Abstract
Background
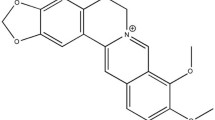
Berberine exhibits numerous pharmacological effects, but the mechanism for its protective effects against ischemia-reperfusion cardiac injury is unknown.
Methods
Male Wistar rats were treated with berberine (100 mg/Kg/day, ig) for 14 days and controls treated with water. Hearts were isolated in vitro and perfused in the Langendorff mode and subjected to 30 min of global ischemia followed by 30 min of reperfusion and hemodynamic data examined. In a separate set of experiments, hearts were subjected in vivo to left anterior descending coronary artery ligation for 30 min followed by 120 min reperfusion and hemodynamic data, type and duration of arrhythmias, and myocardial infarct size determined. AMP-activated protein kinase (AMPK) level, ADP/ATP and AMP/ATP ratios were examined in non-ischemic areas and risk areas of the heart.
Results
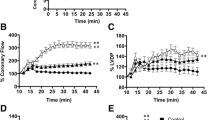
Subsequent to ischemia-reperfusion injury, left ventricular developed pressure, left ventricular end diastolic pressure and maximum rate of intraventricular pressure contractility and relaxation were significantly improved in the berberine treatment groups compared to controls. Berberine treatment decreased infarct size and diminished the duration and incidence of arrhythmias compared to controls. Berberine treatment significantly decreased AMPK protein concentration, and the ratio of ADP/ATP and AMP/ATP in the myocardial risk areas. In contrast, berberine treatment significantly increased AMPK protein concentration, and the ratio of ADP/ATP and AMP/ATP in the non-ischemia areas compared to controls.
Conclusion
These findings suggest that berberine may exert its cardioprotective effect on ischemia-reperfusion injury via regulation of AMPK activity in both non-ischemic areas and risk areas of the heart.







Similar content being viewed by others
Abbreviations
- AAR:
-
area at risk
- AICAR:
-
5-aminoimidazole-4-carboxamide -1-β-D-ribofuranoside
- AMP:
-
adenosine monophosphate
- ADP:
-
adenosine diphosphate
- AMPK:
-
adenosine-5′-monophosphate kinase
- AMPKK:
-
AMPK kinase
- ATP:
-
adenosine triphosphate
- BER:
-
berberine
- CamKK:
-
calmodulin-dependent protein kinase
- CDA:
-
coronary artery disease
- −dp/dtmin:
-
+dp/dtmax, minimum and maximum rate of pressure change in the ventricle
- ECG:
-
electrocardiogram
- GAPDH:
-
glyceraldehydes-3-phosphate dehydrogenates
- HR:
-
heart rate
- IS:
-
infract size
- KHB:
-
Krebs-Henseleit buffer
- LKB1:
-
tumor suppressor kinase
- LV:
-
left ventricular size
- LVDP:
-
left ventricular developed pressure
- LVEDP:
-
left ventricular end-diastolic pressure
- NIA:
-
non-ischemia area
- PBS:
-
phosphate buffered solution
- TTC:
-
triphenyl tetrazolium chloride phosphate buffer
- VAEs:
-
ventricular arrhythmic events
- VF:
-
ventricular fibrillation
- VT:
-
ventricular tachycardia.
References
Ferrari R. Importance of oxygen free radicals during ischemia and reperfusion in the experimental and clinical setting. Oxygen free radicals and the heart. Am J Cardiovasc Pathol. 1992;4(2):103–14.
Koch A, Bingold TM, Oberlander J, Sack FU, Otto HF, Hagl S, Schnabel PA. Capillary endothelia and cardiomyocytes differ in vulnerability to ischemia/reperfusion during clinical heart transplantation. Eur J Cardiothorac Surg. 2001;20(5):996–1001.
Okabe E. Ito H: [Biochemistry of the physiopathologic and clinical aspects of free radicals in heart ischemia: free radicals as a mediator of ischemia-reperfusion injury]. Nihon Rinsho. 1988;46(10):2190–5.
Shibata T, Yamamoto F, Kosakai Y, Kawazoe K, Komai H, Ichikawa H, Ohashi T, Shimada Y, Nakajima N, Kawashima Y. Clinical application of recombinant human SOD for the protection of ischemia reperfusion injury during open heart surgery. Nihon Kyobu Geka Gakkai Zasshi. 1993;41(3):427–32.
Desai AB, Shah KM, Shah DM. Berberine in treatment of diarrhoea. Indian Pediatr. 1971;8(9):462–5.
Khin Maung U, Myo K, Nyunt Nyunt W, Aye K, Tin U. Clinical trial of berberine in acute watery diarrhoea. Br Med J (Clin Res Ed). 1985;291(6509):1601–5.
Lahiri SC, Dutta NK. Berberine and chloramphenicol in the treatment of cholera and severe diarrhoea. J Indian Med Assoc. 1967;48(1):1–11.
Bhamra GS, Hausenloy DJ, Davidson SM, Carr RD, Paiva M, Wynne AM, Mocanu MM, Yellon DM. Metformin protects the ischemic heart by the Akt-mediated inhibition of mitochondrial permeability transition pore opening. Basic Res Cardiol. 2008;103(3):274–84.
Bhutada P, Mundhada Y, Bansod K, Tawari S, Patil S, Dixit P, Umathe S, Mundhada D. Protection of cholinergic and antioxidant system contributes to the effect of berberine ameliorating memory dysfunction in rat model of streptozotocin-induced diabetes. Behav Brain Res. 2011;220(1):30–41.
Hsieh YS, Kuo WH, Lin TW, Chang HR, Lin TH, Chen PN, Chu SC. Protective effects of berberine against low-density lipoprotein (LDL) oxidation and oxidized LDL-induced cytotoxicity on endothelial cells. J Agric Food Chem. 2007;55(25):10437–45.
Meng S, Wang LS, Huang ZQ, Zhou Q, Sun YG, Cao JT, Li YG, Wang CQ. Berberine ameliorates inflammation in patients with acute coronary syndrome following percutaneous coronary intervention. Clin Exp Pharmacol Physiol. 2012;39(5):406–11.
Zhang H, Wei J, Xue R, Wu JD, Zhao W, Wang ZZ, Wang SK, Zhou ZX, Song DQ, Wang YM, et al. Berberine lowers blood glucose in type 2 diabetes mellitus patients through increasing insulin receptor expression. Metabolism. 2010;59(2):285–92.
Wang C, Li J, Lv X, Zhang M, Song Y, Chen L, Liu Y. Ameliorative effect of berberine on endothelial dysfunction in diabetic rats induced by high-fat diet and streptozotocin. Eur J Pharmacol. 2009;620(1–3):131–7.
Wang Q, Zhang M, Liang B, Shirwany N, Zhu Y, Zou MH. Activation of AMP-activated protein kinase is required for berberine-induced reduction of atherosclerosis in mice: the role of uncoupling protein 2. PLoS One. 2011;6(9):e25436.
Folmes KD, Chan AY, Koonen DP, Pulinilkunnil TC, Baczko I, Hunter BE, Thorn S, Allard MF, Roberts R, Gollob MH, et al. Distinct early signaling events resulting from the expression of the PRKAG2 R302Q mutant of AMPK contribute to increased myocardial glycogen. Circ Cardiovasc Genet. 2009;2(5):457–66.
Dyck JR, Lopaschuk GD. AMPK alterations in cardiac physiology and pathology: enemy or ally? J Physiol. 2006;574(Pt 1):95–112.
Xia X, Yan J, Shen Y, Tang K, Yin J, Zhang Y, Yang D, Liang H, Ye J, Weng J. Berberine improves glucose metabolism in diabetic rats by inhibition of hepatic gluconeogenesis. PLoS One. 2011;6(2):e16556.
Yin J, Gao Z, Liu D, Liu Z, Ye J. Berberine improves glucose metabolism through induction of glycolysis. Am J Physiol Endocrinol Metab. 2008;294(1):E148–56.
Turner N, Li JY, Gosby A, To SW, Cheng Z, Miyoshi H, Taketo MM, Cooney GJ, Kraegen EW, James DE, et al. Berberine and its more biologically available derivative, dihydroberberine, inhibit mitochondrial respiratory complex I: a mechanism for the action of berberine to activate AMP-activated protein kinase and improve insulin action. Diabetes. 2008;57(5):1414–8.
Hong Y, Hui SC, Chan TY, Hou JY. Effect of berberine on regression of pressure-overload induced cardiac hypertrophy in rats. Am J Chin Med. 2002;30(4):589–99.
Yang J, Zhou ZY, Xu JG. Protective effect of berberine on cardiac hypertrophy induced by L-thyroxine in rats. Sichuan Da Xue Xue Bao Yi Xue Ban. 2004;35(2):223–5.
Capano M, Crompton M. Bax translocates to mitochondria of heart cells during simulated ischaemia: involvement of AMP-activated and p38 mitogen-activated protein kinases. Biochem J. 2006;395(1):57–64.
Lopaschuk GD. AMP-activated protein kinase control of energy metabolism in the ischemic heart. Int J Obes (Lond). 2008;32 Suppl 4:S29–35.
Paiva MA, Goncalves LM, Providencia LA, Davidson SM, Yellon DM, Mocanu MM. Transitory activation of AMPK at reperfusion protects the ischaemic-reperfused rat myocardium against infarction. Cardiovasc Drugs Ther. 2010;24(1):25–32.
Paiva MA, Rutter-Locher Z, Goncalves LM, Providencia LA, Davidson SM, Yellon DM, Mocanu MM. Enhancing AMPK activation during ischemia protects the diabetic heart against reperfusion injury. Am J Physiol Heart Circ Physiol. 2011;300(6):H2123–34.
Sasaki H, Asanuma H, Fujita M, Takahama H, Wakeno M, Ito S, Ogai A, Asakura M, Kim J, Minamino T, et al. Metformin prevents progression of heart failure in dogs: role of AMP-activated protein kinase. Circulation. 2009;119(19):2568–77.
Gonzalez-Salazar A, Molina-Jijon E, Correa F, Zarco-Marquez G, Calderon-Oliver M, Tapia E, Zazueta C, Pedraza-Chaverri J. Curcumin protects from cardiac reperfusion damage by attenuation of oxidant stress and mitochondrial dysfunction. Cardiovasc Toxicol. 2011;11(4):357–64.
Thuc LC, Teshima Y, Takahashi N, Nishio S, Fukui A, Kume O, Ezaki K, Miyazaki H, Yufu K, Hara M, et al. Cardioprotective effects of pravastatin against lethal ventricular arrhythmias induced by reperfusion in the rat heart. Circ J. 2011;75(7):1601–8.
Hermann R, Marina Prendes MG, Torresin ME, Velez D, Savino EA, Varela A. Effects of the AMP-activated protein kinase inhibitor compound C on the postconditioned rat heart. J Physiol Sci. 2012;62(4):333–41.
Mochizuki T, Yu S, Katoh T, Aoki K, Sato S. Cardioprotective effect of therapeutic hypothermia at 34 degrees C against ischaemia/reperfusion injury mediated by PI3K and nitric oxide in a rat isolated heart model. Resuscitation. 2012;83(2):238–42.
Hong Y, Hui SS, Chan BT, Hou J. Effect of berberine on catecholamine levels in rats with experimental cardiac hypertrophy. Life Sci. 2003;72(22):2499–507.
Saddik M, Gamble J, Witters LA, Lopaschuk GD. Acetyl-CoA carboxylase regulation of fatty acid oxidation in the heart. J Biol Chem. 1993;268(34):25836–45.
Hopkins TA, Dyck JR, Lopaschuk GD. AMP-activated protein kinase regulation of fatty acid oxidation in the ischaemic heart. Biochem Soc Trans. 2003;31(Pt 1):207–12.
Dennis SC, Gevers W, Opie LH. Protons in ischemia: where do they come from; where do they go to? J Mol Cell Cardiol. 1991;23(9):1077–86.
Liu B, Clanachan AS, Schulz R, Lopaschuk GD. Cardiac efficiency is improved after ischemia by altering both the source and fate of protons. Circ Res. 1996;79:940–8.
Russell 3rd R. The Role of AMP-activated protein kinase in fuel selection by the stressed heart. Curr Hypertens Rep. 2003;5(6):459–65.
Russell 3rd RR, Li J, Coven DL, Pypaert M, Zechner C, Palmeri M, Giordano FJ, Mu J, Birnbaum MJ, Young LH. AMP-activated protein kinase mediates ischemic glucose uptake and prevents postischemic cardiac dysfunction, apoptosis, and injury. J Clin Invest. 2004;114(4):495–503.
Calvert JW, Gundewar S, Jha S, Greer JJ, Bestermann WH, Tian R, Lefer DJ. Acute metformin therapy confers cardioprotection against myocardial infarction via AMPK-eNOS-mediated signaling. Diabetes. 2008;57(3):696–705.
Gundewar S, Calvert JW, Jha S, Toedt-Pingel I, Ji SY, Nunez D, Ramachandran A, Anaya-Cisneros M, Tian R, Lefer DJ. Activation of AMP-activated protein kinase by metformin improves left ventricular function and survival in heart failure. Circ Res. 2009;104(3):403–11.
Folmes CD, Wagg CS, Shen M, Clanachan AS, Tian R, Lopaschuk GD. Suppression of 5′-AMP-activated protein kinase activity does not impair recovery of contractile function during reperfusion of ischemic hearts. Am J Physiol Heart Circ Physiol. 2009;297(1):H313–21.
Kudo N, Barr AJ, Barr RL, Desai S, Lopaschuk GD. High rates of fatty acid oxidation during reperfusion of ischemic hearts are associated with a decrease in malonyl-CoA levels due to an increase in 5′-AMP-activated protein kinase inhibition of acetyl-CoA carboxylase. J Biol Chem. 1995;270(29):17513–20.
Zhai P, Sciarretta S, Galeotti J, Volpe M, Sadoshima J. Differential roles of GSK-3beta during myocardial ischemia and ischemia/reperfusion. Circ Res. 2011;109(5):502–11.
Sakamoto K, Zarrinpashneh E, Budas GR, Pouleur AC, Dutta A, Prescott AR, Vanoverschelde JL, Ashworth A, Jovanovic A, Alessi DR, et al. Deficiency of LKB1 in heart prevents ischemia-mediated activation of AMPKalpha2 but not AMPKalpha1. Am J Physiol Endocrinol Metab. 2006;290(5):E780–8.
Anderson KA, Means RL, Huang QH, Kemp BE, Goldstein EG, Selbert MA, Edelman AM, Fremeau RT, Means AR. Components of a calmodulin-dependent protein kinase cascade. Molecular cloning, functional characterization and cellular localization of Ca2+/calmodulin-dependent protein kinase kinase beta. J Biol Chem. 1998;273(48):31880–9.
Hardie DG, Carling D. The AMP-activated protein kinase–fuel gauge of the mammalian cell? Eur J Biochem. 1997;246(2):259–73.
Xiao B, Sanders MJ, Underwood E, Heath R, Mayer FV, Carmena D, Jing C, Walker PA, Eccleston JF, Haire LF, et al. Structure of mammalian AMPK and its regulation by ADP. Nature. 2011;472(7342):230–3.
Hwang JT, Kwon DY, Park OJ, Kim MS. Resveratrol protects ROS-induced cell death by activating AMPK in H9c2 cardiac muscle cells. Genes Nutr. 2008;2(4):323–6.
Wiczer BM, Lobo S, Machen GL, Graves LM, Bernlohr DA. FATP1 mediates fatty acid-induced activation of AMPK in 3T3-L1 adipocytes. Biochem Biophys Res Commun. 2009;387(2):234–8.
Cicero AF, Rovati LC, Setnikar I. Eulipidemic effects of berberine administered alone or in combination with other natural cholesterol-lowering agents. A single-blind clinical investigation. Arzneimittelforschung. 2007;57(1):26–30.
Acknowledgments
This work was supported by Jilin Science & Technology Development Plan (20070728-2, 20090441), Jilin Province Administration of Traditional Chinese medicine science and technology projects No.2010-093, Opening Project of State Key Laboratory of Supramolecular Structure and Materials of Jilin University under Grant No. SKLSSM200912, and the Heart and Stroke Foundation of Manitoba. G.M.H. is a Canada Research Chair in Molecular Cardiolipin Metabolism.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Wenguang Chang and Ming Zhang contributed equally to this and was considered co-first author.
Rights and permissions
About this article
Cite this article
Chang, W., Zhang, M., Li, J. et al. Berberine Attenuates Ischemia-Reperfusion Injury Via Regulation of Adenosine-5′-monophosphate Kinase Activity in Both Non-ischemic and Ischemic Areas of the Rat Heart. Cardiovasc Drugs Ther 26, 467–478 (2012). https://doi.org/10.1007/s10557-012-6422-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-012-6422-0