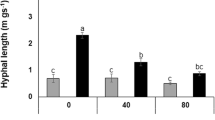
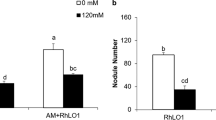
Abstract
Medicago sativa L. is the most important forage crop in arid and semi-arid areas, where increased salinity is a major factor limiting plant growth and crop productivity. The role of arbuscular mycorrhizal (AM) fungus Glomus viscosum H.T. Nicolson strain A6 in protecting alfalfa plants from salt stress, induced by sodium chloride (NaCl), was studied in two ways. Firstly, the root systems of 3-month old M. sativa plants, both mycorrhizal (AM+) and non-mycorrhizal (non-AM) (M. sativa L. var. icon), were placed in solutions of increasing salt concentrations (0, 50, 100, 150, 200 mM NaCl) to study the wilting response. G. viscosum improved the tolerance to salinity stress and the benefit was expressed in terms of the time required to reach the T4 stage in the wilting experiment. Secondly, to evaluate the ability of the Glomus-alfalfa symbiosis to tolerate salt, a pot experiment was set up in a glasshouse in which 3-month old alfalfa plants (M. sativa var. icon) were grown in a peat substratum at three salinity levels (0, 100, 150 mM NaCl). The AM symbiosis stimulated plant height, leaf area, root density, fresh and dry plant weight under saline conditions. Furthermore, proline accumulation was higher in mycorrhizal M. sativa plants than in non-mycorrhizal plants under conditions of salt stress. These and other results indicated that the micropropagated selected clone of M. sativa var. icon, when in symbiosis with G. viscosum H.T. Nicolson strain A6, exhibited better growth and physiological activities under saline conditions than non-AM plants. The AM+ plants also had lower sodium and chloride concentrations in tissues than non-AM plants.

Similar content being viewed by others
References
Abdel Latef AAH, Chaoxing H (2011) Effect of arbuscular mycorrhizal fungi on growth, mineral nutrition, antioxidant enzymes activity and fruit yield of tomato grown under salinity stress. Sci Hort 127:228–233. doi:10.1016/j.scienta.2010.09.020
Al-Karaki GN (2006) Nursery inoculation of tomato with arbuscular mycorrhizal fungi and subsequent performance under irrigation with saline water. Sci Hortic 109:1–7. doi:10.1016/j.scienta.2006.02.019
Al-Karaki GN (2000) Growth of mycorrhizal tomato and mineral acquisition under salt stress. Mycorrhiza 10:51–54. doi:10.1007/s005720000055
Al-Khaliel AS (2010) Effect of salinity stress on mycorrhizal association and growth response of peanut infected by Glomus mosseae. Plant Soil Environ 56:318–324
Anand A, Baig MJ, Mandal PK (2000) Response of alfalfa genotypes to saline water irrigation. Biol Plantarum 43:455–457. doi:10.1023/A:1026752023454
Ashraf M, McNeilly T, Bradshaw D (1986) The response to NaCl and ionic content of selected salt tolerant and normal lines of three legume forage species in sand culture. New Phytol 104:463–471
Augè RM (2001) Water relations, drought and VA mycorrhizal symbiosis. Mycorrhiza 11:3–42. doi:10.1007/s005720100097
Augè RM, Moore JL, Sylvia DM, Cho K (2004) Mycorrhizal promotion of host stomatal conductance in relation to irradiance and temperature. Mycorrhiza 14:85–92. doi:10.1007/s00572-003-0241-x
Augé RM, Toler HD, Sams CE, Nasim G (2008) Hydraulic conductance and water potential gradients in squash leaves showing mycorrhiza-induced increases in stomatal conductances. Mycorrhiza 18:115–121. doi:10.1007/s00572-008-0162-9
Azcón R, El-Atrash F (1997) Influence of arbuscular mycorrhizae and phosphorus fertilization on growth, nodulation and N2 fixation (15N) in Medicago sativa at four salinity levels. Biol Fert Soils 24:81–86. doi:10.1007/BF01420225
Baker DE, Gorsline GW, Smith CG, Thomas WI, Grube WE, Raglan JL (1964) Techniques for rapid analysis of corn leaves for eleven elements. Agron J 56:133–136
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water stress studies. Plant Soil 39:205–207. doi:10.1007/BF00018060
Biermann B, Linderman RG (1981) Quantifying vesicular-arbuscular mycorrhizae: A proposed method towards standardization. New Phytol 87:63–67
Borde M, Dudhane M, Jite P (2011) Growth photosynthetic activity and antioxidant responses of mycorrhizal and non-mycorrhizal bajra (Pennisetum glaucum) crop under salinity stress condition. Crop Prot 30:265–271. doi:10.1016/j.cropro.2010.12.010
Borde M, Dudhane M, Jite P (2010) AM Fungi influences the photosynthetic activity, growth and antioxidant enzymes in Allium sativum L. under salinity condition. Not Sci Biol 2:64–71
Busquets M, Calvet C, Camprubí A, Estaún V (2010) Differential effects of two species of arbuscular mycorrhiza on the growth and water relations of Spartium junceum and Anthyllis cytisoides. Symbiosis 52:95–101. doi:10.1007/s13199-010-0097-8
Cantrell IC, Linderman RG (2001) Preinoculation of lettuce and onion with VA mycorrhizal fungi reduces deleterious effects of soil salinity. Plant Soil 233:269–281. doi:10.1023/A:1010564013601
Cerda A, Pardines J, Botella MA, Martinez V (1995) Effect of potassium on growth, water relations, and the inorganic and organic solute contents for two maize cultivars grown under saline conditions. J Plant Nutr 18:839–851. doi:0.1080/01904169509364942
Chang SX, Robison DJ (2003) Nondestructive and rapid estimation of hardwood foliar nitrogen status using the SPAD-502 chlorophyll meter. Forest Ecol Manag 181:331–338. doi:10.1016/S0378-1127(03)00004-5
Cole DF, Dobrenz AK (1970) Stomatal density of alfalfa (Medicago sativa L.). Crop Sci 10:61–63
Colla G, Rouphael Y, Cardarelli M, Tullio M, Rivera CM, Rea E (2008) Alleviation of salt stress by arbuscular mycorrhizal in zucchini plants grown at low and high phosphorus concentration. Biol Fert Soils 44:501–509. doi:10.1007/s00374-007-0232-8
Dalpé Y, Monreal M (2004) Arbuscular mycorrhiza inoculums to support sustainable cropping systems. Crop Management 10:1094–1104. doi:10.1094/CM-2004-0301-09-RV
Dissing-Nielsen J (1990) The effect of VAM on growth and uptake of nutrients in lucerne. Agric Ecosyst Environ 29:99–102. doi:10.1016/0167-8809(90)90261-B
Djanaguiraman M, Sheeba JA, Shanker AK, Durga Devi D, Bangarusamy U (2006) Rice can acclimate to lethal level of salinity by pretreatment with sublethal level of salinity through osmotic adjustment. Plant Soil 284:363–373
Dudhane M, Borde M, Jite P (2011) Effect of arbuscular mycorrhizal fungi on growth and antioxidant Activity in Gmelina arborea Roxb. under salt stress condition. Not Sci Biol 3:71–78
Esfahani M, Ali Abbasi HR, Rabiei B, Kavousi M (2008) Improvement of nitrogen management in rice paddy fields using chlorophyll meter (SPAD). Paddy Water Environ 6:181–188. doi:10.1007/s10333-007-0094-6
Evelin H, Kapoor R, Giri B (2009) Arbuscular mycorrhizal fungi in alleviation of salt stress: A review. Ann Bot 104:1263–1280. doi:10.1093/aob/mcp251
Gáborčík N (2003) Relationship between contents of chlorophyll (a+b) (SPAD values) and nitrogen of some temperate grasses. Photosynthetica 41:285–287. doi:10.1023/B:PHOT.0000011963.43628.15
Giri B, Kapoor R, Mukerji KG (2007) Improved tolerance of Acacia nilotica to salt stress by arbuscular mycorrhiza, Glomus fasciculatum may be partly related to elevated K/Na ratios in root and shoot tissues. Microbial Ecol 54:753–760. doi:10.1007/s00248-007-9239-9
Giridarakumar S, Matta Reddy A, Sudhakar C (2003) NaCl effects on proline metabolism in two high yielding genotypes of mulberry (Morus alba L.) with contrasting salt tolerance. Plant Science 165:1245–1251. doi:10.1016/S0168-9452(03)00332-7
Goicoechea N, Szalai G, Antolin MC, Sanchez-Diaz M, Paldi E (1998) Influence of arbuscular mycorrhizae and Rhizobium on free polyamines and proline levels in water-stressed alfalfa. J Plant Physiol 153:706–711
Goicoechea N, Antolín MC, Sánchez-Díaz M (1997) Influence of arbuscular mycorrhizae and Rhizobium on nutrient content and water relations in drought stressed alfalfa. Plant Soil 192:261–268. doi:10.1023/A:1004216225159
Graham JH, Syvertsen JP (1985) Host determinants of mycorrhizal dependency of citrus rootstock seedlings. New Phytol 101:667–676. doi:10.1111/j.1469-8137.1985.tb02872.x
Hatimi A (1999) Effect of salinity on the association between root symbionts and Acacia cyanophylla Lind: growth and nutrition. Plant Soil 216:93–101. doi:10.1023/A:1004745707277
Heuer B (1999) Osmoregulatory role of proline in plants exposed to environmental stresses. In: Pessarakli M (ed) Handbook of Plant and Crop Stress, 2nd edn. CRC, pp 675–695. doi: 10.1201/9780824746728.ch32
Huang Z, Zou Z, He C, He Z, Zhang Z, Li J (2011) Physiological and photosynthetic responses of melon (Cucumis melo L.) seedlings to three Glomus species under water deficit. Plant Soil 339:391–399. doi: 10.1007/s11104-010-0591-z
Jaleel CA, Sankar B, Sridharan R, Panneerselvam R (2008) Soil salinity alters growth, chlorophyll content, and secondary metabolite accumulation in Catharanthus roseus. Turk J Biol 32:79–83
Jarrell WM, Beverly RB (1981) The dilution effect in plant nutrition studies. Adv Agron 34:197–224. doi:10.1016/S0065-2113(08)60887-1
Kang Y, Han Y, Torres-Jerez I, Wang M, Tang Y, Monteros M, Udvardi M (2011) System responses to long-term drought and re-watering of two contrasting alfalfa varieties. Plant J 68:71–889. doi:10.1111/j.1365-313X.2011.04738.x
Kao WY, Tsai TT, Tsai HC, Shih CN (2006) Response of three Glycine species to salt stress. Environ Exp Bot 56:120–125. doi:10.1016/j.envexpbot.2005.01.009
Karimi G, Ghorbanli M, Heidari H, Khavari Nejad RA, Assareh MH (2005) The effects of NaCl on growth, water relations, osmolytes and ion content in Kochia prostrata. Biol Plantarum 49:301–304. doi:10.1007/s10535-005-1304-y
Kaya C, Tuna AL, Ashraf M, Altunlu H (2007) Improved salt tolerance of melon (Cucumis melo L.) by the addition of proline and potassium nitrate. Environ Exp Bot 60:397–403. doi:10.1016/j.envexpbot.2006.12.008
Khalil HA, Eissa AM, El-Shazly SM, Aboul Nasr AM (2011) Improved growth of salinity-stressed citrus after inoculation with mycorrhizal fungi. Sci Hortic 130:624–632. doi:10.1016/j.scienta.2011.08.019
Khan MG, Silberbusush M, Lips SH (1997) Response of alfalfa to potassium, calcium, and nitrogen under stress induced by sodium chloride. Biol Plantarum 40:251–259. doi:10.1023/A:1001072704686
Kramer PJ, Boyer JS (1995) Water Relation of Plants and Soils. Academic Press, San Diego, CA, USA. http://dspace.udel.edu:8080/dspace/handle/19716/2830
Krishna H, Singh SK, Sharma RR, Khawale RN, Grover M, Patel VB (2005) Biochemical changes in micropropagated grape (Vitis vinifera L.) plantlets due to arbuscular-mycorrhizal fungi (AMF) inoculation during ex vitro acclimatization. Sci Hort 106:554–567. doi:10.1016/J.scienta.2005.05.009
Kumar A, Sharma S, Mishra S (2010) Influence of arbuscular mycorrhizal (AM) fungi and salinity on seedling growth, solute accumulation, and mycorrhizal dependency of Jatropha curcas L. J Plant Growth Regul 29:297–306. doi:10.1007/s00344-009-9136-1
Larose G, Chênevert R, Moutoglis P, Gagné S, Piché Y, Vierheilig H (2002) Flavonoid levels in roots of Medicago sativa are modulated by the developmental stage of the symbiosis and the root colonizing arbuscular mycorrhizal fungus. J Plant Physiol 159:1329–1339. doi:10.1078/0176-1617-00896
Maas EV, Hoffman GJ (1977) Crop salt tolerance – current assessment. J Irrig Drain Div Am Soc Civ Eng 103:115–134
Maggio A, Quaglietta Chiarandà F, Cefariello R, Fagnano M (2009) Responses to ozone pollution of alfalfa exposed to increasing salinity levels. Environ Pollut 157:1445–1452. doi:10.1016/j.envpol.2008.09.013
Meloni DA, Gulotta MR, Martínez CA, Oliva MA (2004) The effects of salt stress on growth, nitrate reduction and proline and glycinebetaine accumulation in Prosopis alba. Braz J Plant Physiol 16:1–11. doi:10.1590/S1677-04202004000100006
Miller LRG Jr (1981) Simultaneous Statistical Inference. Springer, New York
Netto AT, Campostrini E, Oliveira JG, Bressan-Smith RE (2005) Photosynthetic pigments, nitrose, chlorophyll a fluorescence and SPAD-502 readings in coffee leaves. Sci Hort 104:199–209. doi:10.1016/j.scienta.2004.08.013
Percival GC, Keary IP, Noviss K (2008) The potential of a chlorophyll content SPAD meter to quantify nutrient stress in foliar tissue of Sycamore (Acer pseudoplatanus), English Oak (Quercus robur), and European Beech (Fagus sylvatica). Arboric Urb For 34:89–100
Phillips JM, Hayman DS (1970) Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans Br Mycol Soc 55:158–161
Plenchette C, Fortin JA, Furlan V (1983) Growth responses of several plant species to mycorrhizae in a soil of moderate P fertility. I. Mycorrhizal dependency under field conditions. Plant Soil 70:199–209. doi:10.1007/BF02374780
Porcel R, Aroca R, Ruiz-Lozano JM (2012) Salinity stress alleviation using arbuscular mycorrhizal fungi. A review. Agron Sustain Dev 32:181–200. doi:10.1007/s13593-011-0029-x
Porcel R, Ruiz-Lozano JM (2004) Arbuscular mycorrhizal influence on leaf water potential, solute accumulation, and oxidative stress in soybean plants subjected to drought stress. J Exp Bot 55:1743–1750. doi:10.1093/jxb/erh188
Rabie GH, Almadini AM (2005) Role of bioinoculants in development of salt-tolerance of Vicia faba plants under salinity stress. Afr J Biotechnol 4:210–222
Reddy MP, Vora AB (1985) Effect of salinity on protein metabolism in bajra leaves. Indian J Plant Physiol 28:190–195
Rosendhal CN, Rosendhal S (1991) Influence of vesicular-arbuscular mycorrhizal fungi (Glomus spp.) on the response of cucumber. Environ Exp Bot 31:313–318. doi:10.1016/0098-8472(91)90055-S
Ruiz-Lozano JM, Azcón R, Gomez M (1995) Effects of arbuscular-mycorrhizal Glomus species on drought tolerance: physiological and nutritional plants responses. Appl Environ Microb 61:456–460
Ruiz-Lozano JM, Azcón R, Gomez M (1996) Alleviation of salt stress by arbuscular mycorrhizal Glomus in Lactuca sativa plants. Physiol Plantarum 98:767–772. doi:10.1111/j.1399-3054.1996.tb06683.x
Saif SR (1987) Growth responses of tropical forage plant species to vesicular-arbuscular mycorrhiza. I. Growth mineral uptake and mycorrhizal dependency. Plant Soil 97:25–35. doi:10.1007/s00374-002-0457-5
Sharifi M, Ghorbani M, Ebrahimzadeh H (2007) Improved growth of salinity-stressed soybean after inoculation with salt pre-treated mycorrhizal fungi. J Plant Physiol 164:1144–1151. doi:10.1016/j.jplph.2006.06.016
Sheng M, Tang M, Zhang F, Huang Y (2011) Influence of arbuscular mycorrhiza on organic solutes in maize leaves under salt stress. Mycorrhiza 21:423–430. doi:10.1007/s00572-010-0353-z
Sheng M, Tang M, Chen H, Yang B, Zhang F, Huang Y (2008) Influence of arbuscular mycorrhizae on photosynthesis and water status of maize plants under salt stress. Mycorrhiza 18:287–296. doi:10.1007/s00572-008-0180-7
Smith BR, Fisher PR, Argo WR (2004) Growth and pigment content of container-grown Impatients and Petunia in relation to root substrata pH and applied micronutrient concentration. HortSci 39:141–1425
Sonneveld C, Van Den Ende J, Van Dijk PA (1974) Analysis of growing media by means of a 1:1/2 volume extract. Commun Soil Sci Plant Anal 5:183–202
Srivastava D, Mukerji KG (1995) Field response of mycorrhizal and nonmycorrhizal Medicago sativa var. local in the F1 generation. Mycorrhiza 5:219–222. doi:10.1007/BF00203341
Strain HH, Svec WA (1966) Extraction, separation, estimation and isolation of chlorophylls. In: Vernon LP, Seely GR (eds) The Chlorophylls. Academic, NY, pp 21–66
Talaat NB, Shawky BT (2011) Influence of arbuscular mycorrhizae on yield, nutrients, organic solutes, and antioxidant enzymes of two wheat cultivars under salt stress. J Plant Nutr Soil Sci 174:283–291. doi:10.1002/jpln.201000051
Tennant D (1975) A test of a modified line intersect method of estimating root length. J Ecol 63:995–1001
Trouvelot A, Kouch J, Gianinazzi-Pearson V (1986) Mesure du taux de mycorhization VA d’un système radiculaire: Recherche of method d’estimation ayant une signification fonctionelle. Les Mycorhizes: Phisiologie and Genetique. Ier Seminaire Dijon, Ed. INRA, Paris, pp 217–221
Watson ME, Isaac RA (1990) Analitical instruments for soil and plant analysis. In: Westerman RL (ed) Soil Testing and Plant Analysis, 3rd edn. SSSA Book Series 3, Soil Science Society of America, Madison, W.I, pp 691–740
Wu Q, Xia R (2006) Effects of arbuscular mycorrhizal fungi on leaf solutes and root absorption areas of trifoliate orange seedlings under water stress conditions. Front For China 3:312–317. doi:10.1007/s11461-006-0035-3
Wu J, Wang D, Rosen CJ, Bauer ME (2007) Comparison of petiole nitrate concentrations, SPAD chlorophyll readings, and QuickBird satellite imagery in detecting nitrogen status of potato canopies. Field Crops Res 101:96–103. doi:10.1016/j.fcr.2006.09.014
Yano-Melo AM, Saggin OJ, Maia LC (2003) Tolerance of mycorrhized banana (Musa sp. cv. Pacovan) plantlets to saline stress. Plant Soil Environ 95:343–348. doi:10.1016/S0167-8809(02)00044-0
Zhang Q, Xu L, Tang J, Bai M, Chen X (2011) Arbuscular mycorrhizal mediation of biomass-density relationship of Medicago sativa L. under two water conditions in a field experiment. Mycorrhiza 21:269–277. doi:10.1007/s00572-010-0331-5
Zhu XC, Song FB, Liu SQ, Liu TD (2011) Effects of arbuscular mycorrhizal fungus on photosynthesis and water status of maize under high temperature stress. Plant Soil 346:189–199. doi:10.1007/s11104-011-0809-8
Zuccarini P (2007) Mycorrhizal infection ameliorates chlorophyll content and nutrient uptake of lettuce exposed to saline irrigation. Plant Soil Environ 7:283–289
Acknowledgments
The authors are grateful to Ms. Sara Donahue, English Language Professor in the Faculty of Foreign Languages and Literatures, University of Bari, for her invaluable language assistance in the preparation of this manuscript. Thanks are due to Antonio Corleto, Professor in the Department of Plant Production Sciences, University of Bari, for his precious advice and for providing Medicago sativa seeds. The authors also wish to thank Prof. Manuela Giovannetti of the Department of Crop Plants, University of Pisa (Italy) for providing the arbuscular-mycorrhizal inoculum.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Campanelli, A., Ruta, C., De Mastro, G. et al. The role of arbuscular mycorrhizal fungi in alleviating salt stress in Medicago sativa L. var. icon. Symbiosis 59, 65–76 (2013). https://doi.org/10.1007/s13199-012-0191-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13199-012-0191-1