Summary
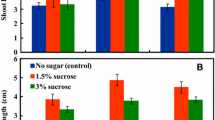
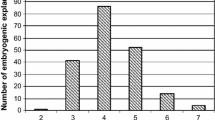
The objectives of the present research were: a) to develop an efficient soybean embryogenic regeneration system characterized by a high frequency of explant response and a large number of somatic embryos per explant; b) to evaluate the factors affecting somatic embryogenesis via somatic embryo cycling; and c) to identify the origin of somatic embryos in the system. A highly improved and efficient system for soybean somatic embryogenesis was established using somatic embryo cotyledons and somatic embryo hypocotyl/radicle explants plated on α-naphthaleneacetic acid (NAA) or 2,4-dichlorophenoxyacetic acid (2,4-D) supplemented MS basal media. The system included somatic embryo cycling between liquid and solid medium and it consistently gave rise to a much higher frequency of explant response and a larger number of embryos per responding explant than those obtained from zygotic cotyledon explant tissues. Genotype, differences were observed for response in some of the treatments with cv “Fayette” being more responsive than “J103”. Histological studies revealed that somatic embryos induced in the somatic embryo cycling system originated almost exclusively from epidermal cells on both 2,4-D and NAA inductive media. The cells of the epidermis proliferated to produce somatic embryos directly without an intervening callus phase. A single-cell origin of somatic embryos was observed in cultures on a 40 mg/liter 2,4-D treatment. A large number of responding cells in the epidermis was also observed in the 10 mg/liter NAA treatment. The single-cell origin of somatic embryos from epidermal layers of the explant tissues should facilitate development of an efficient transformation system for soybean.
Similar content being viewed by others
References
Barwale, U. B.; Kerns, H. R.; Widholm, J. M. Plant regeneration from callus cultures of several soybean genotypes via embryogenesis and organogenesis. Planta 167:473–481; 1986.
Beversdorf, W. D.; Bingham, E. T. Degrees of differentiation obtained in tissue cultures ofGlycine species. Crop Sci. 17:307–311; 1977.
Christianson, M. L.; Warnick, D. A.; Carlson, P. S. A morphologically competent soybean suspension culture. Science 222:632–634; 1983.
Dos Santos, A. V. P.; Cutter, E. G.; Davey, M. R. Origin and development of somatic embryos inMedicago sativa L. (alfalfa). Protoplasma 117:107–115; 1983.
Dudley, M. E.; Jacobs, T. W.; Long, S. R. Microscopic studies of cell divisions induced in alfalfa roots byRhizobium meliloti. Planta 171:289–301; 1987.
Finer, J. J. Apical proliferation of embryogenic tissue of soybeanGlycine max (L.) Merrill]. Plant Cell Rep. 7:238–241; 1988.
Finer, J. J.; McMullen, M. D. Transformation of soybean via particle bombardment of embryogenic suspension culture tissue. In Vitro Cell. Dev. Biol. 27P:175–182; 1991.
Finer, J. J.; Nagasawa, A. Development of an embryogenic suspension culture of soybean (Glycine max Merrill.). Plant Cell Tissue Organ Cult. 15:125–136; 1988.
Gamborg, O. L.; Davis, B. P.; Stahlhut, R. W. Somatic embryogenesis in cell cultures ofGlycine species. Plant Cell Rep. 2:209–212; 1983.
Goldberg, R. B.; Barker, S. J.; Perez-Grau, L. Regulation of gene expression during plant embryogenesis. Cell 56:149–160; 1989.
Hammatt, N.; Davey M. R. Somatic embryogenesis and plant regeneration from cultured zygotic embryos of soybean (Glycine max L. Merr.). J. Plant. Physiol. 128:219–226; 1987.
Hartweck, L. M.; Lazzeri, P. A.; Cui, D., et al. Auxin-orientation effects on somatic embryogenesis from immature soybean cotyledons. In Vitro Cell. Dev. Biol. 24:821–828; 1988.
Hepher, A.; Boulter, M. E.; Harris, N., et al. Development of a superficial meristem during somatic embryogenesis from immature cotyledons of soybean (Glycine max L.). Ann. Bot. 62:513–519; 1988.
Komatsuda, T.; Ko, S-W. Screening of soybean [Glycine max (L.) Merrill] genotypes for somatic embryo production from immature embryo. Jpn. J. Breed. 40:249–251; 1990.
Komatsuda, T.; Ohyama, K. Genotypes of high competence for somatic embryogenesis and plant regeneration in soybeanGlycine max. Theor. Appl. Genet. 75:695–700; 1988.
Lazzeri, P. A.; Hildebrand, D. F.; Collins, G. B. A procedure for plant regeneration from immature cotyledon tissue of soybean. Plant Mol. Biol. Rep. 3:160–167; 1985.
Lazzeri, P. A.; Hildebrand, D. F.; Collins, G. B. Soybean somatic embryogenesis: effects of hormones and culture manipulations. Plant Cell Tissue Organ Cult. 10:197–208; 1987.
Lazzeri, P. A.; Hildebrand, D. F.; Sunega, J. et al. Soybean somatic embryogenesis: interactions between sucrose and auxin. Plant Cell Rep. 7:517–520; 1988.
Li, B. J.; Langridge, W. H. R.; Szalay, A.A. Somatic embryogenesis and plantlet regeneration in the soybeanGlycine max. Plant Cell Rep. 4:344–347; 1985.
Lippmann, B.; Lippmann, G. Induction of somatic embryos in cotyledonary tissue of soybean,Glycine max L. Merr. Plant Cell Rep. 3:215–218; 1984.
Maheswaran, G.; Williams, E. G. Origin and development of somatic embryoids formed directly on immature embryos ofTrifolium repens in vitro. Ann. Bot. 56:619–630; 1985.
Parrott, W. A.; Dryden, G.; Vogt, S., et al. Optimization of somatic embryogenesis and embryo germination in soybean. In Vitro Cell. Dev. Biol. 24:817–820; 1988.
Parrott, W. A.; Hoffman, L. M.; Hildebrand, D. F., et al. Recovery of primary transformants of soybean. Plant Cell Rep. 7:615–617; 1989.
Parrott, W. A.; Williams, E. G.; Hildebrand, D. F., et al. Effect of genotype on somatic embryogenesis from immature cotyledons of soybean. Plant Cell Tissue Organ Cult. 16:15–21; 1989.
Phillips, G. C.; Collins, G. B. Induction and development of somatic embryos from cell suspension cultures of soybean. Plant Cell Tissue Organ Cult. 1:123–129; 1981.
Ranch, J. P.; Oglesby, L.; Zielinski, A. C. Plant regeneration from embryo-derived tissue cultures of soybeans. In Vitro Cell. Dev. Biol. 21:653–657; 1985.
Ranch, J. P.; Oglesby, L.; Zielinski, A. C. Plant regeneration from tissue culture of soybean by somatic embryogenesis. In: Vasil, I. K., ed. Cell culture and somatic cell genetics of plants, vol. 3. New York: Academic Press, Inc.; 1986:97–110.
Santos, M. A.; Torne, J. M. A comparative analysis between totipotency and growth environment conditions of the donor plants in tissue culture ofZea mays L. J. Plant Physiol. 123:299–305; 1986.
Shoemaker, R. C.; Amberger, L. A.; Palmer, R. G., et al. Effect of 2,4-dichlorophenoxyacetic acid concentration on somatic embryogenesis and heritable variation in soybean [Glycine max (L) Merr]. In Vitro Cell. Dev. Biol. 27P:84–88; 1991.
Wright, M. S.; Launis, K. L.; Novitzky, R., et al. A simple method for the recovery of multiple fertile plants from individual somatic embryos of soybean [Glycine max (L.) Merrill]. In Vitro Cell. Dev. Biol. 27P:153–157; 1991.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Liu, W., Moore, P.J. & Collins, G.B. Somatic embryogenesis in soybean via somatic embryo cycling. In Vitro Cell Dev Biol – Plant 28, 153–160 (1992). https://doi.org/10.1007/BF02823065
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02823065