Abstract
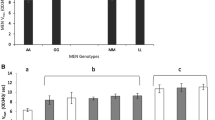
A new variant of alcohol dehydrogenase (ADH 71k) was found in a laboratory stock of Drosophila melanogaster. ADH in this stock had the same electrophoretic mobility as the F variant both on acrylamide and on agar. Activity levels were similar to the levels in F flies at temperatures between 15 and 25 C. But while ADH F enzyme is inactivated rapidly at 40 C, ADH 71k is still active. Also, ADH S is not inactivated at this temperature, but has a far lower activity per fly than ADH 71k. Genetic analysis showed that the new variant is an allele of the Adh locus.
Similar content being viewed by others
References
Ayala, F. J. (1972). Darwinian versus non-Darwinian evolution in natural populations of Drosophila. Proc. Sixth Berkeley Symp. Stat. Prob. 5211.
Bernstein, S. C., Throckmorton, L. H., and Hubby, J. L. (1973). Still more genetic variability in natural populations. Proc. Natl. Acad. Sci. 703928.
Day, T. H., and Needham, L. (1974). Properties of alcohol dehydrogenase isoenzymes in a strain of Drosophila melanogaster homozygous for the Adh-slow allele. Biochem. Genet. 11167.
Day, T. H., Hillier, P. C., and Clarke, B. (1974a). Properties of genetically polymorphic isozymes of alcohol dehydrogenase in Drosophila melanogaster. Biochem. Genet. 11141.
Day, T. H., Hillier, P. C., and Clarke, B. (1974b) The relative quantities and catalytic activities of enzymes produced by alleles at the alcohol dehydrogenase locus in Drosophila melanogaster. Biochem. Genet. 11155.
Florini, J. R., and Vestling, C. S. (1957). Graphical determination of the dissociation constants for two substrate enzyme systems. Biochim. Biophys. Acta 25575.
Frydenberg, O., and Simonsen, V. (1973). Genetics of Zoarces populations. V. Amount of protein polymorphism and degree of genic heterozygosity. Hereditas 75221.
Gibson, J. B. (1970). Enzyme fexibility in Drosophila melanogaster. Nature 227959.
Gibson, J. B. (1972). Differences in the number of molecules produced by two allelic electrophoretic enzyme variants in D. melanogaster. Experientia 28975.
Grell, E. H., Jacobson, K. B., and Murphy, J. B. (1965). Alcohol dehydrogenase in Drosophila melanogaster: Isozymes and genetic variants. Science 14980.
Jacobson, K. B., Murphy, J. B., and Hartman, F. C. (1970). Isozymes of Drosophila alcohol dehydrogenase. J. Biol. Chem. 2451075.
Kimura, M., and Ohta, T. (1971). Theoretical Aspects of Population Genetics, Princeton University Press, Princeton, N.J.
Lewontin, R. C., and Hubby, J. L. (1966). A molecular approach to the study of genetic heterozygosity in natural populations II. Amount of variation and degree of heterozygosity in natural populations of Drosophila pseudoobscura. Genetics 54595.
Lindsley, D. L., and Grell, E. H. (1968). Genetic variations of Drosophila melanogaster. Carnegie Institution, Washington, D.C.
Lineweaver, H., and Burk, D. (1934). The determination of enzyme dissociation constants. J. Am. Chem. Soc. 56658.
Singh, R. S., Hubby, J. L., and Lewontin, R. C. (1974). Molecular heterosis for heat-sensitive enzyme alleles. Proc. Natl. Acad. Sci. 711808.
Vigue, C. L., and Johnson, F. M. (1973). Isozyme variability in species of the genus Drosophila. VI. Frequency-property-environment relationships of allelic alcohol dehydrogenase in D. melanogaster. Biochem. Genet. 9213.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Thörig, G.E.W., Schoone, A.A. & Scharloo, W. Variation between electrophoretically identical alleles at the alcohol dehydrogenase locus in Drosophila melanogaster . Biochem Genet 13, 721–731 (1975). https://doi.org/10.1007/BF00484929
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00484929