Abstract
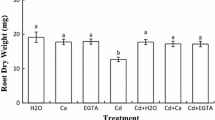
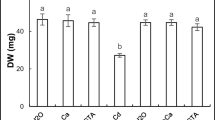
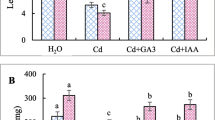
Impact of exogenous calcium and ethylene glycol tetraacetic acid (EGTA) supplement on chickpea (Cicer arietinum L.) germinating seeds exposed to cadmium stress for 6 days was studied. Ca and EGTA late treatment (3 days) alleviated growth inhibition and decreased Cd accumulation as well as lipid peroxidation and protein carbonylation in both root and shoot cells. Exogenous effector application relieved Cd-induced cell death which was associated with a constant level of ATP, which was considered as an apoptotic-like process. Redox balance was examined through the study of the redox state of pyridine nucleotide couples NAD+/NADH and NADP+/NADPH as well as their related oxidative [NAD(P)H-oxidase] and dehydrogenase (glucose-6-phosphate dehydrogenase, 6-phosphogluconate dehydrogenase and malate dehydrogenase) enzyme activities. The present research illustrated an ameliorative effect of Ca and EGTA on growth of Cd-exposed chickpea seedlings that occurs through the protection of sensitive cell sites from Cd-induced oxidation, namely membrane lipids and proteins, rather than the improvement of recycling capabilities of the cellular reducing power.







Similar content being viewed by others
References
Ali B, Xu X, Gill RA, Yang S, Ali S, Tahir M, Zhou W (2014) Promotive role of 5-aminolevulinic acid on mineral nutrients and antioxidative defense system under lead toxicity in Brassica napus. Ind Crop Prod 52:617–626
Atlante A, Giannattasio S, Bobba A, Gagliardi S, Petragallo V, Calissano P, Marra E, Passarella S (2005) An increase in the ATP levels occurs in cerebellar granule cells en route to apoptosis in which ATP derives from both oxidative phosphorylation and anaerobic glycolysis. Biochim Biophys Acta 1708(1):50–62
Bergmeyer HU, Bernt E (1974) Malate dehydrogenase. In: Bergmeyer HU (ed) Methods of enzymatic analysis. Academic Press Inc, New York, pp 1577–1580
Blazka ME, Shaikh ZA (1991) Differences in cadmium and mercury uptakes by hepatocytes: role of calcium channels. Toxicol Appl Pharmacol 110:355–363
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein dye binding. Anal Biochem 72:248–254
Bramm J (1992) Regulated expression of the calmodulin-related TCH genes in cultured Arabidopsis cells: induction by calcium and heat shock. Proc Natl Acad Sci USA 89:3213–3216
Casolo V, Petrussa E, Krajnáková J, Macri F, Vianello A (2005) Involvement of the mitochondrial K+ ATP channel in H2O2- or NO-induced programmed death of soybean suspension cell cultures. J Exp Bot 56:997–1006
Cha JY, Kim JY, Jung IJ, Kim MR, Melencion A, Alam SS, Yun DJ, Lee SY, Kim MG, Kim WY (2014) NADPH-dependent thioredoxin reductase A (NTRA) confers elevated tolerance to oxidative stress and drought. Plant Physiol Biochem 80:184–191
Chaoui A, El Ferjani E (2014) Heavy metal-induced oxidative damage is reduced by β -Estradiol application in lentil seedlings. Plant Growth Regul 74:1–9
Cho SC, Chao YY, Kao CH (2012) Calcium deficiency increases Cd toxicity and Ca is required for heat-shock induced Cd tolerance in rice seedlings. J Plant Physiol 169:892–898
Ernst W (1998) Effects of heavy metals in plants at the cellular and organismic level ecotoxicology. In: Gerrit S, Bernd M III (eds) Bioaccumulation and biological effects of chemicals. Wiley and Spektrum Akademisher Verlag, Berlin, pp 587–620
Farzadfar S, Zarinkamar F, Modarres-Sanavy SAM, Hojati M (2013) Exogenously applied calcium alleviates cadmium toxicity in Matricaria chamomilla L. plants. Environ Sci Pollut Res 20:1413–1422
Hasan SA, Hayat S, Ali B, Ahmad A (2008) 28-homobrassinolide protects chickpea (Cicer arietinum) from cadmium toxicity by stimulating antioxidant. Environ Pollut 151:60–66
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198
Hu KD, Hu LY, Li YH, Zhang FQ, Zhang H (2007) Protective roles of nitric oxide on germination and antioxidant metabolism in wheat seeds under copper stress. Plant Growth Regul 53:173–183
Ishida A, Ookubo K, Ono K (1987) Formation of hydrogen peroxide by NAD(P)H oxidation with isolated cell wall-associated peroxidase from cultured liverwort cells, Marchantia polymorpha L. Plant Cell Physiol 28:723–726
Islam MM, Hoque MA, Okuma E, Banu MNA, Shimoishi Y, Nakamura Y, Murata Y (2009) Exogenous proline and glycinebetaine increase antioxidant enzyme activities and confer tolerance to cadmium stress in cultured tobacco cells. J Plant Physiol 166(15):1587–1597
Kapoor D, Sharma R, Handa N, Kaur H, Rattan A, Yadav P, Gautam V, Kaur R, Bhardwaj R (2015) Redox homeostasis in plants under abiotic stress: role of electron carriers, energy metabolism mediators and proteinaceous thiols. Front environ Sci 3:13
Karmous I, Jaouani K, Chaoui A, El Ferjani E (2012) Proteolytic activities in Phaseolus vulgaris cotyledons under copper stress. Physiol Mol Biol Plants 18(4):337–343
Kinraide TB (1998) Three mechanisms for the calcium alleviation of mineral toxicities. Plant Physiol 118(2):513–520
Lam E (2004) Controlled cell death, plant survival and development. Nat Rev Mol Cell Biol 5:305–315
Leist M, Single B, Castoldi AF, Kühnle S, Nicotera P (1997) Intracellular adenosine triphosphate (ATP) concentration: a switch in the decision between apoptosis and necrosis. J Exp Med 185(8):1481–1486
Levine RL, Williams J, Stadtman ER, Shacter E (1994) Carbonyl assays for determination of oxidatively modified proteins. Methods Enzymol 233:346–357
Leόn AM, Palma JM, Corpas FJ, Gόmez M, Romero-Puertas MC, Chatterjee D, Mateos RM, Del Río LA, Sandalio LM (2002) Antioxidative enzymes in cultivars of pepper plants with different sensitivity to cadmium. Plant Physiol Biochem 40:813–820
Lozano RM, Wong JH, Yee BC, Peters A, Kobrehel K, Buchanan BB (1996) New evidence for a role for thioredoxin h in germination and seedling development. Planta 200:100–106
Matsumura H, Miyachi S (1980) Cycling assay for nicotinamide adenine dinucleotides. Methods Enzymol 69:465–470
Meng H, Hua S, Shamsi IH, Jilani G, Li Y, Jiang L (2009) Cadmium-induced stress on the seed germination and seedling growth of Brassica napus L., and its alleviation through exogenous plant growth regulators. Plant Growth Regul 58:47–59
Noctor G, Queval G, Gakière B (2006) NAD(P) synthesis and pyridine nucleotide cycling in plants and their potential importance in stress conditions. J Exp Bot 57(8):1603–1620
Perfus-Barbeoch L, Leonhardt N, Vavasseur A, Forestier C (2002) Heavy metal toxicity: cadmium permeates through calcium channels and disturbs the plant water status. Plant J 32:539–548
Potters G, Horemans N, Jansen MA (2010) The cellular redox state in plant stress biology: a charging concept. Plant Physiol Biochem 48:292–300
Rahoui S, Ben C, Chaoui A, Martinez Y, Yamchi A, Rickauer M, Gentzbittel L, El Ferjani E (2014) Oxidative injury and antioxidant genes regulation in cadmium-exposed radicles of six contrasted Medicago truncatula genotypes. Environ Sci Pollut Res 21(13):8070–8083
Rahoui S, Chaoui A, Ben C, Rickauer M, Gentzbittel L, El Ferjani E (2015) Effect of cadmium pollution on mobilization of embryo reserves in seedlings of six contrasted Medicago truncatula lines. Phytochemistry 111:98–106
Reinheckel T, Sitte N, Ullrich O, Kuckelkorn U, Davies KJ, Grune T (1998) Comparative resistance of the 20S and 26S proteasome to oxidative stress. Biochem J 335:637–642
Romero-Puertas MC, Rodríguez-Serrano M, Corpas FJ, Gómez M, Del Río LA, Sandalio LM (2004) Cadmium-induced subcellular accumulation of O ·−2 and H2O2 in pea leaves. Plant Cell Environ 27:1122–1134
Scharte J, Schön H, Tjaden Z, Weis E, Von Schaewen A (2009) Isoenzyme replacement of glucose-6-phosphate dehydrogenase in cytosol improves stress tolerance in plants. Proc Natl Acad Sci USA 106(19):8061–8066
Sun Q, Wang XR, Ding SM, Yuan XF (2005) Effects of exogenous organic chelators on phytochelatins production and its relationship with cadmium toxicity in wheat (Triticum aestivum L.) under cadmium stress. Chemosphere 60:22–31
Tian S, Lu L, Zhang J, Wang K, Brown P, He Z, Liang J, Yang X (2011) Calcium protects roots of Sedum alfredii H. against cadmium-induced oxidative stress. Chemosphere 84:63–69
Valderrama R, Corpas FJ, Carreras A, Gómez-Rodríguez MV, Chaki M, Pedrajas JR, Fernández-Ocaña A, Del Río LA, Barroso JB (2006) The dehydrogenase-mediated recycling of NADPH is a key antioxidant system against salt-induced oxidative stress in olive plants. Plant Cell Environ 29(7):1449–1459
Van Breusegem F, Dat JF (2006) Reactive oxygen species in plant cell death. Plant Physiol 141:384–390
Van Engelen DL, Sharpe-Pedler RC, Moorhead KK (2007) Effect of chelating agents and solubility of cadmium complexes on uptake from soil by Brassica juncea. Chemosphere 68:401–408
Wan G, Najeeb U, Jilani G, Naeem MS, Zhou W (2011) Calcium invigorates the cadmium-stressed Brassica napus L. plants by strengthening their photosynthetic system. Environ Sci Pollut Res 18(9):1478–1486
Wang CQ, Song H (2009) Calcium protects Trifolium repens L. seedlings against cadmium stress. Plant Cell Rep 28:1341–1349
Xu W, Li Y, He J, Ma Q, Zhang X, Chen G, Wang H, Zhang H (2010) Cd uptake in rice cultivars treated with organic acids and EDTA. J Environ Sci 22(3):441–447
Zhao Z, Hu X, Ross CW (1987) Comparison of tissue preparation methods for assay of nicotinamide coenzymes. Plant Physiol 84(4):987–988
Ziegler M (2005) A vital link between energy and signal transduction. Regulatory functions of NAD(P). FEBS J 272(18):4561–4564
Acknowledgments
This work was financially supported by the Tunisian Ministry of Higher Education and Scientific Research (UR11ES32) and by Graduate School of Environmental and Life Science, Okayama University (Japan). The authors wish to thank Mr. Bechir Azib and Mr. Abbes Oucherine for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sakouhi, L., Rahoui, S., Ben Massoud, M. et al. Calcium and EGTA Alleviate Cadmium Toxicity in Germinating Chickpea Seeds. J Plant Growth Regul 35, 1064–1073 (2016). https://doi.org/10.1007/s00344-016-9605-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-016-9605-2