2004) Antioxidant responses to arsenic in the arsenic hyperaccumulator Chinese brake fern (Pteris vittata L.). Environ Pollut 128:317–325. doi:10.1016/j.envpol.2003.09.018
Carbonell-Barrachina A, Burlo Carbonell F, Beneyto JM (1995) Arsenic uptake, distribution, and accumulation in tomato plants: effect of arsenite on plant growth and yield. J Plant Nutr 18:1237–1250
Carbonell-Barrachina AA, Aarabi MA, Delaune RD, Gambrell RP, Patrick JWH (1998) Arsenic in wetland vegetation: availability, phytotoxicity, uptake and effects on plant growth and nutrition. Sci Total Environ 217:189–199. doi:10.1016/S0048-9697(98)00195-8
Cenkci S, Cigerci IH, Yıldız M, Ozay C, Bozdag A, Terzi H (2010) Lead contamination reduces chlorophyll biosynthesis and genomic template stability in field mustard (Brassica rapa) L. Environ Exp Bot 67:467–473. doi:10.1016/j.envexpbot.2009.10.001
Chen W, Chang AC, Wu L (2007) Assessing long term environmental risks of trace elements in phosphate fertilizers. Ecotoxicol Environ Saf 67:48–58. doi:10.1016/j.ecoenv.2006.12.013
Cobbet CS, Goldsbrough PB (2002) Phytochelatins and metallothionenins: roles in heavy metal detoxification and homeostasis. Annu Rev Plant Biol 53:159–182. doi:10.1146/annurev.arplant.53.100301.135154
Diwan H, Ahmad A, Iqbal M (2007) Genotypic variation in the phytoremediation potential of Indian mustard for chromium. Environ Manag 41:734–741. doi:10.1007/s00267-007-9020-3
Dutta A, Batra J, Pandey Rai S, Singh D, Kumar S, Sen J (2005) Expression of terpenoid indole alkaloid biosynthetic pathway genes corresponds to accumulation of related alkaloids in Catharanthus roseus (L.) G. Don. Planta 220:376–383. doi:10.1007/s00425-004-1380-9
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82:70–77. doi:10.1016/0003-9861(59)90090-6
Gao Y, Mucci A (2001) Acid base reactions, phosphate and arsenate complexation, and their competitive adsorption at the surface of goethite in 0.7 M NaCl solution. Geochim Cosmochim Acta 65:2361–2378. doi:10.1016/S0016-7037(01)00589-0
Ghosh S, Singh P (2005) Comparative uptake and phytoextraction study of soil induced chromium by accumulator and high biomass weed species. Appl Ecol Environ Res 3:67–79. doi:10.1016/j.envpol.2004.05.015
Greger M (1999) Metal availability and bioconcentration in plants. In: Prasad MNV, Hagemeyer J (eds) Heavy metal stress in plants (from molecules to ecosystems). Springer, Berlin, p 127
Grill E, Winnaker El, Zenk MH (1985) Phytochelatins: the principal heavy metal complexing peptides of higher plants. Science 230:674–676. doi:10.1126/science.230.4726.674
Guo C, Liu CZ, Ye HC, Li GF (2004) Effect of temperature on growth and artemisinin biosynthesis in hairy root cultures of Artemisia annua. Acta Bot Boreal Occident Sin 24:1828–1831
Gupta DK, Tripathi RD, Mishra S, Srivastava S, Dwivedi S, Rai UN, Yang XE, Huanj H, Inouhe M (2008) Arsenic accumulation in root and shoot vis-a-vis its effects on growth and level of phytochelatins in seedlings of Cicer arietinum L. J Environ Biol 29:281–286. doi:10.1016/j.chemosphere.2007.07.038
Gupta M, Sharma P, Sarin NB, Sinha AK (2009) Differential response of arsenic stress in two varieties of Brassica juncea L. Chemosphere 74:1201–1208. doi:10.1016/j.chemosphere.2008.11.023
Hartley-Whitaker J, Woods C, Meharg AA (2001) Is differential phytochelatin production related to decreased arsenate influx in arsenate tolerant Holcus lanatus? New Phytol 155:219–225
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts I: kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198. doi:10.1016/0003-9861(68)90654-1
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. Calif Agric Exp Stn Circ 347:1–3
Jain M, Gadre R (1997) Effect of As on chlorophyll and protein contents and enzymic activities in greening maize tissues. Water Air Soil Pollut 93:109–115. doi:10.1007/BF02404750
Kato M, Shimizu S (1987) Chlorophyll metabolism in higher plants VII. Chlorophyll degradation in senescing tobacco leaves: phenolic dependent peroxidative degradation. Can J Bot 65:729–735. doi:10.1139/b87-097
Khan I, Ahmad A, Iqbal M (2009) Modulation of antioxidant defense system for arsenic detoxification in Indian mustard. Ecotoxicol Environ Saf 72:626–634. doi:10.1016/j.ecoenv.2007.11.016
Knauer K, Behra R, Hemond H (1999) Toxicity of inorganic and methylated arsenic to algal communities from lakes along an arsenic contamination gradient. Aquat Toxicol 46:21–230. doi:10.1016/S0166-445X(98)00131-3
Liu CZ, Guo C, Wang YC, Ouyang F (2002) Effect of light irradiation on hairy root growth and artemisinin biosynthesis of Artemisia annua L. Process Biochem 38:581–585. doi:10.1016/S0032-9592(02)00165-6
Luo XD, Shen CC (1987) The chemistry, pharmacology and clinical applications of Qinghaosu (artemisinin) and its derivatives. Med Res Rev 7:29–52. doi:10.1002/med.2610070103
Mascher R, Lippmann B, Holzinger S, Bergmann H (2002) Arsenate toxicity: effects on oxidative stress response molecules and enzymes in red clover plants. Plant Sci 163:961–969. doi:10.1016/S0168-9452(02)00245-5
Mattina MJI, Lannucci-Berger W, Musante C, White JC (2003) Concurrent plant uptake of heavy metals and persistent organic pollutants from soil. Environ Pollut 124:375–378. doi:10.1016/S0269-7491(03)00060-5
Meharg AA (2004) Arsenic in rice-understanding a new disaster for South-East Asia. Trends Plant Sci 9:415–417. doi:10.1016/j.tplants.2004.07.002
Middleton EM, Teramura AH (1993) The role of flavonol glycosides and carotenoids in protecting soybean from UV-B damage. Plant Physiol 103:741–752. doi:10.1104/pp.103.3.741
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880
Noctor G, Foyer CH (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Physiol Plant Mol Biol 49:249–279. doi:10.1146/annurev.arplant.49.1.249
Odanaka Y, Tsuchiya N, Matano O, Goto S (1987) Absorption, translocation and metabolism of the arsenical fungicides, iron methanearsonate and ammonium iron methane arsonate, in rice plants. J Pestic Sci 12:199–208
Paivoke AEA, Simola LK (2001) Aresenate toxicity to Pisum sativum. Mineral nutrients, chlorophyll content, and phytase activity. Ecotoxicol Environ Saf 49:111–121. doi:10.1006/eesa.2001.2044
Palma JM, Sandalio LM, Corpas FJ, Romero-Puertas MC, McCarthy I, DelRío LA (2002) Plant proteases, protein degradation, and oxidative stress: role of peroxisomes. Plant Physiol Biochem 40:521–530. doi:10.1016/S0981-9428(02)01404-3
Pickering IJ, Price RC, George MJ, Smith RD, George GN, Salt DE (2000) Reduction and coordination of arsenic in Indian mustard. Plant Physiol 122:1171–1177. doi:10.1104/pp.122.4.1171
Pu GB, Ma DM, Chen JL, Ma LQ, Wang H, Li GF, Ye HC, Liu BY (2009) Salicylic acid activates artemisinin biosynthesis in Artemisia annua L. Plant Cell Rep 28:1127–1135. doi:10.1007/s00299-009-0713-3
Qian ZH, Gong K, Zhang L, Lv Jb, Jing FY, Wang YY, Guan SB, Wang GF, Tang KK (2007) A simple and efficient procedure to enhance artemisinin content in Artemisia annua L. by seeding to salinity stress. Afr J Biotechnol 6:1410–1413
Qureshi MI, Israr M, Abdin MZ, Iqbal M (2005) Responses of Artemisia annua L. to lead and salt-induced oxidative stress. Environ Exp Bot 53:185–193. doi:10.1016/j.envexpbot.2004.03.014
Roychowdhury T, Uchino T, Toluanga H, Ando M (2002) Survey of arsenic in food composites from arsenic affected area of West Bengal, India. Food Chem Toxicol 40:1611–1621. doi:10.1016/S0278-6915(02)00104-7
Schaedle M, Bassham JA (1977) Chloroplasts glutathione reductase. Plant Physiol 59:1011–1012. doi:10.1104/pp.59.5.1011
Schat H, Llugany M, Vooijs R, Hartley-Whitaker J, Bleeker PM (2002) The role of phytochelatins in constitutive and adaptive heavy metal tolerances in hyperaccumulator and non-hyperaccumulator metallophytes. J Exp Bot 53:2381–2392. doi:10.1093/jxb/erf107
Schmoger MEV, Oven M, Grill E (2000) Detoxification of arsenic by phytochelatins in plants. Plant Physiol 122:793–801. doi:10.1104/pp.122.3.793
Shaibur MR, Kawai S (2009) Effect of arsenic on visible symptom and arsenic concentration in hydroponic mustard spinach. Environ Exp Bot 67:65–70. doi:10.1016/j.envexpbot.2009.06.001
Shri M, Kumar S, Chakrabarty D, Trivedi PK, Mallick S, Misra P, Shukla D, Mishra S, Srivastava S, Tripathi RD, Tuli R (2009) Effect of arsenic on growth, oxidative stress, and antioxidant system in rice seedlings. Ecotoxicol Environ Saf 72:1102–1110. doi:10.1016/j.ecoenv.2008.09.022
Singh N, Ma LQ (2006) Arsenic speciation and arsenic and phosphate distribution in arsenic hyperaccumulator Pteris vittata and nonhyperaccumulator Pteris ensiformis. Environ Pollut 141:238–246. doi:10.1016/j.envpol.2005.08.050
Singh HP, Batish DR, Kohli RK, Arora K (2007) Arsenic-induced root growth inhibition in mung bean (Phaseolus aureus Roxb.) is due to oxidative stress resulting from enhanced lipid peroxidation. Plant Growth Regul 53:65–73. doi:10.1007/s10725-007-9205-z
Srivastava M, Ma LQ, Singh N, Singh S (2005) Antioxidant responses of hyperaccumulator and sensitive fern species to arsenic. J Exp Bot 56:1335–1342. doi:10.1093/jxb/eri134
Srivastava S, Mishra S, Tripathi RD, Dwivedi S, Trivedi PK, Tandon PK (2007) Phytochelatins and antioxidant systems respond differentially during arsenite and arsenate stress in Hydrilla verticilliata (L.f.). Royle Environ Sci Technol 41:2930–2936. doi:10.1021/es062167j
Sun Q, Ye ZH, Wang XR, Wong MH (2007) Cadmium hyperaccumulation leads to an increase of glutathione rather than phytochelatins in the cadmium hyperaccumulator Sedum alfredii. J Plant Physiol 164:1489–1498. doi:10.1016/j.jplph.2006.10.001
Tang T, Miller DM (1991) Growth and tissue composition of rice grown in soil treated with inorganic copper, nickel, and arsenic. Commun Soil Sci Plant Anal 22:2037–2045. doi:10.1080/00103629109368556
Trease GE, Evans GE (1989) Text book of pharmacogonosy, 2nd edn. Bailliera Tindall, London
Tripathi RD, Srivastava S, Mishra S, Singh N, Tuli R, Gupta DK, Maathuis FJM (2007) Arsenic hazards: strategies for tolerance and remediation by plants. Trends Biotechnol 25:158–165. doi:10.1016/j.tibtech.2007.02.003
Tu C, Ma LQ (2002) Effects of arsenic concentrations and forms on arsenic uptake by the hyperaccumulator ladder brake. J Environ Qual 31:641–647. doi:10.2134/jeq2002.0641
Vatamunik OK, Mari S, Lu YP, Rea PA (2000) Mechanism of heavy metal ion activation of phytochelatin (PC) synthase—blocked thiols are sufficient for PC synthase-catalyzed transpeptidation of glutathione and related thiol peptides. J Biol Chem 275:31451–31459. doi:10.1074/jbc.M002997200
Wallaart TE, Van Uden W, Lubberink HG, Woerdenbag HJ, Pras N, Quax WJ (1999) Isolation and identification of dihydroartemisinic acid from Artemisia annua and its possible role in the biosynthesis of artemisinin. J Nat Prod 62:430–433. doi:10.1021/np980370p
Wallaart TE, Pras N, Beekman AC, Quax WJ (2000) Seasonal variation of artemisinin and its biosynthetic precursors in plants of Artemisia annua of different geographical origin: proof for the existence of chemotypes. Planta Med 66:57–62. doi:10.1055/s-2000-11115
Wang YC, Zhang HX, Zhao B, Yaun XF (2001) Improved growth of Artemisia annua L. hairy roots and artemisinin production under red light conditions. Biotechnol Lett 23:1971–1973
WHO (2006) Monograph on good agricultural and collection practices (GACP) for Artemisia annua L. WHO, Geneva
Wingate VPM, Lawton MA, Lamb CJ (1988) Glutathione causes a massive and selective induction of plant defense genes. Plant Physiol 87:206–210. doi:10.1104/pp.87.1.206
Zhao SS, Zeng M (1985) Spectrometric high pressure liquid chromatography (HPLC) studies on the analysis of Qinghaosu. Planta Med 3:233–237. doi:10.1055/s-2007-969466
Acknowledgments
The authors thank the Department of Science and Technology, New Delhi, India for financial assistance in the form of a project. Rashmi Rai and Sarita Pandey are thankful to CSIR for Senior Research Fellowships. We thank the Head, CAS in Botany, Banaras Hindu University for facilities. Prof. L. C. Rai FNA, Department of Botany, B. H. U., is gratefully acknowledged for his critical reading of the manuscript and valuable suggestions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rai, R., Pandey, S. & Rai, S.P. Arsenic-induced changes in morphological, physiological, and biochemical attributes and artemisinin biosynthesis in Artemisia annua, an antimalarial plant. Ecotoxicology 20, 1900–1913 (2011). https://doi.org/10.1007/s10646-011-0728-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-011-0728-8