Abstract
Purpose
To determine the actions of isoespintanol (Isoesp) on post-ischemic myocardial and mitochondrial alterations.
Methods
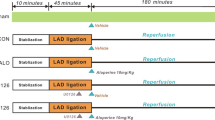
Hearts removed from Wistar rats were perfused by 20 min. After this period, the coronary flow was interrupted by half an hour and re-established during 1 h. In the treated group, Isoesp was administered at the beginning of reperfusion. To assess the participation of ε isoform of protein kinase C (PKCε), protein kinase B (PKB/Akt), and nitric oxide synthase (NOS), hearts were treated with Isoesp plus the respective inhibitors (chelerythrine, wortmannin, and N-nitro-l-arginine methyl ester). Cell death was determined by triphenyl tetrazolium chloride staining technique. Post-ischemic recovery of contractility, oxidative stress, and content of phosphorylated forms of PKCε, Akt, and eNOS were also examined. Mitochondrial state was assessed through the measurement of calcium-mediated response, calcium retention capacity, and mitochondrial potential.
Results
Isoesp limited cell death, decreased post-ischemic dysfunction and oxidative stress, improved mitochondrial state, and increased the expression of PKCε, Akt, and eNOS phosphorylated. All these beneficial effects achieved by Isoesp were annulled by the inhibitors.
Conclusion
These findings suggest that activation of Akt/eNOS and PKCε signaling pathways are involved in the development of Isoesp-induced cardiac and mitochondria tolerance to ischemia-reperfusion.








Similar content being viewed by others

Abbreviations
- Isoesp:
-
Isoespintanol
- NOS:
-
Nitric oxide synthase
- l-NAME:
-
l-NG-Nitroarginine methyl ester
- PKCε:
-
Protein kinase C ε
- Che:
-
Chelerythrine
- Akt:
-
Protein kinase B
- Wort:
-
Wortmannin
- DMSO:
-
Dimethyl sulfoxide
- CR:
-
Coronary resistance
- CPP:
-
Coronary perfusion pressure
- CF:
-
Coronary flow
- GSH:
-
Reduced glutathione
- TBARS:
-
Thiobarbituric acid reactive substances
- LSD:
-
Light scattering decrease
- CRC:
-
Ca2+ retention capacity
- ΔΨ:
-
Mitochondrial membrane potential
- mPTP:
-
Mitochondrial permeability transition pore
- TTC:
-
Triphenyltetrazolium chloride
- LVDP:
-
Left ventricular developed pressure
- +dP/dtmax :
-
Maximal velocity of rise of left ventricular pressure
- LVEDP:
-
Left ventricular end diastolic pressure
- GAPDH:
-
Glyceraldehyde 3-phosphate dehydrogenase
- ROS:
-
Radical oxygen species
References
Baines CP, Song CX, Zheng YT, Wang GW, Zhang J, Wang OL, Guo Y, Bolli R, Cardwell EM, Ping P (2003) Protein kinase Cepsilon interacts with and inhibits the permeability transition pore in cardiac mitochondria. Circ Res 92(8):873–880
Bice JS, Jones BR, Chamberlain GR, Baxter GF (2016) Nitric oxide treatments as adjuncts to reperfusion in acute myocardial infarction: a systematic review of experimental and clinical studies. Basic Res Cardiol 111:23–38
Boengler K, Lochnit G, Schulz R (2018) Mitochondria “THE” target of myocardial conditioning. Am J Physiol Heart Circ Physiol 315(5):H1215–H1231
Britto RM, Silva-Neto JAD, Mesquita TRR, Vasconcelos CML, de Almeida GKM, Jesus ICG, Santos PHD, Souza DS, Miguel-Dos-Santos R, de Sá LA, Dos Santos FSM, Pereira-Filho RN, Albuquerque-Júnior RLC, Quintans-Júnior LJ, Guatimosim S, Lauton-Santos S (2018) Myrtenol protects against myocardial ischemia-reperfusion injury through antioxidant and anti-apoptotic dependent mechanisms. Food Chem Toxicol 111:557–566
Chen Y, Ba L, Huang W, Liu Y, Pan H, Mingyao P, Shi P, Wang Y, Li S, Qi H, Sun H, Cao Y (2017) Role of carvacrol in cardioprotection against myocardial ischemia/reperfusion injury in rats through activation of MAPK/ERK and Akt/eNOS signaling pathways. Eur J Pharmacol 796:90–100
Chen C, Du P, Wang J (2015) Paeoniflorin ameliorates acute myocardial infarction of rats by inhibiting inflammation and inducible nitric oxide synthase signaling pathways. Mo Med Rep 12:3937–3943
Davidson SM, Ferdinandy P, Andreadou I, Bøtker HE, Heusch G, Ibáñez B, Ovize M, Schulz R, Yellon DM, Hausenloy DJ, Garcia-Dorado D (2019) Multitarget strategies to reduce myocardial ischemia/reperfusion injury: JACC review Topic of the Week. J Am Coll Cardiol 73(1):89–99
Fantinelli JC, Cuéllar Álvarez LN, González Arbeláez LF, Ciocci Pardo A, Galeano García PL, Schinella GR, Mosca SM (2017) Acute treatment with copoazú fermented extract ameliorates myocardial ischemia-reperfusion injury via eNOS activation. J Funct Foods 34:470–477
Gavilánez Buñay TC, Colareda GA, Ragone MI, Bonilla M, Rojano BA, Schinella GR, Consolini A (2018) Intestinal, urinary and uterine antispasmodic effects of isoespintanol, metabolite from Oxandra xylopioides leaves. Phytomedicine 51:20–28
González Arbeláez LF, Fantinelli JC, Ciocci Pardo A, Caldiz CI, Ríos JL, Schinella GR, Mosca SM (2016b) Effect of Ilex paraguariensis (yerba mate) extract on infarct size in isolated rat heart: mechanisms involved. Food Funct 7:816–824
González Arbeláez LF, Ciocci Pardo A, Fantinelli JC, Caldiz CI, Ríos JL, Schinella GR, Mosca SM (2016a) Ex vivo treatment with a polyphenol-enriched cocoa extract ameliorates myocardial infarct and postischemic mitochondrial injury in normotensive and hypertensive rats. J Agric Food Chem 64:5180–5187
González Arbeláez LF, Ciocci Pardo A, Fantinelli JC, Schinella GR, Mosca SM, Ríos JL (2018) Cardioprotection and natural polyphenols: an update of clinical and experimental studies. Food & Function 9(12):6129–6145
Heusch G (2015) Molecular basis of cardioprotection: signal transduction in ischemic pre-, post-, and remote conditioning. Circ Res 116(4):674–699
Hocquemiller R, Cortes D, Arango GJ, Myint SH, Cavé A, Angelo A, Muñoz V, Fournet A (1991) Isolement et synthese de l’espintanol, nouveau monoterpene antiparasitaire. J Nat Prod. 54:445–452
Inagaki K, Churchill E, Mochly-Rosen D (2006) Epsilon protein kinase C as a potential therapeutic target for the ischemic heart. Cardiovasc Res 70:222–230
Kawakami Y, Nishimoto H, Kitaura J, Maeda-Yamamoto M, Kato RM, Littman DR, Leitges M, Rawlings DJ, Kawakami T (2004) Protein kinase C betaII regulates Akt phosphorylation on Ser-473 in a cell type- and stimulus-specific fashion. J Biol Chem 279:47720–47725
Kawasaki K, Smith RSJ, Ch-M H, Sun J, Chao J, Liao JK (2003) Activation of the phosphatidylinositol 3-Kinase/protein kinase Akt pathway mediates nitric oxide-induced endothelial cell migration and angiogenesis. Mol Cell Biol 23:5726–5737
Kleinbongard P, Heusch G (2015) Extracellular signalling molecules in the ischaemic/reperfused heart – druggable and translatable for cardioprotection? Br J Pharmacol 172(8):2010–2025
Kloner RA (2011) No-Reflow phenomenon: maintaining vascular integrity. J Cardiovasc Pharmacol Ther 16:244–250
Lesnefsky EJ, Chen Q, Tandler B, Hoppel CL (2017) Mitochondrial dysfunction and myocardial ischemia-reperfusion: implications for novel therapies. Annu Rev Pharmacol Toxicol 57:535–565
Levrand S, Vannay-Bouchiche C, Pesse B, Pacher P, Feihl F, Waeber B, Liaudet L (2006) Peroxynitrite is a major trigger of cardiomyocyte apoptosis in vitro and in vivo. Free Radic Biol Med 41:886–895
Li L, Zhou X, Li N, Sun M, Lv J, Xu Z (2015) Herbal drugs against cardiovascular disease: traditional medicine and modern development. Drug Discov Today 20:1074–1086
Liperoti R, Vetrano DL, Bernabei R, Onder G (2017) Herbal medications in cardiovascular medicine. J Am Coll Cardiol 69:1188–1199
Lopera YE, Fantinelli J, González Arbeláez LF, Rojano B, Ríos JL, Schinella GR, Mosca SM (2013) Antioxidant activity and cardioprotective effect of a nonalcoholic extract of Vaccinium meridionale Swartz during ischemia-reperfusion in rats. Evid Based Complement Alternat Med 2013:516727
Ma XL, Lopez BL, Liu GL, Christopher TA, Ischiropoulos H (1997) Peroxynitrite aggravates myocardial reperfusion injury in the isolated perfused rat heart. Cardiovasc Res 36:195–204
Matsui T, Rosenzweig A (2005) Convergent signal transduction pathways controlling cardiomyocyte survival and function: the role of PI 3-kinase and Akt. J Mol Cell Cardiol 38:63–71
Mela L, Seitz S (1979) Isolation of mitochondria with emphasis on heart mitochondria from small amounts of tissue. Methods Enzymol 55:39–46
Morciano G, Bonora M, Campo G, Aquila G, Rizzo P, Giorgi C, Wieckowski MR, Pinton P (2017) Mechanistic role of mPTP in ischemia-reperfusion injury. Adv Exp Med Biol 982:169–189
Murphy E, Steenbergen C (2008) Mechanisms underlying acute protection from cardiac ischemia-reperfusion injury. Physiol Rev 88:581–609
Murray CJ, Lopez AD (2013) Measuring the global burden of disease. N Engl J Med 369:448–457
Nagoor Meeran MF, Jagadeesh GS, Selvaraj P (2016) Thymol, a dietary monoterpene phenol abrogates mitochondrial dysfunction in β-adrenergic agonist induced myocardial infarcted rats by inhibiting oxidative stress. Chem Biol Interact 244:159–168
Newman DJ, Cragg GM (2016) Natural products as sources of new drugs from 1981 to 2014. J Nat Prod 79:629–661
Niccoli G, Montone RA, Ibanez B, Thiele H, Crea F12, Heusch G, Bulluck H, Hausenloy DJ, Berry C, Stiermaier T, Camici PG, Eitel I (2019) Optimized treatment of ST-elevation myocardial infarction. Circ Res 125:245–258
Ong SB, Samangouei P, Kalkhoran SB, Hausenloy DJ (2015) The mitochondrial permeability transition pore and its role in myocardial ischemia reperfusion injury. J Mol Cell Cardiol 78:23–34
Pacher P, Schulz R, Liaudet L, Szabo C (2005) Nitrosative stress and pharmacological modulation of heart failure. Trends Pharmacol Sci 26:302–310
Pardo AC, Rinaldi GJ, Mosca SM (2015) Mitochondrial calcium handling in normotensive and spontaneously hypertensive rats: correlation with systolic blood pressure levels. Mitochondrion 20:75–81
Ping P, Takano H, Zhang J, Tang XL, Qiu Y, Li RC, Banerjee S, Dawn B, Balafonova Z, Bolli R (1999) Isoform-selective activation of protein kinase C by nitric oxide in the heart of conscious rabbits: a signaling mechanism for both nitric oxide-induced and ischemia-induced preconditioning. Circ Res 84:587–604
Raedschelders K, Ansley DM, Chen DDY (2012) The cellular and molecular origin of reactive oxygen species generation during myocardial ischemia and reperfusion. Pharmacol Ther 133:230–255
Rojano BA, Pérez E, Figadère B, Martin MT, Recio MC, Giner R, Ríos JL, Schinella G, Sáez J (2007) Constituents of Oxandra cf. xylopioides with anti-inflammatory activity. J Nat Prod 70:835–838
Rojano BA, Saez JA, Schinella GR, Quijano J, Vélez E, Gil A, Notario R (2008) Experimental and theoretical determination of the antioxidant properties of isoespintanol (2-isopropyl-3,6-dimethoxy-5-methylphenol). J Mol Struc 877:1–6
Santos MRV, Moreira FV, Fraga BP, Souza DP, Bonjardim LR, Quintans-Junior LJ (2011) Cardiovascular effects of monoterpenes: a review. Rev Bras Farmacogn 21:764–771
Shukla SK, Gupta S, Ojha SK, Sharma SB (2010) Cardiovascular friendly natural products: a promising approach in the management of CVD. Nat Prod Res 24:873–898
Soetkamp D, Nguyen TT, Menazza S, Hirschhäuser C, Hendgen-Cotta UB, Rassaf T, Schlüter KD, Boengler K, Murphy E, Schulz R (2014) S-nitrosation of mitochondrial connexin 43 regulates mitochondrial function. Basic Res Cardiol 109(5):433–452
Suzuki M1, Sasaki N, Miki T, Sakamoto N, Ohmoto-Sekine Y, Tamagawa M, Seino S, Marbán E, Nakaya H. (2002) Role of sarcolemmal K(ATP) channels in cardioprotection against ischemia/reperfusion injury in mice. J Clin Invest 109: 509-516.
Yang M, Camara AK, Wakim BT, Zhou Y, Gadicherla AK, Kwok WM, Stowe DF (2012) Tyrosine nitration of voltage-dependent anion channels in cardiac ischemia-reperfusion: reduction by peroxynitrite scavenging. Biochim Biophys Acta 1817:2049–2059
Zhang J, Baines CP, Zong C, Cardwell EM, Wang G, Vondriska TM, Ping P (2005) Functional proteomic analysis of a three-tier PKCε-Akt-eNOS signaling module in cardioprotection. Am J Physiol Heart Circ Physiol 288:H954–H961
Funding
This work was supported by the Grant M-203 from the National University of La Plata, Argentina, to Dr. S. Mosca.
Author information
Authors and Affiliations
Contributions
L.G.A. performed the western blots; A.C.P. performed the mitochondrial experiments; J.F. performed the isolated hearts experiments; B.J. isolated and provided the drug; G.Sch. contributed to the conception of the study; S.M. wrote the manuscript. All the authors contributed to the analysis and interpretation of data, critically reviewed, and approved the final draft.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
González Arbeláez, L.F., Ciocci Pardo, A., Fantinelli, J.C. et al. Isoespintanol, a monoterpene isolated from oxandra cf xylopioides, ameliorates the myocardial ischemia-reperfusion injury by AKT/PKCε/eNOS-dependent pathways. Naunyn-Schmiedeberg's Arch Pharmacol 393, 629–638 (2020). https://doi.org/10.1007/s00210-019-01761-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-019-01761-9