Abstract
Key message
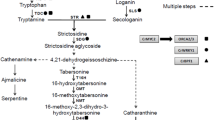
Blue and yellow light affected metabolism and the morphology. Blue and red promote the DOXP/MEP pathway. ADS gene expression was increased in plants cultivated under blue, promoting artemisinin content.
Abstract
Artemisinin-based combination therapies are the most effective treatment for highly lethal malaria. Artemisinin is produced in small quantities in the glandular trichomes of Artemisia annua L. Our aim was to evaluate the effect of light quality in A. annua cultivated in vitro under different light qualities, considering anatomical and morphological changes, the volatile composition, artemisinin content and the expression of two key enzymes for artemisinin biosynthesis. Yellow light is related to the increase in the number of glandular trichomes and this seemed to positively affect the molecular diversity in A. annua. Yellow light-stimulated glandular trichome frequency without triggered area enhancement, whereas blue light stimulated both parameters. Blue light enhanced the thickness of the leaf epidermis. The B-promoting effect was due to increased cell size and not to increased cell numbers. Green and yellow light positively influenced the volatile diversity in the plantlets. Nevertheless, blue and red light seemed to promote the DOXP/MEP pathway, while red light stimulates MVA pathway. Amorpha-4,11-diene synthase gene expression was significantly increased in plants cultivated under blue light, and not red light, promoting artemisinin content. Our results showed that light quality, more specifically blue and yellow light, positively affected secondary metabolism and the morphology of plantlets. It seemed that steps prior to the last one in the artemisinin biosynthesis pathway could be strongly influenced by blue light. Our work provides an alternative method to increase the amount of artemisinin production in A. annua without the use of transgenic plants, by the employment of blue light.



Similar content being viewed by others
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
- ACT:
-
Artemisinin combination therapies
- ADS:
-
Amorpha-4,11-diene synthase
- B:
-
Monochromatic blue light
- CRY:
-
Cryptochrome
- CYP71AV1:
-
Cytochrome P450 monooxygenase
- D:
-
Darkness
- dNTPs:
-
Deoxyribonucleoside 5′-triphosphate
- DOXP/MEP:
-
2Cmethyl-d-erythritol-4-phosphate pathway
- DTT:
-
Dithiothreitol
- FAA:
-
37% formaldehyde, glacial acetic acid, and 70% ethanol solution
- G:
-
Monochromatic green light
- Gl.T:
-
Glandular trichomes
- LED:
-
Light-emitting diode
- LRI:
-
Linear retention indices
- MS:
-
Murashige and Skoog
- MVA:
-
Mevalonic acid pathway
- NIST:
-
National Institute of Standards and Technology
- PAR:
-
Photosynthetically active radiation
- PDMS:
-
Polydimethylsiloxane
- PHOT:
-
Phototropin
- P.P:
-
Palisade parenchyma
- R:
-
Monochromatic red light
- SEM:
-
Scanning electron microscopy
- S.P:
-
Spongy parenchyma
- TT:
-
T-shape trichome
- W:
-
White fluorescent light
- Y:
-
Monochromatic yellow light
References
Adams RP (2007) Identification of essential oil components by gas chromatography/mass spectrometry, vol 456. Allured Publishing Corporation, Carol Stream
Ahmad M, Cashmore AR (1997) The blue-light receptor cryptochrome 1 shows functional dependence on phytochrome A or phytochrome B in Arabidopsis thaliana. Plant J 11:421–427
Ali M, Abbasi BH, Ahmad N, Khan H, Ali GS (2017) Strategies to enhance biologically active-secondary metabolites in cell cultures of Artemisia—current trends. Crit Rev Biotechnol 37(7):833–851
Alvarenga ICA, Pacheco FV, Silva ST, Bertolucci SKV, Pinto JEBP (2015) In vitro culture of Achillea millefolium L.: quality and intensity of light on growth and production of volatiles. Plant Cell Tissue Organ Cult 122(2):299–308
Arena C, Tsonev T, Doneva D, De Micco V, Michelozzi M, Brunetti C, Centritto M, Fineschi S, Velikova V, Loreto F (2016) The effect of light quality on growth, photosynthesis, leaf anatomy and volatile isoprenoids of a monoterpene-emitting herbaceous species (Solanum lycopersicum L.) and an isoprene-emitting tree (Platanus orientalis L.). Environ Exp Bot 130:122–132
Bergstrand KJ, Asp H, Schüssler HK (2015) Different light spectra is affecting growth and morphology of transplants of Solanum lycopersicum. In: International symposium on new technologies and management for greenhouses-GreenSys2015 1170:937–942
Bhakuni RS, Jain DC, Sharma RP, Kumar S (2000) Secondary metabolites of Artemisia annua and their biological activity. Curr Sci 80(1):35–48
Biesboer DD, Mahlberg PG (1978) Accumulation of non-utilizable starch in laticifers of Euphorbia heterophylla and E. myrsinites. Planta 143(1):5–10
Bilia AR, Santomauro F, Sacco C, Bergonzi MC, Donato R (2014) Essential oil of Artemisia annua L.: an extraordinary component with numerous antimicrobial properties. Evid Based Complement Altern Med 2014:7
Booth JK, Page JE, Bohlmann J (2017) Terpene synthases from Cannabis sativa. PLoS One 12:e0173911
Briggs WR, Olney MA (2001) Photoreceptors in plant photomorphogenesis to date. Five phytochromes, two cryptochromes, one phototropin, and one superchrome. Plant Physiol 125(1):85–88
Brisibe EA, Umoren UE, Brisibe F, Magalhäes PM, Ferreira JFS, Luthria D, Wu X, Prior RL (2009) Nutritional characterisation and antioxidant capacity of different tissues of Artemisia annua L. Food Chem 115(4):1240–1246
Brown GD (2010) The biosynthesis of artemisinin (Qinghaosu) and the phytochemistry of Artemisia annua L. (Qinghao). Molecules 15(11):7603–7698
Bryant L, Flatley B, Patole C, Brown GD, Cramer R (2015) Proteomic analysis of Artemisia annua—towards elucidating the biosynthetic pathways of the antimalarial pro-drug artemisinin. BMC Plant Biol 15(1):175
Cain AJ (1947) The use of Nile blue in the examination of lipoids. Q J Microsc Sci 88(3):383–392
Coşge Şenkal B, Kiralan M, Yaman C (2015) The effect of different harvest stages on chemical composition and antioxidant capacity of essential oil from Artemisia annua L. Tarim Bilim Derg 21:71–77
Demarsy E, Fankhauser C (2009) Higher plants use LOV to perceive blue light. Curr Opin Plant Biol 12(1):69–74
Dutta Gupta S, Karmakar A (2017) Machine vision based evaluation of impact of light emitting diodes (LEDs) on shoot regeneration and the effect of spectral quality on phenolic content and antioxidant capacity in Swertia chirata. J Photochem Photobiol B 174:162–172
Ennajdaoui H, Vachon G, Giacalone C, Besse I, Sallaud C, Herzog M, Tissier A (2010) Trichome specific expression of the tobacco (Nicotiana sylvestris) cembratrien-ol synthase genes is controlled by both activating and repressing cis-regions. Plant Mol Biol 73(6):673–685
Falcioni R, Moriwaki T, Bonato CM, de Souza LA, Nanni MR, Antunes WC (2017) Distinct growth light and gibberellin regimes alter leaf anatomy and reveal their influence on leaf optical properties. Environ Exp Bot 140:86–95
Fan X-X, Xu Z-G, Liu X-Y, Tang C-M, Wang L-W, Han X-l (2013) Effects of light intensity on the growth and leaf development of young tomato plants grown under a combination of red and blue light. Sci Hortic 153:50–55
Fiutak G, Michalczyk M, Filipczak-Fiutak M, Fiedor L, Surówka K (2019) The impact of LED lighting on the yield, morphological structure and some bioactive components in alfalfa (Medicago sativa L.) sprouts. Food Chem 285:53–58
Folta K, Carvalho S (2015) Photoreceptors and control of horticultural plant traits. HortScience 50:1274–1280
Folta KM, Pontin MA, Karlin-Neumann G, Bottini R, Spalding EP (2003) Genomic and physiological studies of early cryptochrome 1 action demonstrate roles for auxin and gibberellin in the control of hypocotyl growth by blue light. Plant J 36(2):203–214
Fukuda N, Fujita M, Ohta Y, Sase S, Nishimura S, Ezura H (2008) Directional blue light irradiation triggers epidermal cell elongation of abaxial side resulting in inhibition of leaf epinasty in geranium under red light condition. Sci Hortic 115:176–182
Furr M, Mahlberg PG (1981) Histochemical analyses of laticifers and glandular trichomes in Cannabis sativa. J Nat Prod 44(2):153–159
Galvão VC, Fankhauser C (2015) Sensing the light environment in plants: photoreceptors and early signaling steps. Curr Opin Neurobiol 34:46–53
Ghasemzadeh A, Ghasemzadeh N (2011) Flavonoids and phenolic acids: role and biochemical activity in plants and human. J Med Plant Res 5(31):6697–6703
Goins GD, Yorio NC, Sanwo MM, Brown CS (1997) Photomorphogenesis, photosynthesis, and seed yield of wheat plants grown under red light-emitting diodes (LEDs) with and without supplemental blue lighting. J Exp Bot 48:1407–1413
Gonçalves B, Correia CM, Silva AP, Bacelar EA, Santos A, Moutinho-Pereira JM (2008) Leaf structure and function of sweet cherry tree (Prunus avium L.) cultivars with open and dense canopies. Sci Hortic 116(4):381–387
Gregory M, Baas P (1989) A survey of mucilage cells in vegetative organs of the dicotyledons. Isr J Bot 38(2–3):125–174
Han J, Wang H, Lundgren A, Brodelius PE (2014) Effects of overexpression of AaWRKY1 on artemisinin biosynthesis in transgenic Artemisia annua plants. Phytochemistry 102:89–96
Hayes S, Velanis CN, Jenkins GI, Franklin KA (2014) UV-B detected by the UVR8 photoreceptor antagonizes auxin signaling and plant shade avoidance. Proc Natl Acad Sci USA 111(32):11894–11899
He Q, Fu X, Shi P, Liu M, Shen Q, Tang K (2017) Glandular trichome-specific expression of alcohol dehydrogenase 1 (ADH1) using a promoter-GUS fusion in Artemisia annua L. Plant Cell Tissue Organ Cult 130(1):61–72
Ho WE, Peh HY, Chan TK, Wong WSF (2014) Artemisinins: pharmacological actions beyond anti-malarial. Pharmacol Ther 142(1):126–139
Hong G-J, Hu W-L, Li J-X, Chen X-Y, Wang L-J (2009) Increased accumulation of artemisinin and anthocyanins in Artemisia annua expressing the arabidopsis blue light receptor CRY1. Plant Mol Biol Rep 27(3):334–341
Inthima P, Nakano M, Otani M et al (2017) Overexpression of the gibberellin 20-oxidase gene from Torenia fournieri resulted in modified trichome formation and terpenoid metabolites of Artemisia annua L. Plant Cell Tissue Organ Cult 129:223–236
Izzo LG, Arena C, De Micco V, Capozzi F, Aronne G (2019) Light quality shapes morpho-functional traits and pigment content of green and red leaf cultivars of Atriplex hortensis. Sci Hortic (Amsterdam) 246:942–950
Johansen DA (1940) Plant microtechnique. McGraw-Hill Book Company Inc., New York, p 523
Johkan M, Shoji K, Goto F, Hahida S, Yoshihara T (2012) Effect of green light wavelength and intensity on photomorphogenesis and photosynthesis in Lactuca sativa. Environ Exp Bot 75:128–133
Jolliffe HG, Gerogiorgis DI (2016) Plantwide design and economic evaluation of two continuous pharmaceutical manufacturing (CPM) cases: ibuprofen and artemisinin. Comput Chem Eng 91:269–288
Joulian D, König WA (1998) The atlas of spectral data of sesquiterpene hydrocarbons. Pergamon Press, Hamburg, p 658
Judd R, Bagley MC, Li M et al (2019) Artemisinin biosynthesis in non-glandular trichome cells of Artemisia annua. Mol Plant 12:704–714
Karimi E, Jaafar HZE, Ghasemzadeh A, Ibrahim MH (2013) Light intensity effects on production and antioxidant activity of flavonoids and phenolic compounds in leaves, stems and roots of three varieties of Labisia pumila Benth. Aust J Crop Sci 7(7):1016–1023
Kayser O (2018) Ethnobotany and medicinal plant biotechnology: from tradition to modern aspects of drug development. Planta Med 84(12/13):834–838
Kazaz B, Webster S, Yadav P (2016) Interventions for an artemisinin-based malaria medicine supply chain. Prod Oper Manag 25(9):1576–1600
Kim WS, Choi WJ, Lee S, Kim WJ, Lee DC, Sohn UD, Shin HS, Kim W (2015) Anti-inflammatory, antioxidant and antimicrobial effects of artemisinin extracts from Artemisia annua L. Korean J Physiol Pharmacol 19(1):21–27
Klermund C, Ranftl QL, Diener J, Bastakis E, Richter R, Schwechheimer C (2016) LLM-Domain B-GATA transcription factors promote stomatal development downstream of light signaling pathways in hypocotyls. Plant Cell 28(3):646–660
Kong S-G, Suzuki T, Tamura K, Mochizuki N, Hara-Nishimura I, Nagatani A (2006) Blue light-induced association of phototropin 2 with the Golgi apparatus. Plant J 45(6):994–1005
Lazzarini LES, Bertolucci SKV, Pacheco FV, dos Santos J, Silva ST, de Carvalho AA, Pinto JEBP (2018) Quality and intensity of light affect Lippia gracilis Schauer plant growth and volatile compounds in vitro. Plant Cell Tissue Organ Cult 135(3):367–379
Li H, Xu Z, Tang C (2010) Effect of light-emitting diodes on growth and morphogenesis of upland cotton (Gossypium hirsutum L.) plantlets in vitro. Plant Cell Tissue Organ Cult 103(2):155–163
Linstrom PJ, Mallard WG (2016) NIST Chemistry WebBook, NIST Standard Reference Database Number 69. https://doi.org/10.18434/T4D303. Accessed May 2017
Liu C-Z, Zhou H-Y, Zhao Y (2007) An effective method for fast determination of artemisinin in Artemisia annua L. by high performance liquid chromatography with evaporative light scattering detection. Anal Chim Acta 581(2):298–302
Liu M, Xu Z, Guo S, Tang C, Liu X, Jao X (2014) Evaluation of leaf morphology, structure and biochemical substance of balloon flower (Platycodon grandiflorum (Jacq.) A. DC.) plantlets in vitro under different light spectra. Sci Hortic 174:112–118
Liu M, Shi P, Fu X, Brodelius PE, Shen Q, Jiang W, He Q, Tang K (2016) Characterization of a trichome-specific promoter of the aldehyde dehydrogenase 1 (ALDH1) gene in Artemisia annua. Plant Cell Tissue Organ Cult 126(3):469–480
Liu H, Fu Y, Hu D, Yu J, Liu H (2018) Effect of green, yellow and purple radiation on biomass, photosynthesis, morphology and soluble sugar content of leafy lettuce via spectral wavebands “knock out”. Sci Hortic (Amsterdam) 236:10–17
Lommen WJM, Schenk E, Bouwmeester HJ, Verstappen FWA (2006) Trichome dynamics and artemisinin accumulation during development and senescence of Artemisia annua leaves. Planta Med 72(04):336–345
Ma D-M, Wang Z, Wang L, Alejos-Gonzales F, Sun M-A, Xie D-Y (2015) A genome-wide scenario of terpene pathways in self-pollinated Artemisia annua. Mol Plant 8(11):1580–1598
Ma YN, Chen CJ, Li QQ, Xu FR, Cheng YX, Dong X (2019) Monitoring antifungal agents of Artemisia annua against Fusarium oxysporum and Fusarium solani, associated with Panax notoginseng root-rot disease. Molecules 24(1):213
Macedo AF, Leal-Costa MV, Tavares ES, Lage CLS, Esquibel MA (2011) The effect of light quality on leaf production and development of in vitro-cultured plants of Alternanthera brasiliana Kuntze. Environ Exp Bot 70(1):43–50
Maes L, Van Nieuwerburgh FCW, Zhang Y, Reed DW, Pollier J, Vande Casteele SRF, Inzé D, Covello PS, Deforce DLD, Goossens A (2011) Dissection of the phytohormonal regulation of trichome formation and biosynthesis of the antimalarial compound artemisinin in Artemisia annua plants. New Phytol 189(1):176–189
Maffei M, Scannerini S (1999) Photomorphogenic and chemical responses to blue light in Mentha piperita. J Essent Oil Res 11(6):730–738
Mahmoud SS, Croteau RB (2002) Strategies for transgenic manipulation of monoterpene biosynthesis in plants. Trends Plant Sci 7:366–373
Massa GD, Kim H-H, Wheeler RM, Mitchell CA (2008) Plant productivity in response to LED lighting. HortScience 43(7):1951–1956
Mat Daud Z, Awang Y, Ismail F, Mohamed MTM (2016) Effects of red and blue spectrum of light emitting diodes (LEDs) on the growth and photosynthesis of lemon basil (Ocimum × citriodorum). III Int Conf Agric Food Eng 1152:183–188
McLafferty FW (2009) Wiley registry of mass spectral data. Wiley, New York
Melillo de Magalhães P, Dupont I, Hendrickx A, Joly A, Raas T, Dessy S, Sergent T, Schneider Y-J (2012) Anti-inflammatory effect and modulation of cytochrome P450 activities by Artemisia annua tea infusions in human intestinal Caco-2 cells. Food Chem 134(2):864–871
Mesa LE, Lutgen P, Velez ID, Segura AM, Sara M (2015) Artemisia annua L., potential source of molecules with pharmacological activity in human diseases. Am J Phytomed Clin Ther 3:436–450
Milhous WK, Weina PJ (2010) The botanical solution for malaria. Science 327(5963):279–280
Mishra S, Khurana JP (2017) Emerging roles and new paradigms in signaling mechanisms of plant cryptochromes. Crit Rev Plant Sci 36:89–115
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15(3):473–497
Nguyen KT, Arsenault PR, Weathers PJ (2011) Trichomes + roots + ROS = artemisinin: regulating artemisinin biosynthesis in Artemisia annua L. Vitro Cell Dev Plant 47(3):329–338
O’Brien TP, Feder N, McCully ME (1964) Polychromatic staining of plant cell walls by toluidine blue O. Protoplasma 59(2):368–373
Omer E (2013) Effect of soil type and seasonal variation on growth, yield, essential oil and artemisinin content of Artemisia annua L. Int Res J Hortic 1:15
Pacheco FV, de Paula Avelar R, Alvarenga ICA, Bertolucci SKV, de Alvarenga AA, Pinto JEBP (2016) Essential oil of monkey-pepper (Piper aduncum L.) cultivated under different light environments. Ind Crops Prod 85:251–257
Paerse A (1980) Seeds of plenty, seeds of want: social and economic implications of the green revolution. Oxford University Press, Oxford, p 262
Pashkovskiy PP, Kartashov AV, Zlobin IE, Pogosyan SI, Kuznetsov VV (2016) Blue light alters miR167 expression and microRNA-targeted auxin response factor genes in Arabidopsis thaliana plants. Plant Physiol Biochem 104:146–154
Peplow M (2018) Looking for cheaper routes to malaria medicines. Chem Eng News 96(11):29–31
Perazzo FF, Carvalho JCT, Carvalho JE, Rehder VLG (2003) Central properties of the essential oil and the crude ethanol extract from aerial parts of Artemisia annua L. Pharmacol Res 48(5):497–502
Peterson CA, Peterson RL, Robards AW (1978) A correlated histochemical and ultrastructural study of the epidermis and hypodermis of onion roots. Protoplasma 96(1):1–21
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29(9):e45
Pizzolato TD, Larson PR (1977) Axillary bud development in Populus deltoides. II. Late ontogeny and vascularization. Am J Bot 64(7):849–860
Poudel PR, Kataoka I, Mochioka R (2008) Effect of red- and blue-light-emitting diodes on growth and morphogenesis of grapes. Plant Cell Tissue Organ Cult 92(2):147–153
Pu G-B, Ma D-M, Wang H, Ye H-C, Liu B-Y (2013) Expression and localization of amorpha-4,11-diene synthase in Artemisia annua L. Plant Mol Biol Rep 31(1):32–37
Reale S, Fasciani P, Pace L, Angelis FD, Marcozzi G (2011) Volatile fingerprints of artemisinin-rich Artemisia annua cultivars by headspace solid-phase microextraction gas chromatography/mass spectrometry. Rapid Commun Mass Spectrom 25(17):2511–2516
Ren J, Guo S, Xu C, Yang C, Ai W, Tang Y, Qin L (2014) Effects of different carbon dioxide and LED lighting levels on the antioxidative capabilities of Gynura bicolor DC. Adv Space Res 53(2):353–361
Ruan J-X, Li J-X, Fang X, Wang L-J, Hu W-L, Chen X-Y, Yang C-Q (2016) Isolation and characterization of three new monoterpene synthases from Artemisia annua. Front Plant Sci 7:638. https://doi.org/10.3389/fpls.2016.00638
Schuerger AC, Brown CS, Stryjewski EC (1997) Anatomical features of pepper plants (Capsicum annuum L.) Grown under red light-emitting diodes supplemented with blue or far-red light. Ann Bot 79:273–282
Shi P, Fu X, Liu M, Shen Q, Jiang W, Li L, Sun X, Tang K (2017) Promotion of artemisinin content in Artemisia annua by overexpression of multiple artemisinin biosynthetic pathway genes. Plant Cell Tissue Organ Cult 129(2):251–259
Singh D, Basu C, Meinhardt-Wollweber M, Roth B (2015) LEDs for energy efficient greenhouse lighting. Renew Sustain Energy Rev 49:139–147
Su N, Wu Q, Shen Z, Xia K, Cui J (2013) Effects of light quality on the chloroplastic ultrastructure and photosynthetic characteristics of cucumber seedlings. Plant Growth Regul 73:227–235
Sultana S, Hu H, Gao L, Mao J, Luo J, Jongsma MA, Wang C (2015) Molecular cloning and characterization of the trichome specific chrysanthemyl diphosphate/chrysanthemol synthase promoter from Tanacetum cinerariifolium. Sci Hortic 185:193–199
Tan H, Xiao L, Gao S, Li Q, Chen J, Xiao Y, Ji Q, Chen R, Chen W, Zhang L (2015) TRICHOME AND ARTEMISININ REGULATOR 1 is required for trichome development and artemisinin biosynthesis in Artemisia annua. Mol Plant 8(9):1396–1411
Tariq U, Ali M, Abbasi BH (2014) Morphogenic and biochemical variations under different spectral lights in callus cultures of Artemisia absinthium L. J Photochem Photobiol B 130:264–271
Tzenkova R, Kamenarska Z, Draganov A, Atanassov A (2010) Composition of Artemisia annua essential oil obtained from species growing wild in Bulgaria. Biotechnol Biotechnol Equip 24(2):1833–1835
Untergasser A, Nijveen H, Rao X, Bisseling T, Geurts R, Leunissen JA (2007) Primer3Plus, an enhanced web interface to Primer3. Nucleic Acids Res 35(Web Server issue):W71–W74
van Den Dool H, Kratz PD (1963) A generalization of the retention index system including linear temperature programmed gas—liquid partition chromatography. J Chromatogr A 11:463–471
Wang Y, Zhang H, Zhao B, Yuan X (2001) Improved growth of Artemisia annua L hairy roots and artemisinin production under red light conditions. Biotechnol Lett 23(23):1971–1973
Wang E, Gan S, Wagner GJ (2002) Isolation and characterization of the CYP71D16 trichome-specific promoter from Nicotiana tabacum L. J Exp Bot 53(376):1891–1897
Wang H, Liu Y, Chong K, Liu BY, Ye HC, Li ZQ, Yan F, Li GF (2007) Earlier flowering induced by over-expression of CO gene does not accompany increase of artemisinin biosynthesis in Artemisia annua. Plant Biol 9(3):442–446
Wang H, Han J, Kanagarajan S, Lundgren A, Brodelius PE (2013) Trichome-specific expression of the amorpha-4,11-diene 12-hydroxylase (cyp71av1) gene, encoding a key enzyme of artemisinin biosynthesis in Artemisia annua, as reported by a promoter-GUS fusion. Plant Mol Biol 81(1):119–138
World Health Organization (2016) Artemisinin and artemisinin-based combination therapy resistance: status report, vol WHO/HTM/GMP/2016.5. World Health Organization, Geneva, p 12
World Health Organization (2017) World Malaria Report 2017. World Health Organization, Geneva, p 161
Wu H, Guo J, Chen S, Liu X, Zhou Y, Zhang X, Xu X (2013) Recent developments in qualitative and quantitative analysis of phytochemical constituents and their metabolites using liquid chromatography–mass spectrometry. J Pharm Biomed 72:267–291
Wu Q, Su N, Shen W, Cui J (2014) Analyzing photosynthetic activity and growth of Solanum lycopersicum seedlings exposed to different light qualities. Acta Physiol Plant 36:1411–1420
Xiao L, Tan H, Zhang L (2016) Artemisia annua glandular secretory trichomes: the biofactory of antimalarial agent artemisinin. Sci Bull 61(1):26–36
Xie D-Y, Ma D-M, Judd R, Jones AL (2016) Artemisinin biosynthesis in Artemisia annua and metabolic engineering: questions, challenges, and perspectives. Phytochem Rev 15(6):1093–1114
Xu W, Zou Z, Pei J, Huang L (2018) Longitudinal trend of global artemisinin research in chemistry subject areas (1983–2017). Bioorg Med Chem 26:5379–5387
Yadav RK, Sangwan RS, Sabir F, Srivastava AK, Sangwan NS (2014) Effect of prolonged water stress on specialized secondary metabolites, peltate glandular trichomes, and pathway gene expression in Artemisia annua L. Plant Physiol Biochem 74:70–83
Yu W, Liu Y, Song L, Jacobs DF, Du X, Ying Y, Shao Q, Wu J (2017) Effect of differential light quality on morphology, photosynthesis, and antioxidant enzyme activity in Camptotheca acuminata seedlings. J Plant Growth Regul 36(1):148–160
Zhang D, Sun W, Shi Y, Wu L, Zhang T, Xiang L (2018) Red and blue light promote the accumulation of artemisinin in Artemisia annua L. Molecules 23(6):1329
Zhao S, Fernald RD (2005) Comprehensive algorithm for quantitative real-time polymerase chain reaction. J Comput Biol 12(8):1047–1064
Zheng L, Van Labeke MC (2017) Long-term effects of red-and blue-light emitting diodes on leaf anatomy and photosynthetic efficiency of three ornamental pot plants. Front Plant Sci 8:917
Zhigzhitzhapova SV, Dylenova EP, Gulyaev SM, Randalova TE, Taraskin VV, Tykheev ZA, Radnaeva LD (2019) Composition and antioxidant activity of the essential oil of Artemisia annua L. Nat Prod Res. https://doi.org/10.1080/14786419.2018.1548461
Acknowledgements
We thank Prof. Pedro Melillo (CPQBA/UNICAMP) for providing the seeds and Durvalina Felix-Whipps and Rosângela de Almeida Epifanio (in memoriam) for technical and scientific support.
Funding
This work was supported by Foundation for Research Support of the State of Rio de Janeiro (FAPERJ) [E-26/111.372/2011]; National Council for Scientific and Technological Development (CNPq) [Grant numbers 310474/2015-9, 159779/2013-8]; Coordination of Improvement of Higher Education Personnel (CAPES); and Federal University of Rio de Janeiro State (UNIRIO).
Author information
Authors and Affiliations
Contributions
AFM supervised the project; AFM, EST, MA-F, HRB, ACAA-D and ALV designed the experiments; EML, EST and AFM wrote the manuscript, with the participation of FG-D, ALM and contributions from all authors; tissue culture experiments were conducted by EML and AFM; morphological assays were conducted by AFM, EST, TSSG and ACAA-D; volatile assays were advised by HRB and conducted by EML and AFM; artemisinin analysis was conducted by ALM under the supervision of ALV and MCM; gene expression assays were conducted by EML and FG-D under the supervision of MA-F. All authors critically revised the manuscript and gave their final approval.
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest.
Additional information
Communicated by Stefan Schillberg.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lopes, E.M., Guimarães-Dias, F., Gama, T.d.S.S. et al. Artemisia annua L. and photoresponse: from artemisinin accumulation, volatile profile and anatomical modifications to gene expression. Plant Cell Rep 39, 101–117 (2020). https://doi.org/10.1007/s00299-019-02476-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-019-02476-0