Abstract
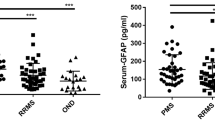
Primary and secondary progressive forms of multiple sclerosis (PPMS and SPMS) have different pathological characteristics. However, it is unknown whether neurodegenerative mechanisms are shared. We measured cerebrospinal fluid (CSF) levels of neurofilament (Nf) light and heavy isoforms and N-acetylaspartic acid (NAA) in 21 PP, 10 SPMS patients and 15 non-inflammatory neurological disease controls (NINDC). Biomarkers were related to Expanded Disability Status Scale (EDSS) and Multiple Sclerosis Severity Score (MSSS) over a long period of follow-up [median (interquartile range) 9 (5.5–12.5) years] in 19 PPMS and 4 SPMS patients, and to T2 lesion load, T1 lesion load, and brain parenchymal fraction at the time of lumbar puncture. Nf light was higher in PPMS (p < 0.005) and Nf heavy was increased in both SPMS and PPMS (p < 0.05 and p < 0.01) compared to NINDC, but were comparable between the two MS subtypes. Nf heavy was a predictor of the ongoing disability measured by MSSS (R 2 = 0.17, β = 0.413; p < 0.05). Conversely, Nf light was the only predictor of the EDSS annual increase (R 2 = 0.195, β = 0.441; p < 0.05). The frequency of abnormal biomarkers did not differ between the two MS progressive subtypes. Our data suggest that PP and SPMS likely share similar mechanisms of axonal damage. Moreover, Nf heavy can be a biomarker of ongoing axonal damage. Conversely, Nf light can be used as a prognostic marker for accumulating disability suggesting it as a good tool for possible treatment monitoring in the progressive MS forms.

Similar content being viewed by others
References
Tallantyre EC, Bø L, Al-Rawashdeh O et al (2009) Greater loss of axons in primary progressive multiple sclerosis plaques compared to secondary progressive disease. Brain 132:1190–1199
Thompson AJ, Kermode AG, Wicks D et al (1991) Major differences in the dynamics of primary and secondary progressive multiple sclerosis. Ann Neurol 29:53–62
Teunissen CE, Iacobaeus E, Khademi M et al (2009) Combination of CSF N-acetylaspartate and neurofilaments in multiple sclerosis. Neurology 72:1322–1329
Montalban X, Sastre-Garriga J, Tintoré M et al (2009) A single-center, randomized, double-blind, placebo-controlled study of interferon beta-1b on primary progressive and transitional multiple sclerosis. Mult Scler 15:1195–1205
Teunissen CE, Petzold A, Bennett JL et al (2009) A consensus protocol for the standardization of cerebrospinal fluid collection and biobanking. Neurology 73:1914–1922
Norgren N, Rosengren L, Stigbrand T (2003) Elevated neurofilament levels in neurological diseases. Brain Res 987:25–31
Petzold A, Altintas A, Andreoni L et al (2010) Neurofilament ELISA validation. J Immunol Methods 352:23–31
Koel-Simmelink MJA, Vennegoor A, Killestein J et al (2014) The impact of pre-analytical variables on the stability of neurofilament proteins in CSF, determined by a novel validated SinglePlex Luminex assay and ELISA. J Immunol Methods 402:43–49
Kuhle J, Leppert D, Petzold A et al (2011) Neurofilament heavy chain in CSF correlates with relapses and disability in multiple sclerosis. Neurology 76:1206–1213
Khalil M, Enzinger C, Langkammer C et al (2013) CSF neurofilament and N-acetylaspartate related brain changes in clinically isolated syndrome. Mult Scler 19:436–442
Norgren N, Sundström P, Svenningsson A et al (2004) Neurofilament and glial fibrillary acidic protein in multiple sclerosis. Neurology 63:1586–1590
Kuhle J, Malmeström C, Axelsson M et al (2013) Neurofilament light and heavy subunits compared as therapeutic biomarkers in multiple sclerosis. Acta Neurol Scand 128:e33–e36
Romme Christensen J, Börnsen L, Khademi M et al (2013) CSF inflammation and axonal damage are increased and correlate in progressive multiple sclerosis. Mult Scler 19:877–884
Petzold A, Eikelenboom MJ, Keir G et al (2005) Axonal damage accumulates in the progressive phase of multiple sclerosis: three year follow up study. J Neurol Neurosurg Psychiatry 76:206–211
Jasperse B, Jakobs C, Eikelenboom MJ et al (2007) N-acetylaspartic acid in cerebrospinal fluid of multiple sclerosis patients determined by gas-chromatography-mass spectrometry. J Neurol 254:631–637
Kuhle J, Plattner K, Bestwick JP et al (2013) A comparative study of CSF neurofilament light and heavy chain protein in MS. Mult Scler 19:1597–1603
Petzold A, Eikelenboom MI, Keir G et al (2006) The new global multiple sclerosis severity score (MSSS) correlates with axonal but not glial biomarkers. Mult Scler 12:325–328
Llufriu S, Kornak J, Ratiney H et al (2014) Magnetic resonance spectroscopy markers of disease progression in multiple sclerosis. JAMA Neurol 71:840–847
Salzer J, Svenningsson A, Sundström P (2010) Neurofilament light as a prognostic marker in multiple sclerosis. Mult Scler 16:287–292
Teunissen C, Menge T, Altintas A et al (2013) Consensus definitions and application guidelines for control groups in cerebrospinal fluid biomarker studies in multiple sclerosis. Mult Scler 19:1802–1809
Eikelenboom MJ, Petzold A, Lazeron RHC et al (2003) Multiple sclerosis: neurofilament light chain antibodies are correlated to cerebral atrophy. Neurology 60:219–223
Ghonemi MO, Rabah AA, Saber HM, Radwan W (2013) Role of Phosphorylated Neurofilament H as a diagnostic and prognostic marker in traumatic brain injury. Egypt J Crit Care Med 1:139–144
Acknowledgments
This study was performed in the context of the BioMS-eu network (http://www.bioms.eu).
Conflicts of interest
The authors declare no financial or other conflicts of interest. In addition, Dr. Korth has a patent EPA 05715297.7-2403 issued. Dr. Teunissen provided consultation for Fujirebio/Innogenetics, and for Roche International Advisory Board; received a grant from Grant of National Science Foundation for joint Programming project BIOMARKAPD, outside the submitted work. In addition, Dr. Teunissen has a patent Novel CSF biomarkers for Alzheimer’s disease pending, and a patent Bri2 as a biomarker for Alzheimer’s disease pending. Dr Killestein received grants from Biogen Idec, Bayer Schering, Teva, Merck-Serono, Genzyme, Novartis, Glaxo SK, UCB. Dr. Tintore reports personal fees and non-financial support for scientific meetings from Biogen-Idec, Bayer, Merck-Serono, Genzyme, Novartis, Teva and Sanofi-Aventis outside the submitted work.
Ethical standard
This study have been approved by the appropriate ethics committee and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. All persons gave their informed consent prior to their inclusion in the study.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Trentini, A., Comabella, M., Tintoré, M. et al. N-Acetylaspartate and neurofilaments as biomarkers of axonal damage in patients with progressive forms of multiple sclerosis. J Neurol 261, 2338–2343 (2014). https://doi.org/10.1007/s00415-014-7507-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-014-7507-4