Abstract
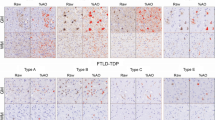
Although frontotemporal lobar degeneration with ubiquitin/TDP-43-positive inclusions (FTLD-TDP) and Pick’s disease are common pathological substrates in sporadic FTLD, clinical differentiation of these diseases is difficult. We performed a retrospective review of medical records and semiquantitative examination of neuronal loss of 20 sporadic FTLD-TDP and 19 Pick’s disease cases. Semantic dementia as the first syndrome developed only in FTLD-TDP patients. Impaired speech output in the early stage was five times more frequent in Pick’s disease than in FTLD-TDP. The total frequency of asymmetric motor disturbances (e.g., parkinsonism, pyramidal signs, and contracture) during the course was significantly more frequent in FTLD-TDP (78%) than in Pick’s disease cases (14%). Asymmetric pyramidal signs were found in 7 of 13 FTLD-TDP cases with corticospinal tract degeneration similar to primary lateral sclerosis. Frontotemporal dementia as the first syndrome was noted in both FTLD-TDP (28%) and Pick’s disease cases (64%); however, only FTLD-TDP cases subsequently developed asymmetric motor disturbances, and some of the cases further exhibited hemineglect. Concordant with these clinical findings, degeneration in the temporal cortex, caudate nucleus, putamen, globus pallidus, substantia nigra, and corticospinal tract was significantly more severe in FTLD-TDP, and degeneration in the frontal cortex tended to be more severe in Pick’s disease. Given these findings, the initial impairment of semantic memory or comprehension and subsequent asymmetric motor disturbances in sporadic FTLD patients predict sporadic FTLD-TDP rather than Pick’s disease, while initial behavioral symptoms or non-fluent aphasia without subsequent asymmetric motor disturbances predict Pick’s disease rather than sporadic FTLD-TDP.






Similar content being viewed by others
References
Arai T, Hasegawa M, Akiyama H, Ikeda K, Nonaka T, Mori H, Mann D, Tsuchiya K, Yoshida M, Hashizume Y, Oda T (2006) TDP-43 is a component of ubiquitin-positive tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Biochem Biophys Res Commun 351:602–611
Arima K, Miyasaka Y (1985) An autopsy case of Pick’s disease, with special reference to light and electron microscopical findings of Pick bodies and ballooned neurons. Neuropathology 6:325–339 in Japanese with English abstract
Baker M, Mackenzie IR, Pickering-Brown SM, Gass J, Rademakers R, Lindholm C, Snowden J, Adamson J, Sadovnick AD, Rollinson S, Cannon A, Dwosh E, Neary D, Melquist S, Richardson A, Dickson D, Berger Z, Eriksen J, Robinson T, Zehr C, Dickey CA, Crook R, McGowan E, Mann D, Boeve B, Feldman H, Hutton M (2006) Mutations in progranulin cause tau-negative frontotemporal dementia linked to chromosome 17. Nature 442:916–919
Beck J, Rohrer JD, Campbell T, Isaacs A, Morrison KE, Goodall EF, Warrington EK, Stevens J, Revesz T, Holton J, Al-Sarraj S, King A, Scahill R, Warren JD, Fox NC, Rossor MN, Collinge J, Mead S (2008) A distinct clinical, neuropsychological and radiological phenotype is associated with progranulin gene mutations in a large UK series. Brain 131:706–720
Boeve BF, Baker M, Dickson DW, Parisi JE, Giannini C, Josephs KA, Hutton M, Pickering-Brown SM, Rademakers R, Tang-Wai D, Jack CR Jr, Kantarci K, Shiung MM, Golde T, Smith GE, Geda YE, Knopman DS, Petersen RC (2006) Frontotemporal dementia and parkinsonism associated with the IVS1+1G->A mutation in progranulin: a clinicopathologic study. Brain 129:3103–3114
Braak H, Braak E (1991) Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol 82:239–259
Broe M, Hodges JR, Schofield E, Shepherd CE, Kril JJ, Halliday GM (2003) Staging disease severity in pathologically confirmed cases of frontotemporal dementia. Neurology 60:1005–1011
Cairns NJ, Perry RH, Jaros E, Burn D, McKeith IG, Lowe JS, Holton J, Rossor MN, Skullerud K, Duyckaerts C, Cruz-Sanchez FF, Lantos PL (2003) Patients with a novel neurofilamentopathy: dementia with neurofilament inclusions. Neurosci Lett 341:177–180
Cairns NJ, Bigio EH, Mackenzie IR, Neumann M, Lee VM, Hatanpaa KJ, White CL 3rd, Schneider JA, Grinberg LT, Halliday G, Duyckaerts C, Lowe JS, Holm IE, Tolnay M, Okamoto K, Yokoo H, Murayama S, Woulfe J, Munoz DG, Dickson DW, Ince PG, Trojanowski JQ, Mann DM, Consortium for Frontotemporal Lobar Degeneration (2007) Neuropathologic diagnostic and nosologic criteria for frontotemporal lobar degeneration: consensus of the Consortium for Frontotemporal Lobar Degeneration. Acta Neuropathol 114:5–22
Cruts M, Gijselinck I, van der Zee J, Engelborghs S, Wils H, Pirici D, Rademakers R, Vandenberghe R, Dermaut B, Martin JJ, van Duijn C, Peeters K, Sciot R, Santens P, De Pooter T, Mattheijssens M, Van den Broeck M, Cuijt I, Vennekens K, De Deyn PP, Kumar-Singh S, Van Broeckhoven C (2006) Null mutations in progranulin cause ubiquitin-positive frontotemporal dementia linked to chromosome 17q21. Nature 442:920–924
Davies RR, Hodges JR, Kril JJ, Patterson K, Halliday GM, Xuereb JH (2005) The pathological basis of semantic dementia. Brain 128:1984–1995
Davion S, Johnson N, Weintraub S, Mesulam MM, Engberg A, Mishra M, Baker M, Adamson J, Hutton M, Rademakers R, Bigio EH (2007) Clinicopathologic correlation in PGRN mutations. Neurology 69:1113–1121
Dickson DW, Josephs KA, Amador-Ortiz C (2007) TDP-43 in differential diagnosis of motor neuron disorders. Acta Neuropathol 114:71–79
Forman MS, Farmer J, Johnson JK, Clark CM, Arnold SE, Coslett HB, Chatterjee A, Hurtig HI, Karlawish JH, Rosen HJ, Van Deerlin V, Lee VM, Miller BL, Trojanowski JQ, Grossman M (2006) Frontotemporal dementia: clinicopathological correlations. Ann Neurol 59:952–962
Fukui T, Sugita K, Kawamura M, Shiota J, Nakano I (1996) Primary progressive apraxia in Pick’s disease: a clinicopathologic study. Neurology 47:467–473
Gass J, Cannon A, Mackenzie IR, Boeve B, Baker M, Adamson J, Crook R, Melquist S, Kuntz K, Petersen R, Josephs K, Pickering-Brown SM, Graff-Radford N, Uitti R, Dickson D, Wszolek Z, Gonzalez J, Beach TG, Bigio E, Johnson N, Weintraub S, Mesulam M, White CL 3rd, Woodruff B, Caselli R, Hsiung GY, Feldman H, Knopman D, Hutton M, Rademakers R (2006) Mutations in progranulin are a major cause of ubiquitin-positive frontotemporal lobar degeneration. Hum Mol Genet 15:2988–3001
Grimes DA, Bergeron CB, Lang AE (1999) Motor neuron disease-inclusion dementia presenting as cortical-basal ganglionic degeneration. Mov Disord 14:674–680
Guerreiro RJ, Santana I, Bras JM, Revesz T, Rebelo O, Ribeiro MH, Santiago B, Oliveira CR, Singleton A, Hardy J (2008) Novel progranulin mutation: screening for PGRN mutations in a Portuguese series of FTD/CBS cases. Mov Disord 23:1269–1273
Hasegawa M, Arai T, Nonaka T, Kametani F, Yoshida M, Hashizume Y, Beach TG, Buratti E, Baralle F, Morita M, Nakano I, Oda T, Tsuchiya K, Akiyama H (2008) Phosphorylated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Ann Neurol 64:60–70
Higaki F, Yokota O, Ohishi M (2008) Factors predictive of survival after percutaneous endoscopic gastrostomy in the elderly: is dementia really a risk factor? Am J Gastroenterol 103:1011–1016
Hodges JR, Davies RR, Xuereb JH, Casey B, Broe M, Bak TH, Kril JJ, Halliday GM (2004) Clinicopathological correlates in frontotemporal dementia. Ann Neurol 56:399–406
Ikeda K (2000) Neuropathological discrepancy between Japanese Pick’s disease without Pick bodies and frontal lobe degeneration type of frontotemporal dementia proposed by Lund and Manchester Group. Neuropathology 20:76–82
Ikeda K, Akiyama H, Arai T, Ueno H, Tsuchiya K, Kosaka K (2002) Morphometrical reappraisal of motor neuron system of Pick’s disease and amyotrophic lateral sclerosis with dementia. Acta Neuropathol 104:21–28
Ikeda M, Ikeda K, Endo M, Haga C, Mizutani Y (1994) An autopsied case of slowly progressive Pick’s disease. Seishin Igaku (Clinical Psychiatry) 36:1167–1171 in Japanese with English abstract
Ikeda M, Ishikawa T, Tanabe H (2004) Epidemiology of frontotemporal lobar degeneration. Dementia Geriatr Cogn Disord 17:265–268
Ishino H, Ikeda H (1972) Four autopsy cases of Pick’s disease—clinicopathological findings (in Japanese with English abstract). Kyushu Neuro psychiatry 18:82–86
Jackson M, Lennox G, Lowe J (1996) Motor neuron disease-inclusion dementia. Neurodegeneration 5:339–350
Josephs KA, Holton JL, Rossor MN, Braendgaard H, Ozawa T, Fox NC, Petersen RC, Pearl GS, Ganguly M, Rosa P, Laursen H, Parisi JE, Waldemar G, Quinn NP, Dickson DW, Revesz T (2003) Neurofilament inclusion body disease: a new proteinopathy? Brain 126:2291–2303
Josephs KA, Holton JL, Rossor MN, Godbolt AK, Ozawa T, Strand K, Khan N, Al-Sarraj S, Revesz T (2004) Frontotemporal lobar degeneration and ubiquitin immunohistochemistry. Neuropathol Appl Neurobiol 30:369–373
Josephs KA, Ahmed Z, Katsuse O, Parisi JF, Boeve BF, Knopman DS, Petersen RC, Davies P, Duara R, Graff-Radford NR, Uitti RJ, Rademakers R, Adamson J, Baker M, Hutton ML, Dickson DW (2007) Neuropathologic features of frontotemporal lobar degeneration with ubiquitin-positive inclusions with progranulin gene (PGRN) mutations. J Neuropathol Exp Neurol 66:142–151
Josephs KA, Lin WL, Ahmed Z, Stroh DA, Graff-Radford NR, Dickson DW (2008) Frontotemporal lobar degeneration with ubiquitin-positive, but TDP-43-negative inclusions. Acta Neuropathol 116:159–167
Kawamura M, Mochizuki S (1999) Primary progressive apraxia. Neuropathology 19:249–258
Kertesz A, McMonagle P, Blair M, Davidson W, Munoz DG (2005) The evolution and pathology of frontotemporal dementia. Brain 128:1996–2005
Knibb JA, Xuereb JH, Patterson K, Hodges JR (2006) Clinical and pathological characterization of progressive aphasia. Ann Neurol 59:156–165
Knopman DS, Mastri AR, Frey WH 2nd, Sung JH, Rustan T (1990) Dementia lacking distinctive histologic features: a common non-Alzheimer degenerative dementia. Neurology 40:251–256
Kosaka K (1976) On aphasia of Pick’s disease—a review of our own 3 cases and 49 autopsy cases in Japan (in Japanese). Seishin Igaku (Clinical Psychiatry) 18:1181–1189
Kosaka K, Matsushita M, Oyanagi S, Hato K, Mehraein P (1980) Pick’s disease with concomitant traumatic brain damages–Five autopsied cases and a critical review of literatures (in Japanese with English abstract). Seishin Shinkeigaku Zasshi (Psychiatria et Neurologia Japonica) 82:33–47
Kosaka K, Ikeda K, Kobayashi K, Hamamoto J, Matsushita M (1985) On pyramidal tract lesions in Pick’s disease (in Japanese). Clinical Psychiatry 27:1171–1178
Le Ber I, Camuzat A, Hannequin D, Pasquier F, Guedj E, Rovelet-Lecrux A, Hahn-Barma V, van der Zee J, Clot F, Bakchine S, Puel M, Ghanim M, Lacomblez L, Mikol J, Deramecourt V, Lejeune P, de la Sayette V, Belliard S, Vercelletto M, Meyrignac C, Van Broeckhoven C, Lambert JC, Verpillat P, Campion D, Habert MO, Dubois B, Brice A, French research network on FTD/FTD-MND (2008) Phenotype variability in progranulin mutation carriers: a clinical, neuropsychological, imaging and genetic study. Brain 131:732–746
Mackenzie IR (2007) The neuropathology and clinical phenotype of FTD with progranulin mutations. Acta Neuropathol 114:49–54
Mackenzie IR, Foti D, Woulfe J, Hurwitz TA (2008) Atypical frontotemporal lobar degeneration with ubiquitin-positive, TDP-43-negative neuronal inclusions. Brain 131:1282–1293
Mackenzie IR, Neumann M, Bigio EH, Cairns NJ, Alafuzoff I, Kril J, Kovacs GG, Ghetti B, Halliday G, Holm IE, Ince PG, Kamphorst W, Revesz T, Rozemuller AJ, Kumar-Singh S, Akiyama H, Baborie A, Spina S, Dickson DW, Trojanowski JQ, Mann DM (2009) Nomenclature for neuropathologic subtypes of frontotemporal lobar degeneration: consensus recommendations. Acta Neuropathol 117:15–18
Masaki K, Maeda M, Furuhashi N, Kowa H (1996) Slowly-progressive aphasia and its symptoms—considered in reference to its related literature (in Japanese with English abstract). Kitasato Med 26:397–411
Masellis M, Momeni P, Meschino W, Heffner R Jr, Elder J, Sato C, Liang Y, St George-Hyslop P, Hardy J, Bilbao J, Black S, Rogaeva E (2006) Novel splicing mutation in the progranulin gene causing familial corticobasal syndrome. Brain 129:3115–3123
Mii T, Sudo K, Hayashi Y (1969) Ein Fall der Pickschen Krankheit (in Japanese with German abstract). Kyushu Neuro-psychiatry 15:106–113
Munoz-Garcia D, Ludwin SK (1984) Classic and generalized variants of Pick’s disease: a clinicopathological, ultrastructural, and immunocytochemical comparative study. Ann Neurol 16:467–480
Munoz DG, Dickson DW, Bergeron C, Mackenzie IR, Delacourte A, Zhukareva V (2003) The neuropathology and biochemistry of frontotemporal dementia. Ann Neurol 54(Suppl 5):S24–S28
Neary D, Snowden JS, Gustafson L, Passant U, Stuss D, Black S, Freedman M, Kertesz A, Robert PH, Albert M, Boone K, Miller BL, Cummings J, Benson DF (1998) Frontotemporal lobar degeneration: a consensus on clinical diagnostic criteria. Neurology 51:1546–1554
Neumann M, Sampathu DM, Kwong LK, Truax AC, Micsenyi MC, Chou TT, Bruce J, Schuck T, Grossman M, Clark CM, McCluskey LF, Miller BL, Masliah E, Mackenzie IR, Feldman H, Feiden W, Kretzschmar HA, Trojanowski JQ, Lee VM (2006) Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science 314:130–133
Obi K, Akiyama H, Kondo H, Shimomura Y, Hasegawa M, Iwatsubo T, Mizuno Y, Mochizuki H (2008) Relationship of phosphorylated alpha-synuclein and tau accumulation to Abeta deposition in the cerebral cortex of dementia with Lewy bodies. Exp Neurol 210:409–420
Oda T, Tsuchiya K, Arai T, Togo T, Uchikado H, de Silva R, Lees A, Akiyama H, Haga C, Ikeda K, Kato M, Kato Y, Hara T, Onaya M, Hori K, Teramoto H, Tominaga I (2007) Pick’s disease with Pick bodies: an unusual autopsy case showing degeneration of the pontine nucleus, dentate nucleus, Clarke’s column, and lower motor neuron. Neuropathology 27:81–89
Onari K, Spatz H (1926) Anatomische Beiträge zur Lehre von Pickschen umschriebenen Größhirnrinden Atrophie (Picksche Krankheit). Z Ges Neurol Psychiatr 101:470–511
Ota S, Tsuchiya K, Anno M, Akiyama H (2005) An autopsy case of dementia lacking distinctive histologic features showing semantic dementia. No To Shinkei 57:233–238
Oyanagi S, Tanaka M, Omori T, Matsuchita M, Ishii T (1979) Regular arrangements of tubular structures in Pick bodies formed in an autopsied case of Pick’s disease (in Japanese with English abstract). Shinkei Kenkyū no Shimpo (Adv in Neurol Sci) 23:441–451
Paviour DC, Lees AJ, Josephs KA, Ozawa T, Ganguly M, Strand C, Godbolt A, Howard RS, Revesz T, Holton JL (2004) Frontotemporal lobar degeneration with ubiquitin-only-immunoreactive neuronal changes: broadening the clinical picture to include progressive supranuclear palsy. Brain 127:2441–2451
Pick A (1892) Ueber die Beziehungen der senilen Hirnatrophie zur Aphasie. Prager Med Wochenschr 17:165–167
Pickering-Brown SM (2007) Progranulin and frontotemporal lobar degeneration. Acta Neruopathol 114:39–47
Pickering-Brown SM, Rollinson S, Du Plessis D, Morrison KE, Varma A, Richardson AM, Neary D, Snowden JS, Mann DM (2008) Frequency and clinical characteristics of progranulin mutation carriers in the Manchester frontotemporal lobar degeneration cohort: comparison with patients with MAPT and no known mutations. Brain 131:721–731
Ratnavalli E, Brayne C, Dawson K, Hodges JR (2002) The prevalence of frontotemporal dementia. Neurology 58:1615–1621
Rabinovici GD, Furst AJ, O’Neil JP, Racine CA, Mormino EC, Baker SL, Chetty S, Patel P, Pagliaro TA, Klunk WE, Mathis CA, Rosen HJ, Miller BL, Jagust WJ (2007) 11C-PIB PET imaging in Alzheimer disease and frontotemporal lobar degeneration. Neurology 68:1205–1212
Roeber S, Mackenzie IR, Kretzschmar HA, Neumann M (2008) TDP-43-negative FTLD-U is a significant new clinico-pathological subtype of FTLD. Acta Neuropathol 116:147–157
Rossor MN, Revesz T, Lantos PL, Warrington EK (2000) Semantic dementia with ubiquitin-positive tau-negative inclusion bodies. Brain 123:267–276
Shi J, Shaw CL, Du Plessis D, Richardson AM, Bailey KL, Julien C, Stopford C, Thompson J, Varma A, Craufurd D, Tian J, Pickering-Brown S, Neary D, Snowden JS, Mann DM (2005) Histopathological changes underlying frontotemporal lobar degeneration with clinicopathological correlation. Acta Neuropathol 110:501–512
Snowden JS, Pickering-Brown SM, Mackenzie IR, Richardson AM, Varma A, Neary D, Mann DM (2006) Progranulin gene mutations associated with frontotemporal dementia and progressive non-fluent aphasia. Brain 129:3091–3102
Snowden J, Neary D, Mann D (2007) Frontotemporal lobar degeneration: clinical and pathological relationships. Acta Neuropathol 114:31–38
Spina S, Murrell JR, Huey ED, Wassermann EM, Pietrini P, Grafman J, Ghetti B (2007) Corticobasal syndrome associated with the A9D progranulin mutation. J Neuropathol Exp Neurol 66:892–900
Tsuchiya K, Ishizu H, Nakano I, Kita Y, Sawabe M, Haga C, Kuyama K, Nishinaka T, Oyanagi K, Ikeda K, Kuroda S (2001) Distribution of basal ganglia lesions in generalized variant of Pick’s disease: a clinicopathological study of four autopsy cases. Acta Neuropathol 102:441–448
Tsuchiya K, Ikeda M, Hasegawa K, Fukui T, Kuroiwa T, Haga C, Oyanagi S, Nakano I, Matsushita M, Yagishita S, Ikeda K (2001) Distribution of cerebral cortical lesions in Pick’s disease with Pick bodies: a clinicopathological study of six autopsy cases showing unusual clinical presentations. Acta Neuropathol 102:553–571
Tsuchiya K, Piao YS, Oda T, Mochizuki A, Arima K, Hasegawa K, Haga C, Kakita A, Hori K, Tominaga I, Yagishita S, Akiyama H, Takahashi H (2006) Pathological heterogeneity of the precentral gyrus in Pick’s disease: a study of 16 autopsy cases. Acta Neuropathol 112:29–42
Uchihara T, Tsuchiya K (2008) Neuropathology of Pick body disease. In: Duyckaerts C, Litvan I (eds) Handbook of clinical neurology, vol 89 (Third series), Dementias. Elsevier, Amsterdam, pp 415–430
Yokota O, Sasaki K, Fujisawa Y, Takahashi J, Terada S, Ishihara T, Nakashima H, Kugo A, Ata T, Ishizu H, Kuroda S (2005) Frequency of early and late-onset dementias in a Japanese memory disorders clinic. Eur J Neurol 12:782–790
Yokota O, Tsuchiya K, Oda T, Ishihara T, de Silva R, Lees AJ, Arai T, Uchihara T, Ishizu H, Kuroda S, Akiyama H (2006) Amyotrophic lateral sclerosis with dementia: an autopsy case showing many Bunina bodies, tau-positive neuronal and astrocytic plaque-like pathologies, and pallido-nigral degeneration. Acta Neuropathol 112:633–645
Yokota O, Tsuchiya K, Itoh Y, Ishizu H, Akiyama H, Ikeda M, Kuzuhara S, Otomo E (2006) Frontotemporal lobar degeneration with ubiquitin pathology: an autopsy case presenting with semantic dementia and upper motor neuron signs with a clinical course of 19 years. Acta Neuropathol 112:739–749
Yokota O, Tsuchiya K, Terada S, Ishizu H, Uchikado H, Ikeda M, Oyanagi K, Nakano I, Murayama S, Kuroda S, Akiyama H (2008) Basophilic inclusion body disease and neuronal intermediate filament inclusion disease: a comparative clinicopathological study. Acta Neuropathol 115:561–575
Acknowledgments
We would like to thank Ms. C. Haga (Department of Neuropathology, Tokyo Institute of Psychiatry) and Mr. A. Sasaki for help with the production of the manuscript. This work was supported by a grant-in-aid for scientific research from the Ministry of Education, Culture, Sports, Science and Technology (14570957) and a research grant from the Zikei Institute of Psychiatry. This work was supported by a grant-in-aid for scientific research from the Ministry of Education, Culture, Sports, Science and Technology (14570957) and a research grant from the Zikei Institute of Psychiatry.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yokota, O., Tsuchiya, K., Arai, T. et al. Clinicopathological characterization of Pick’s disease versus frontotemporal lobar degeneration with ubiquitin/TDP-43-positive inclusions. Acta Neuropathol 117, 429–444 (2009). https://doi.org/10.1007/s00401-009-0493-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-009-0493-4