Abstract
Spillover of fish from marine reserves to adjacent harvested waters may be mediated by density-independent movement, density-dependent movement, or both. If dispersal is by random movement, populations within the reserve must be regulated by density-dependent population growth (DDG). Density-dependent movement (DDM) can also regulate the population if accelerated emigration from a reserve to the surrounding fishing grounds leads to substantially increased mortality. Using spatially explicit models, we show that stock per unit area is bounded for DDG and increases with size for DDM. With DDG, spillover rate per unit area of reserve is maximized with reserves around 50% larger in linear dimension than the minimum size for population persistence. With DDM, spillover per unit area of reserve increases with reserve size. The results highlight the need for the mechanism of population regulation to be incorporated into theoretical and empirical investigations of marine reserve ecology.




Similar content being viewed by others
References
Abesamis RA, Russ GR (2005) Density-dependent spillover from a marine reserve: long-term evidence. Ecol Appl 15:1798–1812
Alcala AC, Russ GR (1990) A direct test of the effects of protective management on abundance and yield of tropical marine resources. J Cons 47:40–47
Anderson TW (2001) Predator responses, prey refuges, and density-dependent mortality of a marine fish. Ecology 82:245–257
Armsworth PR, Roughgarden JE (2005) The impact of directed versus random movement on population dynamics and biodiversity patterns. Am Nat 165:449–465
Attwood CG, Bennett BA (1994) Variation in dispersal of galjoen (Coracinus capensis) (Teleostei: Coracinidae) from a marine reserve. Can J Fish Aquat Sci 51:1247–1257
Baskett ML, Micheli F, Levin SA (2007) Designing marine reserves for interacting species: insights from theory. Biol Conserv 137:163–179
Béné C, Tewfik A (2003) Biological evaluation of marine protected area: evidence of crowding effect on a protected population of queen conch in the Caribbean. Mar Ecol 24:45–58
Botsford LW, Hastings A, Gaines SD (2001) Dependence of sustainability on the configuration of marine reserves and larval dispersal distance. Ecol Lett 4:144–150
Cantrell RS, Cosner C (2003) Spatial ecology via reaction–diffusion equations. Wiley series in mathematical and computational biology. Wiley, Chichester, UK
Chapman MR, Kramer DL (2000) Movements of fishes within and among fringing coral reefs in Barbados. Environ Biol Fish 57:11–24
Conrad JM (1999) The bioeconomics of marine sanctuaries. J Bioecon 1:205–217
Fisher RA (1937) The wave of advance of advantageous genes. Ann Eugenics 7:355–369
Fryxell JM, Lynn DH, Chris PJ (2006) Harvest reserves reduce extinction risk in an experimental microcosm. Ecol Lett 9:1025–1031
Gaggiotti OE, Jones F, Lee WM, Amos W, Harwood J, Nichols RA (2002) Patterns of colonization in a metapopulation of grey seals. Nature 416:424–427
Gaines SD, Gaylord B, Largier JL (2003) Avoiding current oversights in marine reserve design. Ecol Appl 13:S32–S46
Gitschlag GR (1986) Movement of pink shrimp Penaeus duorarum in relation to the Tortugas sanctuary gulf of Mexico. N Am J Fish Manage 6:328–338
Grosholz ED (1996) Contrasting rates of spread for introduced species in terrestrial and marine systems. Ecology 77:1680–1686
Gurney WSC, Nisbet RM (1975) The regulation of inhomogeneous populations. J Theor Biol 52:441–458
Gurney WSC, Nisbet RM (1998) Ecological dynamics. Oxford University Press, New York
Halpern BS, Warner RR (2002) Marine reserves have rapid and lasting effects. Ecol Lett 5:361–366
Halpern BS, Warner RR (2003) Matching marine reserve design to reserve objectives. Proc R Soc Lond B Biol Sci 270:1871–1878
Halpern BS, Regan HM, Possingham HP, McCarthy MA (2006) Accounting for uncertainty in marine reserve design. Ecol Lett 9:2–11
Hannesson R (1998) Marine reserves: what would they accomplish? Mar Resour Econ 13:159–170
Hixon MA (1998) Population dynamics of coral-reef fishes: controversial concepts and hypotheses. Aust J Ecol 23:192–201
Hixon MA, Carr MH (1997) Synergistic predation, density dependence, and population regulation in marine fish. Science 277:946–949
Holbrook SJ, Schmitt RJ (2002) Competition for shelter space causes density-dependent predation mortality in damselfishes. Ecology 83:2855–2868
Holmes EE, Lewis MA, Banks JE, Veit RR (1994) Partial differential equations in ecology: spatial interactions and population dynamics. Ecology 75:17–29
Johnson DR, Funicelli NA, Bohnsack JA (1999) Effectiveness of an existing estuarine no-take fish sanctuary within the Kennedy Space Center, Florida. N Am J Fish Manage 19:436–453
Kaplan DM, Botsford LW (2005) Effects of variability in spacing of coastal marine reserves on fisheries yield and sustainability. Can J Fish Aquat Sci 62:905–912
Kelly S, Scott D, MacDiarmid AB (2002) The value of a spillover fishery for spiny lobsters around a marine reserve in Northern New Zealand. Coast Manage 30:153–166
Kellner JB, Tetreault I, Gaines SD, Nisbet RM (2007) Fishing-the-line near marine reserves in single and multi-species fisheries. Ecol Appl 17:1039–1054
Kierstead H, Slobodkin LB (1953) The size of water masses containing plankton blooms. J Mar Res 12:141–147
Koslow JA, Bell J, Virtue P, Smith DC (1995) Fecundity and its variability in Orange Roughy: effects of population density, condition, egg size, and senescence. J Fish Biol 47:1063–1080
Kot M (2001) Elements of mathematical ecology. Cambridge University Press, New York
Lima SL, Zollner PA (1996) Towards a behavioral ecology of ecological landscapes. Trends Ecol Evol 11:131–135
Lizaso JLS, Goni R, Renones O, Charton G, Galzin R, Bayle JT, Jerez PS, Ruzafa AP, Ramos AA (2000) Density dependence in marine protected populations: a review. Environ Conserv 27:144–158
McClanahan TR, Kaunda-Arara B (1996) Fishery recovery in a coral-reef marine park and its effect on the adjacent fishery. Conserv Biol 10:1187–1199
McClanahan TR, Mangi S (2000) Spillover of exploitable fishes from a marine park and its effect on the adjacent fishery. Ecol Appl 10:1792–1805
Murawski SA, Brown R, Lai HL, Rago PJ, Hendrickson L (2000) Large-scale closed areas as a fishery-management tool in temperate marine systems: the Georges Bank experience. Bull Mar Sci 66:775–798
Murray JD, Sperb RP (1983) Minimum domains for spatial patterns in a class of reaction–diffusion equations. J Math Biol 18:169–184
Murray SN, Ambrose RF, Bohnsack JA, Botsford LW, Carr MH, Davis GE, Dayton PK, Gotshall D, Gunderson DR, Hixon MA, Lubchenco J, Mangel M, MacCall A, McArdle DA, Ogden JC, Roughgarden J, Starr RM, Tegner MJ, Yoklavich MM (1999) No-take reserve networks: sustaining fishery populations and marine ecosystems. Fisheries 24:11–25
National Research Council (2001) Marine protected areas: tools for sustaining ocean ecosystems. National Academy, Washington, DC
Neubert MG (2003) Marine reserves and optimal harvesting. Ecol Lett 6:843–849
Nisbet RM, Gurney WSC (1982) Modelling fluctuating populations. Wiley, Chichester, New York
Okubo A (1980) Diffusion and ecological problems: mathematical models. Springer, New York
Okubo A, Levin SA (2001) Diffusion and ecological problems: modern perspectives, 2nd edn. Springer, New York
Pollock DE (1993) Recruitment overfishing and resilience in spiny lobster populations. ICES J Mar Sci 50:9–14
Polunin NVC, Roberts CM (1993) Greater biomass and value of target coral-reef fishes in 2 small Caribbean marine reserves. Mar Ecol Prog Ser 100:167–176
Rakitin A, Kramer DL (1996) Effect of a marine reserve on the distribution of coral reef fishes in Barbados. Mar Ecol Prog Ser 131:97–113
Ramos-Espla AA, McNeill SE (1994) The status of marine conservation in Spain. Ocean Coast Manag 24:125–138
Roberts CM, Branch G, Bustamante RH, Castilla JC, Dugan J, Halpern BS, Lafferty KD, Leslie H, Lubchenco J, McArdle D, Ruckelshaus M, Warner RR (2003) Application of ecological criteria in selecting marine reserves and developing reserve networks. Ecol Appl 13:S215–S228
Rose KA, Cowan JH Jr, Winemiller KO, Myers RA, Hilborn R (2001) Compensatory density dependence in fish populations: importance, controversy, understanding and prognosis. Fish Fish 2:293–327
Rosenberg R, Nilsson HC, Hollertz K, Hellman B (1997) Density-dependent migration in an Amphiura filiformis (Amphiuridae, Echinodermata) infaunal population. Mar Ecol Prog Ser 159:121–131
Sanchirico JN, Wilen JE (2001) A bioeconomic model of marine reserve creation. J Environ Econ Manage 42:257–276
Shepherd SA (1990) Studies on Southern Australian Abalone (Genus Haliotis). 12. Long-term recruitment and mortality dynamics of an unfished population. Aust J Mar Fresh Res 41:475–492
Shorthouse B (1990) The Great Barrier Reef Marine Park: how does it work for fishermen? Australian Fisheries, December, 16–17
Skellam JG (1951) Random dispersal in theoretical populations. Biometrika 38:196–218
Sparrevohn CR, Nielsen A, Stottrup JG (2002) Diffusion of fish from a single release point. Can J Fish Aquat Sci 59:844–853
St Mary CM, Osenberg CW, Frazer TK, Lindberg WJ (2000) Stage structure, density dependence and the efficacy of marine reserves. Bull Mar Sci 66:675–690
Travis JMJ, Murrell DJ, Dytham C (1999) The evolution of density-dependent dispersal. Proc R Soc Lond B Biol Sci 266:1837–1842
Turchin P (1991) Translating foraging movements in heterogeneous environments into the spatial-distribution of foragers. Ecology 72:1253–1266
Turchin P (1998) Quantitative analysis of movement: measuring and modeling population redistribution in animals and plants. Sinauer, Sunderland, MA
Yamasaki A, Kuwahara A (1990) Preserved area to effect recovery of over-fished Zuwai crab stocks off Kyoto Prefecture. In: Proceedings of the International Symposium on King and Tanner Crabs. Alaska Sea Grant College Program, University of Alaska, Fairbanks, Anchorage, AK, pp 575–586
Zabel RW (2002) Using “travel time” data to characterize the behavior of migrating animals. Am Nat 159:372–387
Acknowledgments
Support was provided by the Coastal Toxicology Lead Campus Component of the University of California Toxic Substances Research and Teaching Program, the University of California – Santa Barbara, US NSF (grant DEB01-08450), Santa Barbara Coastal Long Term Ecological Research, the Pew Charitable Trusts, the Andrew W. Mellon Foundation, and the Partnership for Interdisciplinary Studies of Coastal Oceans (PISCO) funded primarily by the Gordon and Betty Moore Foundation and the David and Lucile Packard Foundation. This is PISCO contribution number 282. The authors thank Bruce Kendall, Bob Warner, Shaun Belward, and Elizaveta Pachepsky for helpful discussions.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
This appendix contains analyses to support the findings in Table 1.
Recasting equations in non-dimensional form
Reduction in the DDG model to a non-dimensional form is described by Gurney and Nisbet (1998, pp 53–54). We define base units of length, time, and population density by
and define dimensionless variables
The dynamics of the DDG model within the reserve can now be expressed as
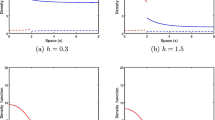
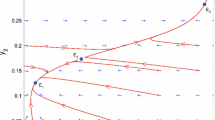
with \(\tilde n = 0\) at the reserve boundary. The entire behavior of the system is now determined by a single parameter group \(\tilde L = {L \mathord{\left/ {\vphantom {L {L_{\text{s}} }}} \right. \kern-\nulldelimiterspace} {L_{\text{s}} }}\). Because the length scale L s is proportional to the critical reserve size (see Eq. 7), this implies that the dynamics are controlled completely by the ratio \({L \mathord{\left/ {\vphantom {L {L_{\text{c}} }}} \right. \kern-\nulldelimiterspace} {L_{\text{c}} }}\) used as independent variables in Figs. 2 and 3. The results in Table 1 regarding the dependence of system behavior on parameters other than L (i.e., r, D 0, K) are obtained by using Eqs. 8 and 9 to convert properties of the dimensionless equations back to the original unscaled variables.
A similar rescaling is used for the DDM model, the only difference being that the base unit of population density is \(n_{\text{s}} = {{D_0 } \mathord{\left/ {\vphantom {{D_0 } {D_n }}} \right. \kern-\nulldelimiterspace} {D_n }}\). The DDM model dynamics within the reserve are then described by the equation
Analysis of DDG model
The steady states of this model have been extensively studied in 1D (Kot 2001, pp 294–301). For any model whose steady states are defined by an equation of the form
with absorbing boundary conditions at \( \pm {{\tilde L} \mathord{\left/ {\vphantom {{\tilde L} 2}} \right. \kern-\nulldelimiterspace} 2}\), there is a first integral
and \(\tilde n_{\max } \) is the maximum population density (attained at the center of the reserve), and v is a dummy integration variable.
For the DDM model, \(f\left( {\tilde n} \right) = \tilde n\left( {1 - \tilde n} \right)\), so \(F\left( {\tilde n} \right) = \frac{1}{2}\tilde n^2 - \frac{1}{3}\tilde n^3 \). At the reserve boundaries, \(F\left( {\tilde n} \right) = 0\), and the total (scaled) spillover rate from the two edges of the reserve is given by
which has a finite upper bound as \(\tilde n_{\max } \leqslant 1\). For small reserves (L close to L c ), the flux is easily shown to be directly proportional to L − L c . Thus, as L increases, spillover rate per unit length initially rises, then falls asymptotically to zero as L→∞.
For the 2D circular system considered in the paper, we know of no first integral analogous to Eq. 13. In this case, it follows from previous work by Murray and Sperb (1983, p 180) that Eq. 14 is replaced by an inequality
For small reserves (L close to L c), the flux is easily shown to be directly proportional to \(\left( {L - L_{\text{c}} } \right)^2 \). Thus, spillover rate per unit area will have a unimodal form analogous to the 1D situation. This was demonstrated numerically in Fig. 3c.
Analysis of the DDM model
A change of variable transforms the scaled DDM in 1D to the form in Eq. 12. If we define
then, after some algebra, we obtain
The maximum value of u (attained at the center of the reserve) is related to the scaled size by (cf. Kot 2001, Eq. 17.17)
In contrast with the DDG case, there is no upper bound to u max for the DDM model, and with some algebra, it can be shown that as L→∞, \(u_{\max } \propto L^4 \). To see this, first note from Eqs. 17 and 18 that
Then, substitute \(z = {u \mathord{\left/ {\vphantom {u {u_{\max } }}} \right. \kern-\nulldelimiterspace} {u_{\max } }}\) in Eq. 18 to obtain
The last integral does not involve u max, so the result follows.
Equation 16 then implies that the maximum scaled population density \(\tilde n_{\max } \propto L^2 \), and hence total population size (TPS) is proportional to L 3. In this model, spillover rate is proportional to TPS, so spillover rate per unit length increases as the reserve increases, and there is no optimum reserve size that maximizes spillover.
We are unable to construct a proof of the analogous result for 2D because of the lack of a simple first integral (the use of the Murray–Sperb inequality does not help here), but the very strong 1D result makes very plausible our numerically-based conclusion that there is no optimum reserve size for DDM.
Rights and permissions
About this article
Cite this article
Kellner, J.B., Nisbet, R.M. & Gaines, S.D. Spillover from marine reserves related to mechanisms of population regulation. Theor Ecol 1, 117–127 (2008). https://doi.org/10.1007/s12080-008-0012-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12080-008-0012-6