Abstract
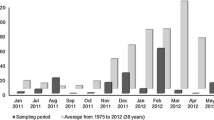
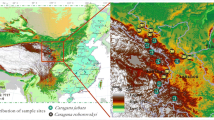
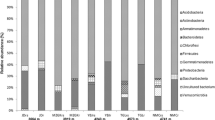
The rhizosphere is viewed as a deterministic environment led by the interaction between plants and microorganisms. In the case of semi-arid plants, this interaction is strengthened by the harshness of the environment. We tested the hypothesis that dry season represents a constraint on the bacterial diversity of the rhizosphere from semi-arid plants. To accomplish this, we sampled two leguminous species at five locations during the dry and rainy seasons in the Caatinga biome and characterised bacterial community structures using qPCR and 16S rRNA sequencing. We found that the main differences between seasons were due to reduced phylogenetic diversity caused by dryness. Variation partitioning indicated that environmental characteristics significant impacts in β-diversity. Additionally, distance decay relationship and taxa area relationship indicate a higher spatial turnover at the rainy season. During the dry season, decreased bacterial abundance is likely due to the selection of resistant or resilient microorganisms; with the return of the rain, the sensitive populations start to colonise the rhizosphere by a process that is strongly influenced by environmental characteristics. Thus, we propose that the reduction of PD and strong influence of environmental parameters on the assemblage of these communities make them prone to functional losses caused by climatic disturbances.






Similar content being viewed by others
References
Rotenberg E, Yakir D (2010) Contribution of semi-arid forests to the climate system. Science 327:451–454. doi:10.1126/science.1179998
Kavamura VN, Santos SN, Da Silva JL et al (2012) Screening of Brazilian cacti rhizobacteria for plant growth promotion under drought. Microbiol Res. doi:10.1016/j.micres.2012.12.002
Taketani RG, Kavamura VN, Mendes R (2014) Functional congruence of rhizosphere microbial communities associated to leguminous tree from Brazilian semiarid region. Environ Microbiol Rep. doi:10.1111/1758-2229.12187
Kavamura VN, Taketani RG, Lançoni MD et al (2013) Water regime influences bulk soil and rhizosphere of cereus jamacaru bacterial communities in the Brazilian caatinga biome. PLoS One 8, e73606. doi:10.1371/journal.pone.0073606
Placella SA, Brodie EL, Firestone MK (2012) Rainfall-induced carbon dioxide pulses result from sequential resuscitation of phylogenetically clustered microbial groups. Proc Natl Acad Sci U S A 109:10931–10936. doi:10.1073/pnas.1204306109
Barnard RL, Osborne CA, Firestone MK (2014) Changing precipitation pattern alters soil microbial community response to wet-up under a Mediterranean-type climate. ISME J 9:946–957. doi:10.1038/ismej.2014.192
Oliveira G, Araújo MB, Rangel TF et al (2012) Conserving the Brazilian semiarid (Caatinga) biome under climate change. Biodivers Conserv 21:2913–2926. doi:10.1007/s10531-012-0346-7
Engelbrecht BMJ, Comita LS, Condit R et al (2007) Drought sensitivity shapes species distribution patterns in tropical forests. Nature 447:80–82. doi:10.1038/nature05747
Chase JM (2007) Drought mediates the importance of stochastic community assembly. Proc Natl Acad Sci U S A 104:17430–17434. doi:10.1073/pnas.0704350104
Philippot L, Raaijmakers JM, Lemanceau P, van der Putten WH (2013) Going back to the roots: the microbial ecology of the rhizosphere. Nat Rev Microbiol 11:789–799. doi:10.1038/nrmicro3109
Souza DC, Oyama MD (2010) Climatic consequences of gradual desertification in the semi-arid area of northeast Brazil. Theor Appl Climatol 103:345–357. doi:10.1007/s00704-010-0302-y
Cavalcanti ER, Coutinho SFS (2005) Desertification in the northeast of brazil: the natural resources use and the land degradation. Soc Nat 1:891–900
Leal IR, Cardoso M, Silva DA et al (2005) Changing the course of biodiversity conservation in the Caatinga of northeastern Brazil. Conserv Biol 19:701–706
Lançoni MD, Taketani RG, Kavamura VN, de Melo IS (2013) Microbial community biogeographic patterns in the rhizosphere of two Brazilian semi-arid leguminous trees. World J Microbiol Biotechnol. doi:10.1007/s11274-013-1286-4
Barnard RL, Osborne C a, Firestone MK (2014) Changing precipitation pattern alters soil microbial community response to wet-up under a Mediterranean-type climate. ISME J 1–12. doi: 10.1038/ismej.2014.192
Barnard RL, Osborne CA, Firestone MK (2013) Responses of soil bacterial and fungal communities to extreme desiccation and rewetting. ISME J 7:2229–2241. doi:10.1038/ismej.2013.104
Queiroz LP (2006) The Brazilian caatinga: phytogeographical patterns inferred from distribution data of the Leguminosae. In: Penninigton RT, Lewis GP, Ratter JA (eds). Royal Botanical Garden, Edinburgh, pp 113–149
Caruso T, Chan Y, Lacap DC et al (2011) Stochastic and deterministic processes interact in the assembly of desert microbial communities on a global scale. ISME J 5:1406–1413. doi:10.1038/ismej.2011.21
Taketani RGRGG, dos Santos HF, van Elsas JD et al (2009) Characterisation of the effect of a simulated hydrocarbon spill on diazotrophs in mangrove sediment mesocosm. Antonie Van Leeuwenhoek 96:343–354. doi:10.1007/s10482-009-9351-6
Sogin MLL, Morrison HGG, Huber JAA et al (2006) Microbial diversity in the deep sea and the underexplored “rare biosphere”. Proc Natl Acad Sci 103:12115
Caporaso JG, Kuczynski J, Stombaugh J et al (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7:335–336. doi:10.1038/nmeth0510-335
Edgar RC (2010) Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26:2460–2461. doi:10.1093/bioinformatics/btq461
Caporaso JG, Bittinger K, Bushman FD et al (2010) PyNAST: a flexible tool for aligning sequences to a template alignment. Bioinformatics 26:266–267. doi:10.1093/bioinformatics/btp636
Wickham H (2009) ggplot2: elegant graphics for data analysis. Springer, New York
Hammer Ø, Harper DAT, Ryan PD (2001) Paleontological statistics software package for education and data analysis. Palaeontol Electron 4:9
Quince C, Lanzen A, Davenport RJ, Turnbaugh PJ (2011) Removing noise from pyrosequenced amplicons. BMC Bioinforma 12:38. doi:10.1186/1471-2105-12-38
McMurdie PJ, Holmes S (2014) Waste not, want not: why rarefying microbiome data is inadmissible. PLoS Comput Biol 10, e1003531. doi:10.1371/journal.pcbi.1003531
ter Braak Šmilauer, P. CJF (2002) CANOCO Reference Manual and CanoDraw for Windows User’s Guide: Software for Canonical Community Ordination (version 4.5). 500.
Winter C, Matthews B, Suttle CA (2013) Effects of environmental variation and spatial distance on Bacteria, Archaea and viruses in sub-polar and arctic waters. ISME J 7:1507–1518. doi:10.1038/ismej.2013.56
Oksanen P (2010) Vegan 1.17-0
Legendre P, Borcard D, Peres-Neto P (2005) Analyzing beta diversity: partitioning the spatial variation of community composition data. Ecol Monogr 75:435–450
Arrhenius O (1921) Species and area. J Ecol 9:95–99
Zinger L, Boetius A, Ramette A (2014) Bacterial taxa-area and distance-decay relationships in marine environments. Mol Ecol 23:954–964. doi:10.1111/mec.12640
Nekola JC, White PS (1999) The distance decay of similarity in biogeography and ecology. J Biogeogr 26:867–878
Manzoni S, Schimel JP, Porporato A (2012) Responses of soil microbial communities to water stress: results from a meta-analysis. Ecology 93:930–938
Bachar A, Al-Ashhab A, Soares MIM et al (2010) Soil microbial abundance and diversity along a low precipitation gradient. Microb Ecol 60:453–461. doi:10.1007/s00248-010-9727-1
Silver WL, Lugo AE, Keller M (1999) Soil oxygen availability and biogeochemistry along rainfall and topographic gradients in upland wet tropical forest soils. Biogeochemistry 44:301–328. doi:10.1007/BF00996995
Carson JK, Gonzalez-Quiñones V, Murphy DV et al (2010) Low pore connectivity increases bacterial diversity in soil. Appl Environ Microbiol 76:3936–3942. doi:10.1128/AEM.03085-09
Hu S, Chapin FS, Firestone MK et al (2001) Nitrogen limitation of microbial decomposition in a grassland under elevated CO2. Nature 409:188–191
Holmes A, Bowyer J, Holley M et al (2000) Diverse, yet-to-be-cultured members of the Rubrobacter subdivision of the Actinobacteria are widespread in Australian arid soils. FEMS Microbiol Ecol 33:111–120
Lennon JT, Jones SE (2011) Microbial seed banks: the ecological and evolutionary implications of dormancy. Nat Rev Microbiol 9:119–130. doi:10.1038/nrmicro2504
Evans SE, Wallenstein MD (2014) Climate change alters ecological strategies of soil bacteria. Ecol Lett 17:155–164. doi:10.1111/ele.12206
Rodrigues JLM, Pellizari VH, Mueller R et al (2013) Conversion of the Amazon rainforest to agriculture results in biotic homogenization of soil bacterial communities. Proc Natl Acad Sci U S A 110:988–993. doi:10.1073/pnas.1220608110
Griffiths BS, Philippot L (2013) Insights into the resistance and resilience of the soil microbial community. FEMS Microbiol Rev 37:112–129. doi:10.1111/j.1574-6976.2012.00343.x
Barthès A, Ten-Hage L, Lamy A et al (2014) Resilience of aggregated microbial communities subjected to drought—small-scale studies. Microb Ecol. doi:10.1007/s00248-014-0532-0
Thuiller W, Lavergne S, Roquet C et al (2011) Consequences of climate change on the tree of life in Europe. Nature 470:531–534. doi:10.1038/nature09705
Acknowledgments
The authors acknowledge the support of Cosme Corrêa dos Santos (UEFS) and Jorge Yoshio Tamashiro (University of Campinas) in identifying the plant species Mimosa tenuiflora and Piptadenia stipulacea (Benth.) Ducke, respectively. RGT was a recipient of a postdoctoral grant from FAPESP (2010/50799-7). This study was supported by Embrapa. The authors thank João Luiz da Silva, Dr. Rodrigo Mendes and Dr. Suikinai Santos for their support during sampling.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This project was conducted with the authorisation of the Institute of Environment and Renewable Natural Resources (IBAMA), process number 02001.004527/2011-90, and did not involve endangered or protected species.
Electronic Supplementary Material
Additional Supporting Information may be found in the online version of this article:
Appendix S1 Variation partitioning of bacterial community composition using partial redundancy analysis (RDA).
ESM 1
(DOC 181 kb)
Rights and permissions
About this article
Cite this article
Taketani, R.G., Lançoni, M.D., Kavamura, V.N. et al. Dry Season Constrains Bacterial Phylogenetic Diversity in a Semi-Arid Rhizosphere System. Microb Ecol 73, 153–161 (2017). https://doi.org/10.1007/s00248-016-0835-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-016-0835-4