Abstract
Background
There is some in vitro evidence that the adult ciliary body might harbor an inactive population of stem/retinal progenitor cells (RPC), or that ciliary epithelial (CE) cells might have the capacity to trans-differentiate, which may result in a balance between neural and epithelial properties. We have reported alterations in the ciliary body (CB) and adjacent vitreous in vivo by endoscopic evaluation of human eyes with a history of retinal detachment (RD) and anterior proliferative vitreoretinopathy (PVR).
Methods
The present study examined with light microscopy three paraffin–embedded phthisic human eyes with RD and anterior PVR. One normal eye, exenterated for an orbital tumor, served as the control. All specimens were stained with hematoxilin and eosin safran (HES), and serial sections were immunostained with antibodies against EGFR, Ki67, CD133, NSE, rhodopsin, and GFAP.
Results
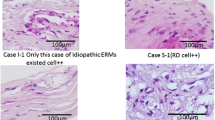
We observed: (1) an intense proliferation and displacement of clusters of CE cells into the vitreous base in a “neurosphere-like” fashion; (2) differentiation of CE cells towards early and late neuronal [photoreceptor (PR)] lineages; and (3) strong staining of EGF and EGFR in the CE. Such proliferation, migration, and differentiation were not present in the CE of the control eye. GFAP staining was intensely positive in the three detached retinae, and was negative in the CE of eyes with RD, as well as in the retina of the control eye.
Conclusions
Our observations suggest that EGFR-positive CE cells in the adult human eye in vivo with RD and PVR form “neurosphere-like” structures; their differentiation seems to be directed towards the neural and photoreceptor lineage, and not towards glial formation. In the adult human eye, the CE in a pathological retinal environment such as RD might provide a spontaneous source of donor cells for retinal transplantation.













Similar content being viewed by others
References
Tropepe V, Coles BL, Chiasson BJ, Horsford DJ, Elia AJ, McInnes RR, Van der Kooy D (2000) Retinal stem cells in the adult mammalian eye. Science 287:2032–2036
Ahmad I, Tang L, Pham H (2000) Identification of neural progenitors in the adult mammalian eye. Biochem Biophys Res Commun 270:517–521
Fischer AJ, Reh TA (2000) Indentification of a proliferating marginal zone of retinal progenitors in postnatal chickens. Dev Biol 220:197–210
Fischer AJ, Hendrickson A, Reh TA (2001) Immunocytochemical characterization of cysts in the peripheral retina and pars plana of the adult primate. Invest Ophthalmol Vis Sci 42:3256–3263
Moshiri A, Reh TA (2004) Persistent progenitors at the retinal margin of ptc+/- mice. J Neurosci 24:229–237
Das AV, James J, Rahnenführer J, Thoreson WB, Bhattacharya S, Zhao X, Ahmad I (2005) Retinal properties and potential of the adult mammalian ciliary epithelium stem cells. Vision Research 45:1653–1666
Inoue Y, Yanagi Y, Tamaki Y, Uchida S, Kawase Y, Araie M, Okochi H (2005) Clonogenic analysis of ciliary epithelial derived retinal progenitor cells in rabbits. Exp Eye Res 81(4):437–445
Gu P, Harwood LJ, Zhang X, Wylie M, Curry WJ, Cogliati T (2007) Isolation of retinal progenitor and stem cells from the porcine eye. Mol Vis 13:1045–1057
Hollyfield JG (1968) Differential addition of cells to the retina in Rana pipiens tadpoles. Dev Biol 18:163–179
Coles BL, Angénieux B, Inoue T, Del Rio K, Spence JR, McInnes RR, Arsenijevic Y, Van der Kooy D (2004) Facile isolation and the characterization of human retinal stem cells. Proc Natl Acad Sci USA 101(44):15772–15777
Mayer EJ, Carter DA, Ren Y, Hugues EH, Rice CM, Halfpenny CA, Scolding NJ, Dick AD (2005) Neural progenitor cells from postmortem adult human retina. Br J Ophthalmol 89(1):102–106
Coles BL, Horsford DJ, Mclnnes R, Van der Kooy D (2006) Loss of retinal progenitor cells leads to an increase in the retinal stem cell population in vivo. Eur J Neurosci 23:75–82
Klassen H, Zjaeian B, Kirov I, Young MJ, Schwartz PH (2007) Isolation of retinal progenitor cells from postmortem human tissue and comparison with autologous brain progenitors. J Neurosci Res 77:334–343
Xu H, Drina D, Sta I, Kielczewski JL, Valenta DF, Pease ME, Zack DJ, Quigley HA (2007) Characteristics of progenitor cells derived from adult ciliary body in mouse, rat, and human eyes. Invest Ophthalmol Vis Sci 48:1674–1682
MacNeil A, Pearson RA, MacLaren RE, Smith AJ, Sowden JC, Ali RR (2007) Comparative analysis of progenitor cells isolated from the iris, pars plana, and ciliary body of the adult porcine eye. Stem Cells 10:2430–2438
Wetts R, Serbedzija GN, Fraser SE (1989) Cell lineage analysis reveals multipotent precursors in the ciliary margin of the frog retina. Dev Biol 136:254–263
Perron M, Kanekar S, Vetter ML, Harris WA (1998) The genetic sequence of retinal development in the ciliary margin of the Xenopus eye. Dev Biol 199:185–200
Moe M, Kolberg R, Sandberg C, Vilk-Mo E, Olstorn H, Varghese M, Langmoen I, Nicolaissen B (2009) A comparison of epithelial and neural properties in progenitor cells derived from the adult human ciliary body and brain. Experimental Eye Research: 88(1):30–38
Cicero SA, Johnson D, Reyntjens S, Frase S, Connell S, Chow LML, Baker SJ, Sorrentino BP, Dyer MA (2009) Cells previously identified as retinal stem cells are pigmented epithelial cells. PNAS 106(16):6685–6690
Bathia B, Singhal S, Lawrence J, Khaw P, Limb A (2009) Distribution Of Muller stem cells within the neural retina: evidence for the existence of a ciliary margin-like zone in the adult human eye. Exp Eye Res 89:373–382
Boscher C (2007) Endoscopy. In: Kuhn F (ed) Ocular traumatology. Springer, Berlin, pp 473-484, section II, chapter 2.20
Boscher C (2001) Endoscopy for anterior proliferative vitreoretinopathy. AAO subspecialty day, Retina: A Retina Odyssey, pp 151-157
Bauer N, Fonseca AV, Florek M, Freund D, Jászai J, Bornhäuser M, Fargeas CA, Corbeil D (2008) New insights into the cell biology of hematopoietic progenitors by studying Prominin-1 (CD133). Cells Tissues Organs 188(1–2):127–138
Mizrak D, Brittan M, Alison MR (2008) CD133: molecule of the moment. J Pathol 214(1):3–9
Coskun V, Wu H, Blanchi B, Tsao S, Kim K, Zhao J, Biancotti JC, Hutnick L, Krueger RC Jr, Fan G, de Vellis J, Sun YE (2008) CD133+ neural stem cells in the ependyma of mammalian postnatal forebrain. Proc Natl Acad Sci USA 105(3):1026–1031
Carter DA, Dick AD, Mayer EJ (2009) CD 133+ adult human retinal cells remain undifferentiated in Leukaemia Inhibitory Factor (LIF). BMC Opthtalmology 9:1. doi:10.1186/1471-2415-9-1
Weigmann A, Corbeil D, Hellwig A, Huttner WB (1997) Prominin, a novel microvilli-specific polytopic membrane protein of the apical surface of epithelial cells, is targeted to plasmalemmal protrusions of non-epithelial cells. Proc Natl Acad Sci USA 94:12425–12430
Simmons PJ, Peault B, Buck DW, Huttner WB (2000) The human AC133 hematopoietic stem cell antigen is also expressed in epithelial cells and targeted to plasma membrane protrusions. J Biol Chem 275:5512–5520
Corbeil D, Röper K, Fargeas CA, Joester A, Huttner WB (2001) Prominin: a story of cholesterol, plasma membrane protrusions and human pathology. Traffic 2:82–91
Fargeas CA, Joester A, Missol-Kolka E, Hellwig A, Huttner WB, Denis Corbeil D (2004) Identification of novel Prominin-1/CD133 splice variants with alternative C-termini and their expression in epididymis and testis. Journal of Cell Science 117:4301–4311
Maw MA, Corbeil D, Koch J, Hellwig A, Wilson-Wheeler JC, Bridges RJ, Kumaramanickavel G, John S, Nancarrow D, Röper K, Weigmann A, Huttner WB, Denton MJ (2000) A frameshift mutation in prominin (mouse)-like 1 causes human retinal degeneration. Hum Mol Genet 9:27–34
Sun Y, Kong W, Falk A, Hu J, Zhou L, Pollard S, Smith A (2009) CD133 (Prominin) negative human neural stem cells are clonogenic and tripotent. PLoS One 4(5):e5498, Epub 2009 May 11
Fischer AJ, Reh TA (2001) Muller glia are a potential source of neural regeneration in the postnatal chicken retina. Nat Neurosci 4:247–252
Fischer AJ, McGuire CR, Dierks BD, Reh TA (2002) Insulin and fibroblast growth factor 2 activate a neurogenic program in Muller glia of the chicken retina. J Neurosci 22:9387–9398
Wan J, Zheng H, Xiao H, She ZJ, Zhou GM (2007) Sonic hedgehog promotes stem-cell potential of Müller glia in the mammalian retina. Biochem Biophys Res Commun 363(2):347–354
Bernados RL, Barthel LK, Meyers JR, Raymond PA (2007) Late-stage neuronal progenitors in the retina are radial müller glia that function as retinal stem cells. J Neurosci 27(26):7028–7040
Monnin J, Morand-Villeneuve N, Michel G, Hicks D, Versaux-Botteri C (2007) Production of neurospheres from mammalian Müller cells in culture. Neurosci Lett 421(1):22–26
Lawrence JM, Singhal S, Bhatia B, Keegan DJ, Reh TA, Luthert PJ, Khaw PT, Limb GA (2007) MIO-M1 cells and similar muller glial cell lines derived from adult human retina exhibit neural stem cell characteristics. Stem Cells 25(8):2033–2043
Draberova E, Del Valle L, Gordon J, Markova V, Smejkalova B, Bertrand L, de Chadarevian JP, Agamanolis DP, Legido A, Khalili K, Dráber P, Katsetos C (2008) Class III beta-tubulin is constitutively coexpressed with glial fibrillary acidic protein and nestin in midgestational human fetal astrocytes: implications for phenotypic identity. J Neuropathol Exp Neurol 67(4):341–354
Alvarez-Buylla A, Garcia-Verdugo JM (2002) Neurogenesis in adult sub-ventricular zone. J Neurosci 22:629–634
Zhao X, Das AV, Soto-Leon F, Ahmad I (2005) Growth factor-responsive progenitors in the postnatal mammalian retina. Dev Dyn 232(2):349–358
Rapaport DH, Wong LL, Wood ED, Yasumura D, La Vail MM (2004) Timing and topography of cell genesis in the rat retina. J Comp Neurol 474(2):304–324
MacLaren RE, Pearson RA, MacNeil A, Douglas RH, Salt TE, Akimoto M, Swaroop A, Sowden JC, Ali RR (2006) Retinal repair by transplantation of photoreceptor precursors. Nature 444(7116):203–207
Lamba DA, Karl MO, Ware CB, Reh TA (2006) Efficient generation of retinal progenitor cells from human embryonic stem cells. Proc Natl Acad Sci USA 103:12769–12774
Martins RAP, Pearson RA (2008) Control of cell proliferation by neurotransmitters in the developing vertebrate retina. Brain Res 1192:37–60
McFarlane S, Zuber ME, Holt CE (1998) A role for the fibroblast growth factor receptor in cell fate decisions in the developing vertebrate retina. Development 125(20):3967–3975
Fischer AJ, Reh TA (2003) Growth factors induce neurogenesis in the ciliary body. Dev Biol 259:225–240
Giordano F, De Marzo A, Vetrini F, Marigo V (2007) Fibroblast growth factor and epidermal growth factor differently affect differentiation of murine retinal stem cells in vitro. Mol Vis 2(13):1842–1850
Reh TA, Tully T (1986) Regulation of Tyrosine hydroxylase-containing amacrine cell number in larval frog retina. Dev Biol 114:463–469
Ooto S, Akagi T, Kageyam R, Mandai M, Honda Y, Takahasi M (2004) Potential for neural regeneration after neurotoxic injury in the adult mammalian retina. Proc Natl Acad Sci USA 101:13654–13659
Nickerson PE, Emsley JG, Myers T, Clarke DB (2007) Proliferation and expression of progenitor and mature retinal phenotypes in the adult mammalian ciliary body after retinal ganglion cell injury. Invest Ophthalmol Vis Sci 48(11):5266–5275
Nishiguchi KM, Kaneko H, Nakamura M, Kachi S, Terasaki H (2008) Identification of photoreceptor precursors in the pars plana during ocular development and after retinal injury. Invest Ophthalmol Vis Sci 49(1):422–428
Jens FK, Kiilgaard J, Prause U (2007) Subretinal posterior pole injury induces selective proliferation of RPE cells in the periphery in vivo studies in pigs. Invest Ophthalmol Vis Sci 48(1):355–360
Moe M, Varghese M, Danilov A, Westerlund U, Ram-Pettersen J, Brundin L, Svensson M, Berg-Johnsen J, Langmoen I (2005) Multipotent progenitor cells from the adult human brain: neurophysiological differentiation to mature neurons. Brain 128:2189–2199
Logan A, Ahmed Z, Baird A, Gonzalez AM, Berry M (2006) Neurotrophic factor synergy is required for neuronal survival and disinhibited axon regeneration after CNS injury. Brain 129(2):490–502
Author information
Authors and Affiliations
Corresponding author
Additional information
The Institutional IRB did not require a permit for this study. The work is in accordance with the Code of Ethics of the World Medical Association (Declaration of Helsinki) for experiments involving humans.
This study was not sponsored.
The authors do not have any financial interest with regard to this manuscript.
The authors have full control of all primary data, and they agree to allow Graefe’s Archive for Clinical and Experimental Ophthalmology to review their data if requested.
Rights and permissions
About this article
Cite this article
Ducournau, Y., Boscher, C., Adelman, R.A. et al. Proliferation of the ciliary epithelium with retinal neuronal and photoreceptor cell differentiation in human eyes with retinal detachment and proliferative vitreoretinopathy. Graefes Arch Clin Exp Ophthalmol 250, 409–423 (2012). https://doi.org/10.1007/s00417-011-1797-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-011-1797-3