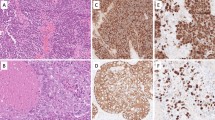
Abstract
Neuroendocrine tumors of the lung are classified into low-grade typical and intermediate-grade atypical carcinoids, and high-grade poorly differentiated neuroendocrine carcinomas of the large and small cell types. This scheme is strongly predictive of patients’ prognosis but relies on few and scarcely reproducible pathological parameters (namely mitotic count and assessment of the presence of necrosis), which have been demonstrated to affect the inter-observer agreement of the classification. Moreover, tumor and nodal staging schemes are not specific for lung carcinoids, at variance with neuroendocrine tumors of the gastro-entero-pancreatic system, despite these tumors have specific features that strongly differ from conventional lung cancer. Finally, there is no grading for lung neuroendocrine neoplasms and prognostication, as well as the definition of treatment modalities and clinical strategies, which are based on tumor histotypes, only. However, literature data indicate that the evaluation of Ki-67 proliferation index may be a reliable and useful tool to determine the biological and clinical behavior of neuroendocrine tumors, with special reference to carcinoids, both in pre-operative and surgical samples.
Similar content being viewed by others
References
W.D. Travis, E. Brambilla, H.K. Muller-Hermelink, C.C. Harris, Pathology and genetics of tumours of the lung, pleura, thymus and heart (IARC Press, Lyon, 2004)
G. Pelosi, J. Rodriguez, G. Viale, J. Rosai, Typical and atypical pulmonary carcinoid tumor overdiagnosed as small-cell carcinoma on biopsy specimens: a major pitfall in the management of lung cancer patients. Am. J. Surg. Pathol. 29, 179–187 (2005)
W.D. Travis, D.J. Giroux, K. Chansky, J. Crowley, H. Asamura, E. Brambilla, J. Jett, C. Kennedy, R. Rami-Porta, V.W. Rusch, P. Goldstraw, International Staging Committee and Participating Institutions: the IASLC Lung Cancer Staging Project: proposals for the inclusion of broncho-pulmonary carcinoid tumors in the forthcoming (seventh) edition of the TNM Classification for Lung Cancer. J. Thorac. Oncol. 3, 1213–1223 (2008)
P. Ferolla, N. Daddi, M. Urbani, A. Semeraro, R. Ribacchi, P. Giovenali, S. Ascani, V. De Angelis, L. Crinò, F. Puma, G. Daddi, Regional multidisciplinary group for the diagnosis and treatment of neuroendocrine tumors, CRO, Umbria region cancer network, Italy: tumorlets, multicentric carcinoids, lymph-nodal metastases, and long-term behavior in bronchial carcinoids. J. Thorac. Oncol. 4, 383–387 (2009)
A.A. Nassar, D.E. Jaroszewski, R.A. Helmers, T.V. Colby, B.M. Patel, F. Mookadam, Diffuse idiopathic pulmonary neuroendocrine cell hyperplasia: a systematic overview. Am. J. Respir. Crit. Care Med. 184, 8–16 (2011)
S.M. Liu, H.H. Wu, C.J. Wu, C.L. Kuo, L.R. Mo, Adrenocorticotropin-producing pulmonary tumorlets with lymph node metastasis. Pathol. Int. 53, 883–886 (2003)
B.G. Skov, M. Krasnik, S. Lantuejoul, T. Skov, E. Brambilla, Reclassification of neuroendocrine tumors improves the separation of carcinoids and the prediction of survival. J. Thorac. Oncol. 3, 1410–1415 (2008)
D.R. Swarts, R.J. van Suylen, M.A. den Bakker, M.F. van Oosterhout, F.B. Thunnissen, M. Volante, A.M. Dingemans, M.R. Scheltinga, G.P. Bootsma, H.M. Pouwels, B.E. van den Borne, F.C. Ramaekers, E.J. Speel, Interobserver variability for the WHO classification of pulmonary carcinoids. Am. J. Surg. Pathol. 38, 1429–1436 (2014)
M.A. den Bakker, S. Willemsen, K. Grünberg, L.A. Noorduijn, M.F. van Oosterhout, R.J. van Suylen, W. Timens, B. Vrugt, A. Wiersma-van Tilburg, F.B. Thunnissen, Small cell carcinoma of the lung and large cell neuroendocrine carcinoma interobserver variability. Histopathology 56, 356–363 (2010)
A.M. Marchevsky, A.A. Gal, S. Shah, M.N. Koss, Morphometry confirms the presence of considerable nuclear size overlap between “small cells” and “large cells” in high-grade pulmonary neuroendocrine neoplasms. Am. J. Clin. Pathol. 116, 466–472 (2001)
K. Tsuta, D.C. Liu, N. Kalhor, I.I. Wistuba, C.A. Moran, Using the mitosis-specific marker anti-phosphohistone H3 to assess mitosis in pulmonary neuroendocrine carcinomas. Am. J. Clin. Pathol. 136, 252–259 (2011)
A. Warth, L. Fink, A. Fisseler-Eckhoff, D. Jonigk, M. Keller, G. Ott, R.J. Rieker, P. Sinn, S. Söder, A. Soltermann, K. Willenbrock, W. Weichert, Pulmonary Pathology Working Group of the German Society of Pathology: interobserver agreement of proliferation index (Ki-67) outperforms mitotic count in pulmonary carcinoids. Virchows Arch. 462, 507–513 (2013)
P.L. Filosso, A. Oliaro, E. Ruffini, G. Bora, P. Lyberis, S. Asioli, L. Delsedime, A. Sandri, F. Guerrera, Outcome and prognostic factors in bronchial carcinoids: a single-center experience. J. Thorac. Oncol. 8, 1282–1288 (2013)
E. Aydin, U. Yazici, M. Gulgosteren, Y. Agackiran, S. Kaya, E. Gulhan, I. Tastepe, N. Karaoglanoglu, Long-term outcomes and prognostic factors of patients with surgically treated pulmonary carcinoid: our institutional experience with 104 patients. Eur. J. Cardiothorac. Surg. 39, 549–554 (2011)
C. Cao, T.D. Yan, C. Kennedy, N. Hendel, P.G. Bannon, B.C. McCaughan, Bronchopulmonary carcinoid tumors: long-term outcomes after resection. Ann. Thorac. Surg. 91, 339–343 (2011)
M.A. Cañizares, J.M. Matilla, A. Cueto, J. Algar, I. Muguruza, N. Moreno-Mata, R. Moreno-Balsalobre, R. Guijarro, R. Arrabal, E. Garcia-Fontan, A. Gonzalez-Piñeiro, M. Garcia-Yuste, EMETNE-SEPAR Members: Atypical carcinoid tumours of the lung: prognostic factors and patterns of recurrence. Thorax 69, 648–653 (2014)
N. Daddi, M. Schiavon, P.L. Filosso, G. Cardillo, M.C. Ambrogi, A. De Palma, L. Luzzi, A. Bandiera, C. Casali, A. Ruffato, V. De Angelis, L.G. Andriolo, F. Guerrera, F. Carleo, F. Davini, M. Urbani, S. Mattioli, U. Morandi, P. Zannini, G. Gotti, M. Loizzi, F. Puma, A. Mussi, A. Ricci, A. Oliaro, F. Rea, Multi-Institutional Italian Pathology Group: Prognostic factors in a multicentre study of 247 atypical pulmonary carcinoids. Eur. J. Cardiothorac. Surg. 45, 677–686 (2014)
R. Johnson, S. Trocha, M. McLawhorn, M. Worley, G. Wheeler, L. Thompson, N. Schisler, D. Schammel, C. Schammel, J. Stephenson, W. Bolton, Histology, not lymph node involvement, predicts long-term survival in bronchopulmonary carcinoids. Am. Surg. 77, 1669–1674 (2011)
G. Pelosi, G. Rindi, W.D. Travis, M. Papotti, Ki-67 antigen in lung neuroendocrine tumors: unraveling a role in clinical practice. J. Thorac. Oncol. 9, 273–284 (2014)
T. Zahel, S. Krysa, E. Herpel, A. Stenzinger, B. Goeppert, P. Schirmacher, H. Hoffmann, P.A. Schnabel, A. Warth, Phenotyping of pulmonary carcinoids and a Ki-67-based grading approach. Virchows Arch. 460, 299–308 (2012)
G. Rindi, C. Klersy, F. Inzani, G. Fellegara, L. Ampollini, A. Ardizzoni, N. Campanini, P. Carbognani, T.M. De Pas, D. Galetta, P.L. Granone, L. Righi, M. Rusca, L. Spaggiari, M. Tiseo, G. Viale, M. Volante, M. Papotti, G. Pelosi, Grading the neuroendocrine tumors of the lung: an evidence-based proposal. Endocr. Relat. Cancer 21, 1–16 (2014)
F. Grimaldi, D. Muser, C.A. Beltrami, P. Machin, A. Morelli, S. Pizzolitto, G. Talmassons, F. Marciello, A.A. Colao, R. Monaco, G. Monaco, A. Faggiano, Partitioning of bronchopulmonary carcinoids in two different prognostic categories by Ki-67 score. Front Endocrinol. (Lausanne) 2, 20 (2011)
A.E. Walts, D. Ines, A.M. Marchevsky, Limited role of Ki-67 proliferative index in predicting overall short-term survival in patients with typical and atipica pulmonary carcinoid tumors. Mod. Pathol. 25, 1258–1264 (2012)
K. Tsuta, M.G. Raso, N. Kalhor, D.D. Liu, I.I. Wistuba, C.A. Moran, Histologic features of low- and intermediate-grade neuroendocrine carcinoma (typical and atipica carcinoid tumors) of the lung. Lung Cancer 71, 34–41 (2011)
P. Perkins, B.L. Kemp, J.B. Putnam Jr, J.D. Cox, Pretreatment characteristics of carcinoid tumors of the lung which predict aggressive behavior. Am. J. Clin. Oncol. 20, 285–288 (1997)
S.Y. Ha, J.J. Lee, J. Cho, J. Hyeon, J. Han, H.K. Kim, Lung parenchymal invasion in pulmonary carcinoid tumor: an important histologic feature suggesting the diagnosis of atypical carcinoid and poor prognosis. Lung Cancer 80, 146–152 (2013)
Acknowledgments
This study was partially supported by a grant from the Italian Association for Cancer Research (AIRC) (grant nr. IG 13567 to MV).
Conflict of interest
All Authors declare the absence of any potential conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Volante, M., Gatti, G. & Papotti, M. Classification of lung neuroendocrine tumors: lights and shadows. Endocrine 50, 315–319 (2015). https://doi.org/10.1007/s12020-015-0578-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-015-0578-x