Abstract
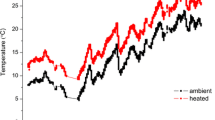
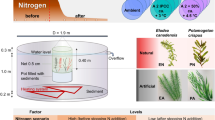
Submerged macrophytes are important contributors to primary production in clear water arctic and low arctic lakes. However, their production and potential coverage are hampered by the harsh climate conditions. In this study, we investigated the potential for increased macrophyte production and coverage in arctic lakes in a future warmer climate. In situ growth experiments with Callitriche hamulata were performed at 2, 4, 8 and 12 m depth in combination with nutrient assay experiments at 2 m depth. In addition, growth experiments were performed in the laboratory at four temperatures (5, 10, 15 and 20 °C) under saturated and light-limited conditions (150 and 25 µmol m−2 s−1). The results show that macrophyte growth in the low arctic lake, Badesø, is phosphorus limited, but they also indicate that nutrients are not the limiting factor for the macrophyte depth distribution. Rather the short growing season combined with low summer temperatures may limit the expansion of C. hamulata. Our study also shows that C. hamulata is a very temperature-sensitive plant, particularly around 10 °C. In a future warmer climate in the arctic, the thermocline in clear lakes is expected to expand deeper into the water column. Thus, at the present light conditions we can expect an expansion of both colonisation depth and coverage, which may affect the overall primary production in arctic lakes and the carbon flux and carbon cycling in the lake systems. Our findings strongly support recent predictions of increased growth and a more northerly distribution range of cold-temperature submerged macrophytes.






Similar content being viewed by others
References
Barko JW, Smart MR (1981) Comparative influences of light and temperature on the growth and metabolism of selected submersed freshwater macrophytes. Ecol Monogr 51:219–235. https://doi.org/10.2307/2937264
Bay C, Aastrup P, Nymand J (2008) The NERO line. A vegetation transect in Kobbefjord, West Greenland. National Environmental Research Institute, Aarhus University, Denmark.
Bay C, Raundrup K, Nymand J, Aastrup P, Krogh PH, Lauridsen TL, Johansson LS et al. (2012) Nuuk Basic: the BioBasis programme. In: Jensen LM (ed.) Nuuk Ecological Research Operations, 5th Annual Report 2011. Aarhus University, DCE—Danish Centre for Environment and Energy, pp 33–46.
Bay C, Nymand J, Aastrup P, Raundrup K, Krogh PH, Lauridsen TL, Lund M et al. (2013) Nuuk Basic: the BioBasis programme. In: Jensen LM, Rasch M (eds.) Nuuk Ecological Research Operations, 6th Annual Report 2012. Aarhus University, DCE—Danish Centre for Environment and Energy, pp 33-46.
Beca-Carretero P, Olesen B, Marbà N, Krause-Jensen D (2018) Response to experimental warming in northern eelgrass populations: comparison across a range of temperature adaptations. Mar Ecol Prog Ser 589:59–72. https://doi.org/10.3354/meps12439
Bennike O (2000) Palaeoecological studies of Holocene lake sediments from West Greenland. Palaeogeogr Palaeoclimatol Palaeoecol 155:285–304
Blindow I (1992) Decline of charophytes during eutrophication: comparison with angiosperms. Freshw Biol 28:9–14. https://doi.org/10.1111/j.1365-2427.1992.tb00557.x
Breton J, Vallieres C, Laurion I (2009) Limnological properties of permafrost thaw ponds in northeastern Canada. Can J Fish Aquat Sci 66:1635–1648
Brown JH, Gillooly JF, Allen AP, Savage VM, West GB (2004) Toward a metabolic theory of ecology. Ecology 85:1771–1789. https://doi.org/10.1890/03-9000
Brutemark A, Rengefors K, Anderson NJ (2006) An experimental investigation of phytoplankton nutrient limitation in two contrasting low artic lakes. Polar Biol 29:487–494. https://doi.org/10.1007/s00300-005-0079-0
Carr GM, Champers PA (1998) Macrophyte growth and sediment phosphorus and nitrogen in a Canadian prairie river. Freshw Biol 39:525–536. https://doi.org/10.1046/j.1365-2427.1998.00300.x
Canfield DE, Langeland KA, Linda SB, Haller WT (1985) Relations between water transparency and maximum depth of macrophyte colonization in lakes. J Aquat Plant Manag 23:25–28
Cazzanelli M, Forsström L, Rautio M, Michelsen A, Christoffersen KS (2012) Benthic production is the key to Daphnia middendorffiana survival in a shallow high arctic freshwater ecosystem. Freshw Biol 57:541–551
Chambers PA, Kalff J (1985) Depth distribution of biomass of submerged aquatic macrophyte communities in relation to Secchi depth. Can J Fish Aquat Sci 42:701–709. https://doi.org/10.2307/2260665
Christoffersen KS, Amsinck SL, Landkildehus F, Lauridsen TL, Jeppesen E (2008) Lake flora and fauna in relation to ice-melt, water temperature and chemistry at Zackenberg. Adv Ecol Res 40:371–389. https://doi.org/10.1016/S0065-2504(07)00016-5
Daniels WC, Kling GW, Giblin AE (2015) Benthic community metabolism in deep and shallow arctic lakes during 13 years of whole-lake fertilization. Limnol Oceanogr 60:1604–1618. https://doi.org/10.1002/lno.10120
Dale H (1986) Temperature and light: the determining factors in maximum depth distribution of aquatic macrophytes in Ontario, Canada. Hydrobiologia 133:73–77. https://doi.org/10.1007/BF00010804
Davison IR (1991) Environmental effects on algal photosynthesis: temperature. J Phycol 27:2–8. https://doi.org/10.1111/j.0022-3646.1991.00002.x
Dieffenbacher-Krall AC, Nurse AM (2005) Late-glacial and Holocene record of lake levels of Mathews Pond aand Whitehead Lake, northern Maine, USA. J Paleolimnol 34:283–310. https://doi.org/10.1007/s10933-005-4958-8
Duarte CM, Kalff J (1987) Latitudinal influences on the depths of maximum colonization and maximum biomass of submerged angiosperms in lakes. Can J Fish Aquat Sci 44:1758–1759. https://doi.org/10.1139/f87-215
Duarte CM, Kalff J (1990) Patterns in the submerged macrophyte biomass of lakes and the importance of the scale of analysis in the interpretation. Can J Fish Aquat Sci 47:357–363. https://doi.org/10.1139/f90-037
Duarte CM (1992) Nutrient concentration of aquatic plants, patterns across species. Limnol Oceanogr 37:882–889. https://doi.org/10.4319/lo.1992.37.4.0882
Finlay JC, Bowden WB (1994) Controls on production of bryophytes in an arctic stream. Freshw Biol 32:455–466. https://doi.org/10.1111/j.1365-2427.1994.tb01139.x
Friberg N, Dybkjær JB, Olafsson JS, Gislasons GM, Larsen SE, Lauridsen TL (2009) Relationships between structure and function in streams contrasting in temperature. Freshw Biol 54:2051–2068. https://doi.org/10.1111/j.1365-2427.2009.02234.x
GEM (2017) Greenland ecological monitoring data provided by Asiaq 2017. https://data.g-e-m.dk/. Accessed 12 June 2017
Gerloff GC, Krombholz PH (1966) Tissue analysis as a measure of nutrient availability for the growth of angiosperm aquatic plants. Limnol Oceanogr 11:529–537. https://doi.org/10.4319/lo.1966.11.4.0529
Grasshoff K, Erhardt M, Krembling K (eds) (1983) Methods of seawater analysis. Verlag Chemie, Weinheim, pp 159–228
Guo C, Ochyra R, Wu P-C, Seppelt RD, Yao Y-F, Bian L-G (2013) Warnstofia exannulata, an aquatic moss in the arctic: seasonal growth response. Clim Change 119:407–419. https://doi.org/10.1007/s10584-013-0724-5
Güsewell S (2004) N: P ratios in terrestrial plants: variation and functional significance. New Phytol 164:243–266. https://doi.org/10.1111/j.1469-8137.2004.01192.x
Hollis JA, van Gool JAM, Steenfelt A, Gaarde AA (2005) Greenstone belts in the central Godthåbsfjord region, southern West Greenland. Geol Surv Den Greenl Bull 7:65–68
Hyldgaard B, Sorrell B, Brix H (2014) Closely related freshwater species, Ceratophyllum demersum and C. submersum, differ in temperature response. Freshw Biol 59:777–788. https://doi.org/10.1111/fwb.12303
Jackson LJ, Lauridsen TL, Søndergaard M, Jeppesen E (2007) A comparison of shallow Danish and Canadian lakes and implications of climate change. Freshw Biol 52:1782–1792. https://doi.org/10.1111/j.1365-2427.2007.01809.x
Jassby AD, Platt T (1976) Mathematical formulation of the relationship between photosynthesis and light for phytoplankton. Limnol Oceanogr 21:540–547. https://doi.org/10.4319/lo.1976.21.4.0540
Jeppesen E, Jensen JP, Jensen C, Faafeng B, Hessen DO, Søndergaard M, Lauridsen T, Brettum P, Christoffersen K (2003) The impact of nutrient state and lake depth on top-down control in the pelagic zone of lakes: a study of 466 lakes from the temperate zone to the arctic. Ecosystems 6:313–325. https://doi.org/10.1007/PL00021503
Jeppesen E, Lauridsen TL, Christoffersen KS, Landkildehus F, Geertz-Hansen P, Amsinck SL, Søndergaard M et al (2017) The structuring role of fish in Greenland lakes: An overview based on contemporary and paleoecological studies of 87 lakes from the low and the high arctic. Hydrobiologia 800:99–113. https://doi.org/10.1007/s10750-017-3279-z
Kirk JTO (2011) Light and photosynthesis in aquatic ecosystems. Cambridge University Press, Cambridge, pp 1–665
Krause-Jensen D, Jensen C, Nielsen K, Petersen MF, Hansen DF, Laursen M, Platz EM et al (2002) Næringssaltbegrænsning af makroalger i danske kystområder. Miljøministeriet, Danmarks Miljøundersøgelser 392:1–114
Krause-Jensen D, Marbà N, Olesen B, Sejr M, Christensen PB, Rodrigues J, Renaud PE et al (2012) Seasonal sea ice cover as principal driver of spatial and temporal variation in depth extension and annual production of kelp in Greenland. Glob Change Biol 18:2981–2994. https://doi.org/10.1111/j.1365-2486.2012.02765
Lambers H, Chapin FS, Pons TL (2008) Plant physiological ecology. Springer, Berlin
Lauridsen TL, Jeppesen E, Landkildehus F, Søndergaard M (2001) Horizontal distribution of cladocerans in arctic Greenland lakes—impact of macrophytes and fish. Hydrobiologia 442:107–116. https://doi.org/10.1023/A:1017517919996
Lauridsen TL, Raundrup K, Nymand J, Jeppesen E (2011) Sø-monitering for vurdering af klimaændringer i lavarktis ved Nuuk. Vand & Jord 4:116–119 (in Danish)
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Meth Enzymol 148:350–382. https://doi.org/10.1016/0076-6879(87)48036-1
Maberly SC (1985) Photosynthesis by Fontinalis antipyreticaI. Interaction between photon irradiance, concentration of carbon dioxide and temperature. New Phytol 100(2):127–140. https://doi.org/10.1111/j.1469-8137.1985.tb02765.x
Maberly SC (1993) Morphological and photosynthetic characteristics of Potamogeton obtusifolius from different depths. J Aquat Plant Manag 31:34–39. https://doi.org/10.1080/02705060.2018.1443841
Madsen TV, Brix H (1997) Growth, photosynthesis and acclimation by two submerged macrophytes in relation to temperature. Oecologia 110:320–327. https://doi.org/10.1007/s004420050165
Marbà N, Krause-Jensen D, Olesen B, Christensen PB, Merzouk A, Rodrigues J, Wegeberg S, Wilce RT (2017) Climate change stimulates the growth of the intertidal macroalgae Ascophyllum nodosum near the northern distribution limit. Ambio 46:119–131. https://doi.org/10.1007/s13280-016-0873-7
Mesquita PS, Wrona FJ, Prowse TD (2010) Effects of retrogressive permafrost thaw slumping on sediment chemistry and submerged macrophytes in arctic tundra lakes. Freshw Biol 55:2347–2358. https://doi.org/10.1111/j.1365-2427.2010.02450.x
Middelboe AL, Markager S (1997) Depth limits and minimum light requirements of freshwater macrophytes. Freshw Biol 37:553–568. https://doi.org/10.1046/j.1365-2427.1997.00183.x
Olesen B, Madsen TV (2000) Growth and physiological acclimation to temperature and inorganic carbon availability by two submerged aquatic macrophyte species, Callitriche cophocarpa and Elodea canadensis. Func Ecol 14:252–260. https://doi.org/10.1046/j.1365-2435.2000.00412.x
Olesen B, Krause-Jensen D, Marbà N, Bondo-Christensen P (2015) Eelgrass Zostera marina in subarctic Greenland: dense meadows with slow biomass turnover in cold waters. Mar Ecol Prog Ser 518:107–121. https://doi.org/10.3354/meps11087
Pilon J, Santamaría L (2002) Clonal variation in the thermal response of the submerged aquatic macrophyte Potamogeton pectinatus. J Ecol 90:141–152. https://doi.org/10.1046/j.0022-0477.2001.00645.x
Prowse TD, Wrona FJ, Reist JD, Hobbie JE, Lévesque LM, Vincent WF (2006) General features of the arctic relevant to climate change in freshwater ecosystems. Ambio 35:330–338. https://doi.org/10.1579/0044-7447(2006)35[330:GFOTAR]2.0.CO;2
Rautio M, Dufresne F, Laurion I, Bonilla S, Vincent WF, Christoffersen KS (2011) Shallow freshwater ecosystems of the circumpolar Arctic. Ecoscience 18:204–222. https://doi.org/10.2980/18-3-3463
Riget F, Böcher J (2000) The fresh water environment. In: Born EW, Böcher J (eds) The ecology of Greenland. Grønlands Miljø og Naturforvaltning, Atuakkiorfik Undervisning, Nuuk, Greenland, pp 221–236
Riis T, Christoffersen KS, Baattrup-Pedersen A (2015) Mosses in high arctic lakes: in situ measurements of annual primary production and decomposition. Polar Biol 39:543–552. https://doi.org/10.1111/fwb.12368
Riis T, Olesen B, Clayton JS, Lambertini C, Brix H, Sorrell BK (2012) Growth and morphology in relation to temperature and light availability during the establishment of three invasive aquatic plant species. Aquat Bot 102:56–64. https://doi.org/10.1016/j.aquabot.2012.05.002
Riis T, Olesen B, Katborg C, Christoffersen KS (2010) Growth rate of an aquatic bryophyte (Warnstorfia fluitans (Hedw.) Loeske) from a high arctic lake: effect of nutrient concentration. Arctic 63:100–110. https://doi.org/10.14430/arctic650
Rooney N, Kalff J (2000) Inter-annual variation in submerged macrophyte community biomass and distribution: the influence of temperature and lake morphometry. Aquat Bot 68:321–335. https://doi.org/10.1016/S0304-3770(00)00126-1
Sand-Jensen K, Madsen TV (1991) Minimum light requirements of submerged freshwater macrophytes in laboratory growth experiments. J Ecol 79:749–764. https://doi.org/10.2307/2260665
Sand-Jensen K, Riis T, Markager S, Vincent WF (1999) Slow growth and decomposition of mosses in arctic lakes. Can J Fish Aquat Sci 56:388–393
Santamaría L, van Vierssen W (1997) Photosynthetic temperature responses of fresh- and brackish-water macrophytes: a review. Aquat Bot 58:135–150. https://doi.org/10.1016/S0304-3770(97)00015-6
Sheldon RB, Boylen CW (1977) Maximum depth inhabited by aquatic vascular plants. Am Midl Nat 97:248–254. https://doi.org/10.2307/2424706
Smart RM, Barko JW (1985) Laboratory culture of submersed freshwater macrophytes on natural sediments. Aquat Bot 21:251–326. https://doi.org/10.1016/0304-3770(85)90053-1
Squires MM, Lesack LFW, Hubert D (2002) The influence of water transparency on the distribution and abundance of macrophytes among lakes of the Mackenzie Delta, Western Canadian Arctic. Freshw Biol 47:2123–2135. https://doi.org/10.1046/j.1365-2427.2002.00959.x
Stendel M, Christensen JH, Petersen D (2008) Arctic climate and climate change with a focus on Greenland. Adv Ecol Res 40:13–43. https://doi.org/10.1016/S0065-2504(07)00002-5
Søndergaard M, Phillips G, Hellsten S, Kolada A, Ecke A, Mäemets H, Mjelde M et al (2013) Maximum growing depth of submerged macrophytes in European lakes. Hydrobiologia 704:165–177. https://doi.org/10.1007/s10750-012-1389-1
Tank SE, Lesack LFW, Hesslein RH (2009) Northern delta lakes as summertime CO2 absorbers within the arctic landscape. Ecosystems 12:144–157. https://doi.org/10.1007/s10021-008-9213-5
Thiemer K, Christiansen DM, Petersen NS, Mortensen SM, Christoffersen KS (2018) Reconstruction of annual growth in relation to summer temperatures and translocation of nutrients in the aquatic moss Drepanocladus trifarius from West Greenland. Polar Biol 41:2311–2321. https://doi.org/10.1007/s00300-018-2371-9
Vadeboncoeur Y, Jeppesen E, Vander Zanden MJ, Schierup H-H, Christoffersen K, Lodge DM (2003) From Greenland to green lakes: Cultural eutrophication and the loss of benthic pathways in lakes. Limnol Oceanogr 48:1408–1418. https://doi.org/10.4319/lo.2003.48.4.1408
Vincent WF, Hobbie JE, Laybourn-Parry J (2008) Introduction to the limnology of high-latitude lake and river ecosystems. In: Vincent WF, Laybourn-Parry J (eds) Polar lakes and rivers. Limnology of arctic and Antarctic Aquatic Ecosystems. Oxford University Press, Oxford, pp 1–25
WCSP (2017) World Checklist of Selected Plant Families. Facilitated by the Royal Botanic Gardens, Kew. Published on the Internet; https://apps.kew.org/wcsp/. Accessed 12 June 2017
Wauthy M, Rautio M, Christoffersen KS, Forsström L, Laurion I, Mariash H, Peura S, Vincent WF (2018) Increasing dominance of terrigenous organic matter in circumpolar freshwaters due to permafrost thaw. Limnol Oceanogr Lett. https://doi.org/10.1002/lol2.10063
Wetzel RG (2001) Limnology lake and river ecosystems, 3rd edn. Elsevier Academic Press, London
Xiong FSS, Mueller EC, Day TA (2000) Photosynthetic and respiratory acclimation and growth response of antarctic vascular plants to contrasting temperature regimes. Am J Bot 87:700–710. https://doi.org/10.2307/2656856
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lauridsen, T.L., Mønster, T., Raundrup, K. et al. Macrophyte performance in a low arctic lake: effects of temperature, light and nutrients on growth and depth distribution. Aquat Sci 82, 18 (2020). https://doi.org/10.1007/s00027-019-0692-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00027-019-0692-6